Advanced Therapy Medicinal Products Market Size and Trends 2026 to 2035
The advanced therapy medicinal products market size was exhibited at USD 42.04 billion in 2025 and is projected to hit around USD 193.75 billion by 2035, growing at a CAGR of 16.51% during the forecast period 2026 to 2035.
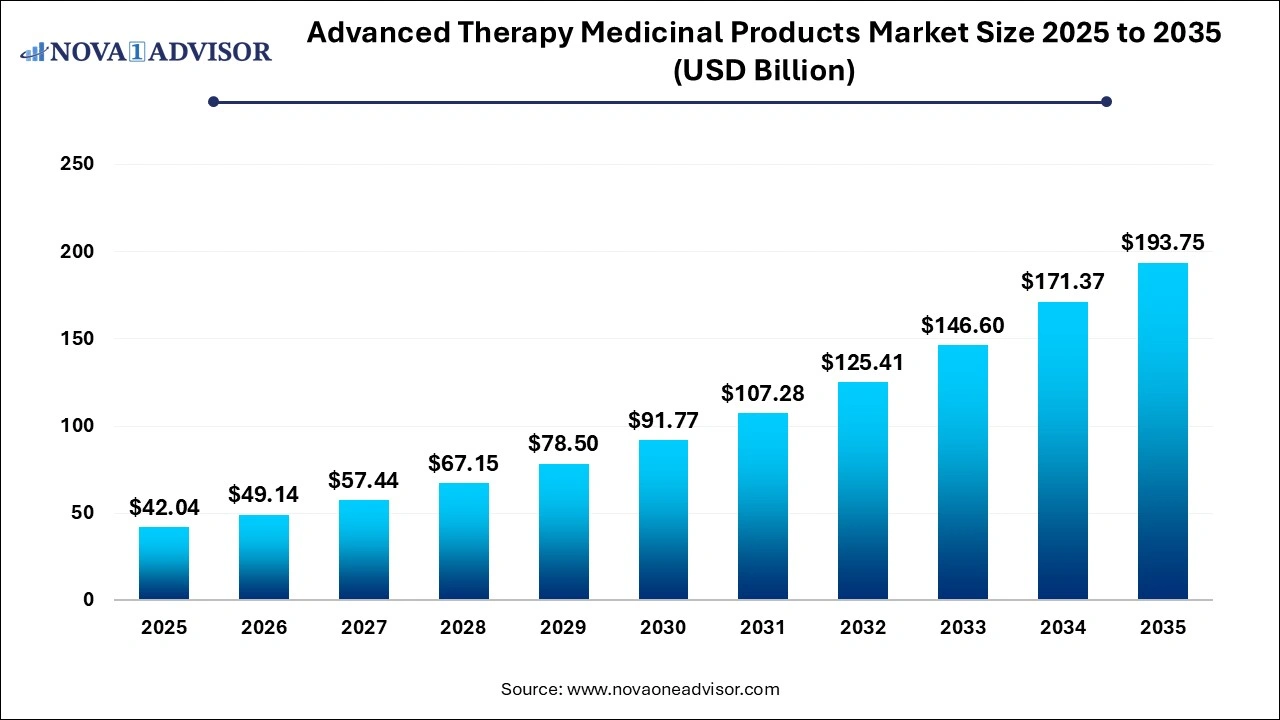
Advanced Therapy Medicinal Products Market Key Takeaways:
- North America dominated the advanced therapy medicinal products industry and accounted for the largest revenue share of 45.0% in 2025.
- Asia Pacific is expected to grow at the fastest CAGR of 21.4%during the forecast period.
Advanced Therapy Medicinal Products Market Outlook
- Market Growth Overview: The advanced therapy medicinal products market is expected to grow significantly between 2026 and 2035, driven by technological advancements in gene and cell therapy, the rising prevalence of chronic and genetic diseases, and increased investment and strategic collaborations.
- Sustainability Trends: Sustainability trends involve manufacturing scalability and automation, environmental sustainability, economic sustainability, and market access.
- Major Investors: Major investors in the market include Novartis AG, Gilead Sciences, Inc. (Kite Pharma), Bristol Myers Squibb (BMS), Pfizer Inc., and Bluebird Bio, Inc.
