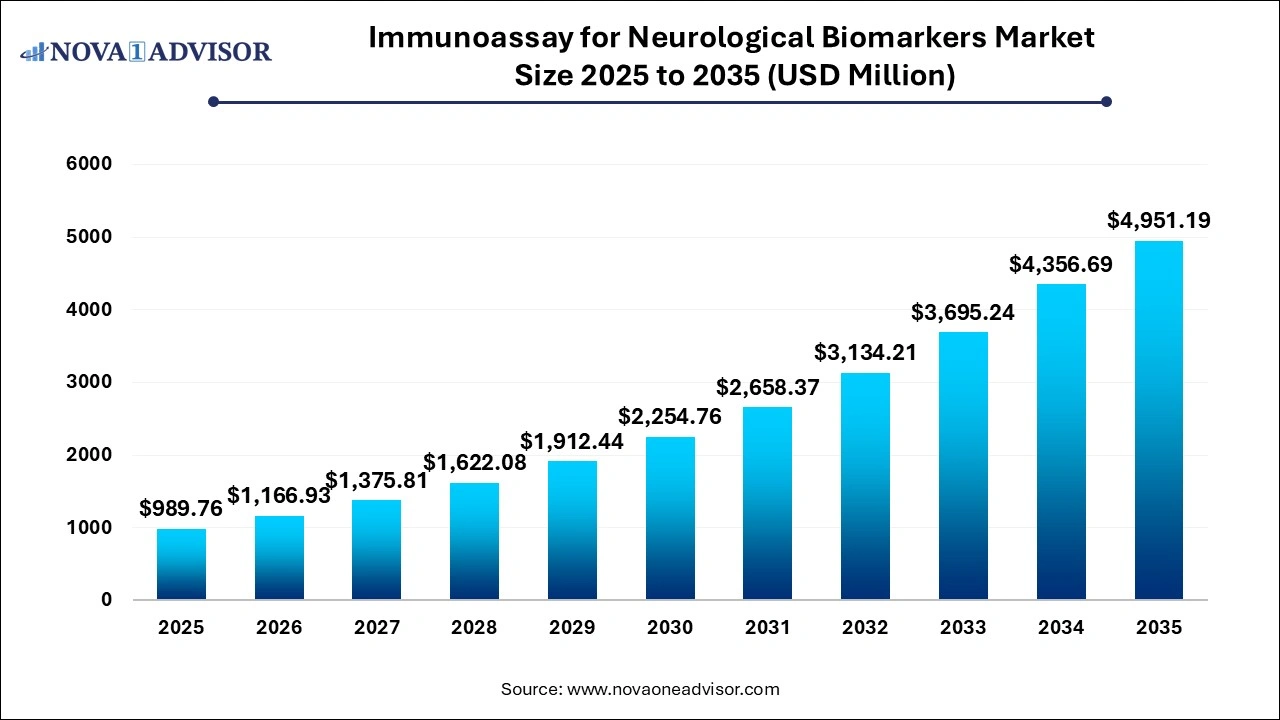
The immunoassay for neurological biomarkers market size was exhibited at USD 989.76 million in 2025 and is projected to hit around USD 4,951.19 million by 2035, growing at a CAGR of 17.47% during the forecast period 2026 to 2035.
| Report Coverage | Details |
| Market Size in 2026 | USD 1,166.93 Million |
| Market Size by 2035 | USD 4,951.19 Million |
| Growth Rate From 2026 to 2035 | CAGR of 17.47% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | By Product, By Disease, By Application |
| Market Analysis (Terms Used) | Value (USD Million/Billion) or (Volume/Units) |
| Regional scope | North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled | QIAGEN; Abbott; Merck & Co., Inc.; Johnson & Johnson Services, Inc.; Thermo Fisher Scientific, Inc.; Bio-Rad Laboratories, Inc.; Sysmex Corporation; Merck KGaA; F. Hoffmann La-Roche Ltd.; Nimble Therapeutics |
Immunoassays are protein-based tests that work by quantification and detection of antigen–antibodies interactions. These assays play a crucial role in diagnosis and treatment planning for a variety of neurological diseases. In recent years, there has been a significant rise in investments in the R&D of treatments for neurological diseases, which is expected to accelerate the market growth during the forecast period.
Furthermore, rising prevalence of neurological diseases such as Parkinson's disease, Alzheimer's Disease (AD), dementia, brain tumors, epilepsy, and others present significant unmet needs. AD is a global epidemic. According to Alzheimer's Association, approximately 55.0 million individuals globally have dementia, which is anticipated to rise to 98.0 million by 2030 and around 139.0 million by 2050. Furthermore, approximately 6.5 million individuals in the U.S., will be living with AD in 2024. Therefore, the demand for biomarkers is increasing for drug development, diagnosis, disease progression and research in neurological diseases.
The development of therapeutics used for neurological diseases has been challenging, mostly due to the blood-brain barrier. The key players are focusing on new therapies to prevent, defer, slow, or improve the symptoms of neurological diseases. Companies such as Biogen, AbbVie, AC Immune, Novartis AG, TauRx, Eli Lilly & Co., and others are highly active in developing novel therapies for the treatment of AD. Many market players have their candidate drugs in phase lll, which may be launched during the forecast period. Thus, increasing focus on the development of therapeutics for neurological diseases is expected to boost the demand of immunoassays for neurological biomarkers.
Moreover, key players engaged in the development of neurological biomarkers and partnerships between international companies are expected to fuel the market growth. Government initiatives such as investments in R&D of high-quality, cost-effective & standardized products for diagnosis and treatment of neurodegenerative disease research is also expected to drive market growth. For instance, in November 2021, the Department of Health and Social Care announced an investment of USD 456.62 million in neurodegenerative research in the UK. Moreover, the use of clinical data informatics platforms by productive research organizations is anticipated to propel market growth in the coming years.
The reagents segment held the largest share of 66.78% in 2025 in the immunoassays for neurological biomarkers. The market is witnessing a shift from traditional reagents to ultrasensitive reagents, which is driving the segment. For instance, the use of single-molecule counting assay kits for detecting neurological conditions such as multiple sclerosis and Alzheimer’s disease is significantly increasing. Furthermore, the approval and launch of novel immunoassay reagents are expected to fuel segment growth.
The instruments segment is anticipated at a CAGR of 17.6% during the forecast period. Increasing approval and the launch of immunoassay instruments are expected to fuel the segment's growth. For instance, in December 2025, the FDA approved Roche’s AD assay employing cobas immunoassay analyzer. Similarly, in July 2024, FDA designated Elecsys Amyloid Plasma Panel as a breakthrough device that allows the measurement of Alzheimer’s disease biomarkers from a blood sample.
The Alzheimer’s disease segment held the largest share of 38.04% in 2025. A rise in the prevalence of AD is expected to drive the market. The percentage of people with Alzheimer's dementia increases with age. Thus, this disease mostly occurs in people aged 65 and above. According to Alzheimer’s Association, globally, approximately 6.5 million individuals are living with Alzheimer’s disease in the U.S. as of 2024. Owing to the increasing prevalence of AD, government and nonprofit organizations are providing a plethora of research opportunities, which may fuel market growth.
The Alzheimer’s disease segment is expected to expand at the highest CAGR of 18.4% during the forecast period. Key players are strategically undertaking initiatives to increase their share with the development of advanced biomarker technology for diagnostics and treatment research. For instance, in March 2024, Quanterix signed a licensing agreement with Eli Lilly and Company to use pTau 217 for near-term clinical research and in long term for IVD applications.
The FDA granted breakthrough device designation to Quanterix’s Simoa pTau-181 assay for the diagnosis of Alzheimer’s disease. Thus, such developments are expected to govern the future of Simoa assay development across diseases like Alzheimer’s disease and drive the immunoassay for neurological biomarkers market.