Advanced Therapy Medicinal Products Market Overview
The advanced therapy medicinal products (ATMP) market represents a revolutionary frontier in the global healthcare industry, merging biotechnology, regenerative medicine, and personalized therapies. ATMPs encompass cell therapies, gene therapies, and tissue-engineered products, offering curative potential for previously intractable diseases such as rare genetic disorders, certain cancers, and degenerative conditions.
Unlike conventional pharmaceutical that primarily manage symptoms, ATMPs aim to repair, replace, or regenerate diseased tissues or genetic anomalies at a fundamental level. Landmark approvals of products like CAR-T cell therapies, gene replacement therapies (e.g., Luxturna, Zolgensma), and bioengineered skin grafts have validated ATMPs’ clinical promise.
Rising incidences of genetic disorders, cancers, and chronic diseases, coupled with breakthroughs in stem cell research, CRISPR gene-editing technologies, and biomaterials science, have created an unprecedented innovation ecosystem. Regulatory agencies such as the FDA (U.S.) and EMA (Europe) have established specialized pathways (e.g., Regenerative Medicine Advanced Therapy – RMAT designation) to accelerate the clinical development and commercialization of ATMPs.
Despite technical and regulatory challenges, massive investments by biopharmaceutical giants, venture capitalists, and government bodies are fueling rapid market expansion. ATMPs are poised not just to transform therapeutic paradigms but also to reshape the economics of healthcare in the coming decades.
How AI is Impacting the Advanced Therapy Medicinal Products Market
AI and ML are transforming the advanced therapy medicinal products industry by accelerating the discovery, design, and manufacturing of complex gene and cell therapies. In R&D, AI algorithms significantly speed up target identification and optimize viral vector engineering, which are critical for developing effective, customized therapies. In manufacturing, AI and ML facilitate digital twins and real-time monitoring to optimize production processes, enhancing yield and consistency. These technologies streamline clinical trials by improving patient recruitment through rapid data analysis and predicting patient-specific responses.
Supply Chain Analysis of the Advanced Therapy Medicinal Products Market
- R&D and Discovery: This initial stage involves foundational research, target identification, vector engineering (for gene therapies), and preclinical studies.
Key Players: Spark Therapeutics, UniQure N.V., and CRISPR Therapeutics.
- Clinical Development & Regulatory Affairs: This phase involves translating research into human trials, requiring strict adherence to Good Clinical Practice (GCP) and navigating regulatory pathways like the FDA’s RMAT or EMA’s PRIME.
Key Players: PharmaLex (AmerisourceBergen), Voisin Consulting Life Sciences (VCLS), and QBD Group.
- Manufacturing and Quality Control: This stage involves specialized, sterile manufacturing of viral vectors and cell modification (GMP facilities), which is frequently outsourced to contract development and manufacturing organizations (CDMOs).
Key Players: Lonza Group AG, Catalent, Inc., WuXi Advanced Therapies, FUJIFILM Diosynth Biotechnologies, and AGC Biologics.
Major Trends in the Advanced Therapy Medicinal Products Market
-
Increased Approvals of Gene and Cell Therapies: Momentum gaining with a growing number of ATMPs securing regulatory green lights.
-
Expansion of Allogeneic ("Off-the-Shelf") Therapies: Addressing manufacturing and scalability challenges of autologous therapies.
-
Advances in CRISPR and Gene-editing Technologies: Facilitating the development of next-generation gene therapies.
-
Adoption of Innovative Manufacturing Techniques: Automation, closed-system processing, and modular manufacturing platforms scaling ATMP production.
-
Emergence of Personalized Regenerative Medicine: Tailoring ATMPs to individual genetic or molecular profiles.
-
Strategic Partnerships and M&A Activity: Biopharma consolidations aimed at building robust ATMP pipelines.
-
Increased Investment in Supply Chain Infrastructure: Cold chain logistics and GMP-compliant facilities expanding globally.
-
Focus on Rare and Orphan Diseases: ATMP developers increasingly targeting smaller patient populations with high unmet needs.