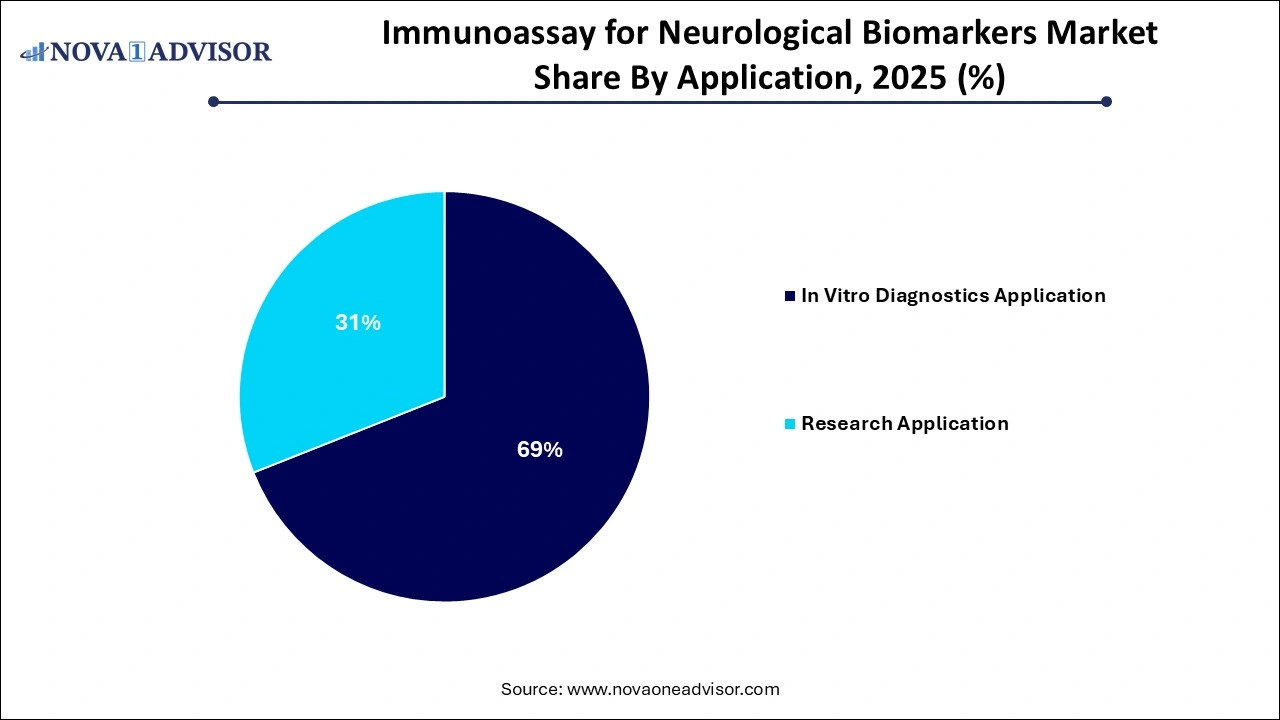
The research application segment captured the highest revenue share of 69% in 2025. Immunoassay plays a crucial role in neurological R&D; it is being used to determine pharmacokinetics of biologics and measure biomarkers & the levels of host cell proteins. Moreover, researchers are involved in identifying key biomarkers in serum and plasma to develop assays that are highly sensitive for determining the low levels of biomarkers.
Merck’s Single Molecule Counting, an immunoassay kit, is one such example, which is highly sensitive in measuring neurological proteins in samples. All these factors are increasing the number of research organizations working on biomarker research & validation; pharmaceutical companies that are using biomarkers for drug discovery & development; and ambulatory surgery centers, thereby driving market growth.
Furthermore, in vitro diagnostics application segment is anticipated to expand at rapid pace with a CAGR of 23.9% during the forecast period. The rising number of disease-modifying therapies for neurological disorders is boosting the demand for biomarker testing in the market. For instance, in June 2021, FDA granted accelerated approval to Aduhelm indicated for Alzheimer’s disease. Furthermore, the government is undertaking various initiatives to provide better facilities, such as reimbursement for neurological diagnostic tests, which is likely to drive the market. Many healthcare institutions are working with laboratories to integrate different tests.
North America held the largest share of 49% in the immunoassay for neurological biomarkers market in 2025. Owing to the increased healthcare expenditure, rise in customer awareness about the use of biomarkers in diagnosing neurological disorders, availability of technologically advanced products, proactive government initiatives, and improved healthcare infrastructure. Moreover, key players are focusing on new product launches of novel immunoassays for the identification of neurological biomarkers.
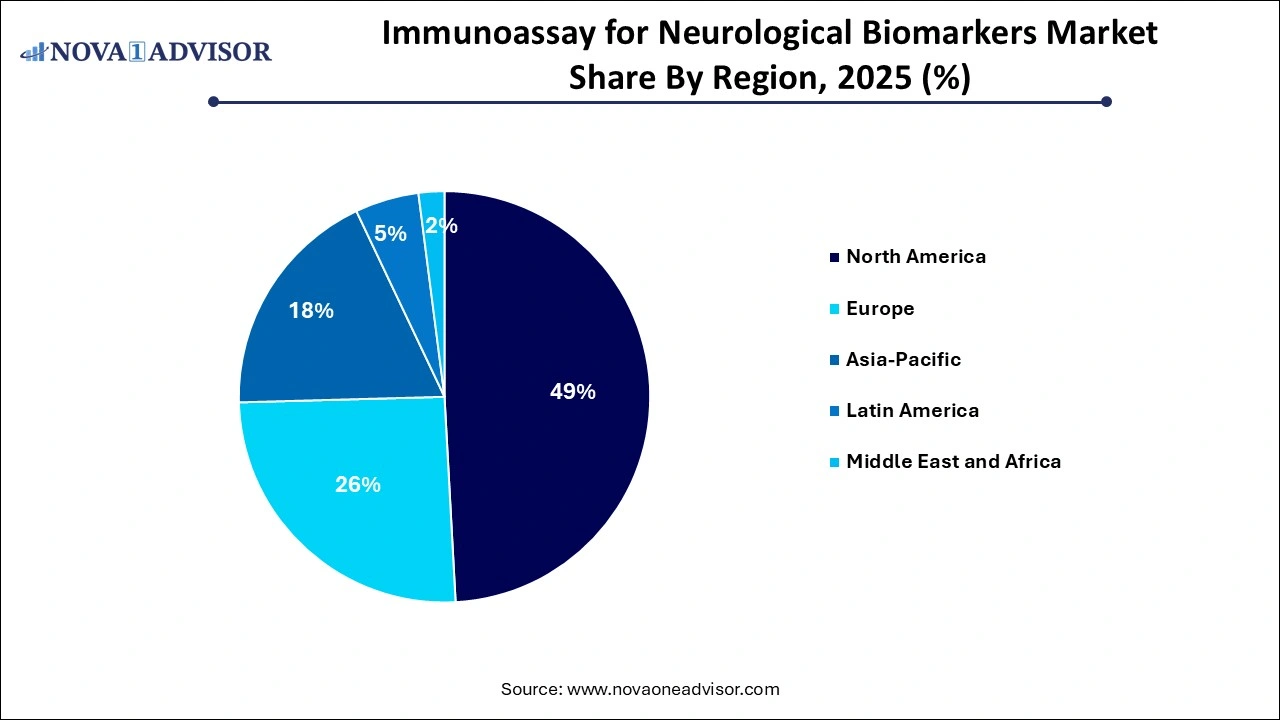
Asia Pacific is estimated to expand at the fastest CAGR of 18.6% from 2026-2034. This high growth can be attributed to the rising prevalence of neurological diseases and increasing number of clinical trials. The need for early diagnosis of neurological diseases to reduce treatment costs & mortality and abundance of raw materials for manufacturing immunoassay products can increase manufacturer interest. These factors are expected to fuel the market demand in Asia Pacific. In addition, increasing healthcare investments in countries, such as China and India, is also positively influencing market growth.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2026 to 2035. For this study, Nova one advisor, Inc. has segmented the immunoassay for neurological biomarkers market
By Product
By Disease
By Application
By Regional