Report Scope of Advanced Therapy Medicinal Products Market
| Report Coverage |
Details |
| Market Size in 2026 |
USD 49.14 Billion |
| Market Size by 2035 |
USD 193.75 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 16.51% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Therapy Type |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Covered |
North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled |
Spark Therapeutics, Inc.; Bluebird Bio, Inc.; Novartis AG; UniQure N.V.; Celgene Corporation; Gilead Lifesciences, Inc.; Kolon TissueGene, Inc.; JCR Pharmaceuticals Co., Ltd.; MEDIPOST; Vericel Corporation; PHARMICELL Co., Ltd; Organogenesis Inc. |
Advanced Therapy Medicinal Products Market Dynamics
Driver
Rising Demand for Curative Therapies for Rare Diseases and Cancers
The primary driver accelerating the ATMP market is the growing demand for curative therapies addressing rare diseases and oncology indications.
Rare genetic disorders and aggressive cancers often have limited or no treatment options, leaving patients with unmet medical needs. ATMPs such as gene replacement therapies for spinal muscular atrophy (SMA) or CAR-T therapies for refractory leukemias offer durable or even curative outcomes by addressing root causes rather than merely controlling symptoms.
Patient advocacy, regulatory incentives like orphan drug designation, and high reimbursement rates for successful ATMPs are creating strong commercial incentives. Consequently, the ATMP pipeline is expanding rapidly, with hundreds of clinical trials underway globally, spanning indications such as hemophilia, retinal dystrophy, glioblastoma, and Duchenne muscular dystrophy.
Restraint
High Costs and Complex Manufacturing Processes
Despite the promise of ATMPs, high treatment costs and complex manufacturing processes remain significant barriers to widespread adoption.
Manufacturing ATMPs, especially autologous cell Therapy, involves individualized, labor-intensive procedures requiring stringent GMP compliance, cold chain management, and high process variability. The cost of production often translates into extraordinarily high list prices—upwards of USD 500,000 to USD 2 million per treatment creating affordability and reimbursement challenges even in high-income markets.
Additionally, scaling up manufacturing while ensuring product consistency and regulatory compliance is a significant hurdle, particularly for early-stage biotech companies. Solving these challenges through technological innovations, centralized production hubs, or allogeneic therapy platforms will be crucial for sustainable market growth.
Opportunity
Expansion of Allogeneic and Off-the-Shelf ATMPs
A transformative opportunity lies in the expansion of allogeneic (“off-the-shelf”) ATMPs, which can be manufactured in large batches and stored until needed, overcoming many limitations of autologous (patient-specific) therapies.
Allogeneic stem cells, T-cells, and engineered tissues derived from healthy donors offer scalability, cost reduction, and broader patient access. Companies are increasingly investing in genome editing technologies to reduce immunogenicity and graft-versus-host risks in allogeneic therapies.
For example, the approval of allogeneic CAR-T therapies under investigation by companies like Allogene Therapeutics and Celyad Oncology indicates a shift towards more commercially viable ATMP models. Allogeneic approaches will likely dominate future pipelines in oncology, regenerative orthopedics, and rare disease therapeutics.
Advanced Therapy Medicinal Products Market Segmental Insights
By Therapy Type Insights
Cell therapy dominates the therapy type segment, holding the largest share owing to the early clinical success and commercial approvals of therapies like Novartis' Kymriah and Gilead’s Yescarta. Cell therapies involve modifying or transplanting live cells to replace damaged tissues or reprogram immune responses, particularly in cancer treatment (CAR-T), orthopedic applications, and cardiovascular regeneration.
Gene therapy is growing fastest, fueled by breakthroughs in delivery vectors (AAVs, lentivirus), gene-editing platforms (CRISPR/Cas9), and regulatory approvals such as Zolgensma (SMA) and Luxturna (retinal dystrophy). Gene Therapy promise single-administration cures, transforming patient outcomes and drawing substantial investment attention, particularly for monogenic disorders and inherited blindness.
Advanced Therapy Medicinal Products Market By Regional Insights
Europe currently leads the ATMP market, supported by a strong regulatory framework under the European Medicines Agency (EMA) and the Committee for Advanced Therapies (CAT), which specifically addresses ATMP evaluation and approval.
Countries like Germany, the UK, Spain, and France have active clinical trial ecosystems, sophisticated healthcare infrastructures, and favorable reimbursement policies for ATMPs, especially under health technology assessment (HTA) frameworks for rare diseases. Initiatives like the European Advanced Therapies Investor Day and Horizon Europe funding further boost Europe's leadership.
Europe’s experience in early approvals (e.g., Holoclar, an autologous stem cell therapy for corneal burns) and collaborative regulatory models (e.g., PRIME designation) continue to make it a preferred region for ATMP development and commercialization.
Asia-Pacific is witnessing the fastest growth, driven by increasing investments in regenerative medicine, expanding healthcare infrastructure, and rising demand for innovative therapeutics.
China, Japan, South Korea, and Australia are investing heavily in stem cell research, gene editing technologies, and ATMP manufacturing capabilities. Regulatory agencies like Japan's Pharmaceuticals and Medical Devices Agency (PMDA) have introduced expedited pathways for regenerative medicine products, accelerating market entry.
Moreover, the rising prevalence of genetic diseases, coupled with growing affordability and government support, positions Asia-Pacific as a key ATMP growth engine for the coming decade.
U.K Advanced Therapy Medicinal Products Market Trends
The U.K. is leveraging the cell and gene therapy catapult and ATTC network to create a seamless infrastructure for clinical trials and commercialization. By shifting focus from rare disorders to common chronic diseases, such as diabetes, these firms are exponentially expanding the therapeutic reach of viral vectors. Significant CDMO capacity expansion and heavy outsourcing are solving the manufacturing complexities of next-generation ATMPs, while the MHRA's accelerated regulatory frameworks reduce the time-to-market.
China Advanced Therapy Medicinal Products Market Trends
China’s shift from small molecules to advanced biological modalities is turning the region into a high-speed engine for global biotech innovation. By leveraging the NMPA’s streamlined priority reviews, these firms are accelerating the R&D cycle for innovative oncology products, positioning China as a global leader in personalized medicine. In response to geopolitical pressures like the BIOSECURE Act, startups are securing growth through self-reliant supply chain strategies and robust domestic manufacturing.
Key Players in the Advanced Therapy Medicinal Products Market
- Spark Therapeutics, Inc.: Spark Therapeutics pioneered gene therapy by gaining FDA approval for Luxturna, the first adeno-associated viral (AAV) vector gene therapy for a genetic disease in the US.
- Bluebird Bio, Inc.: Bluebird Bio is a leader in ex-vivo gene therapy, utilizing lentiviral vectors to treat severe genetic disorders like sickle cell disease and beta-thalassemia. Their FDA-approved products, including ZYNTEGLO and LYFGENIA, represent a major shift toward one-time functional cures for rare diseases.
- Novartis AG: Novartis is a major player in the ATMP market through its early adoption and commercialization of CAR-T cell therapy, notably Kymriah for leukemia, and the gene therapy Zolgensma for spinal muscular atrophy.
- UniQure N.V.: UniQure specializes in AAV-based gene therapies for rare diseases, successfully developing and partnering for the commercialization of HEMGENIX for hemophilia B.
- Celgene Corporation: Now part of Bristol-Myers Squibb, Celgene has historically played a critical role in the advancement of cellular therapies, particularly CAR-T and TCR technologies, in hematology. Their portfolio and collaborations, such as with Bluebird Bio in earlier oncology research, have been instrumental in pushing forward immune-based cancer treatments.
Recent Developments
-
March 2025: Novartis announced plans to expand its manufacturing capacity for Kymriah CAR-T therapy with a new facility in Spain, boosting European supply chain resilience.
-
February 2025: Bluebird Bio received FDA approval for Skysona, a gene therapy for cerebral adrenoleukodystrophy, marking a major milestone in rare neurogenetic disorders.
-
January 2025: CRISPR Therapeutics and Vertex Pharmaceuticals initiated Phase 3 trials of their CRISPR-based gene therapy CTX001 for sickle cell disease and beta-thalassemia.
-
December 2024: Orchard Therapeutics launched Libmeldy commercially in Europe, the first ex vivo gene therapy for metachromatic leukodystrophy (MLD).
-
November 2024: Celyad Oncology received fast-track designation from the FDA for CYAD-211, an allogeneic CAR-T therapy for multiple myeloma.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2026 to 2035. For this study, Nova one advisor, Inc. has segmented the advanced therapy medicinal products market
By Therapy Type
-
- Stem Cell Therapy
- Non-stem Cell Therapy
- Gene Therapy
- Tissue Engineered Product
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)


