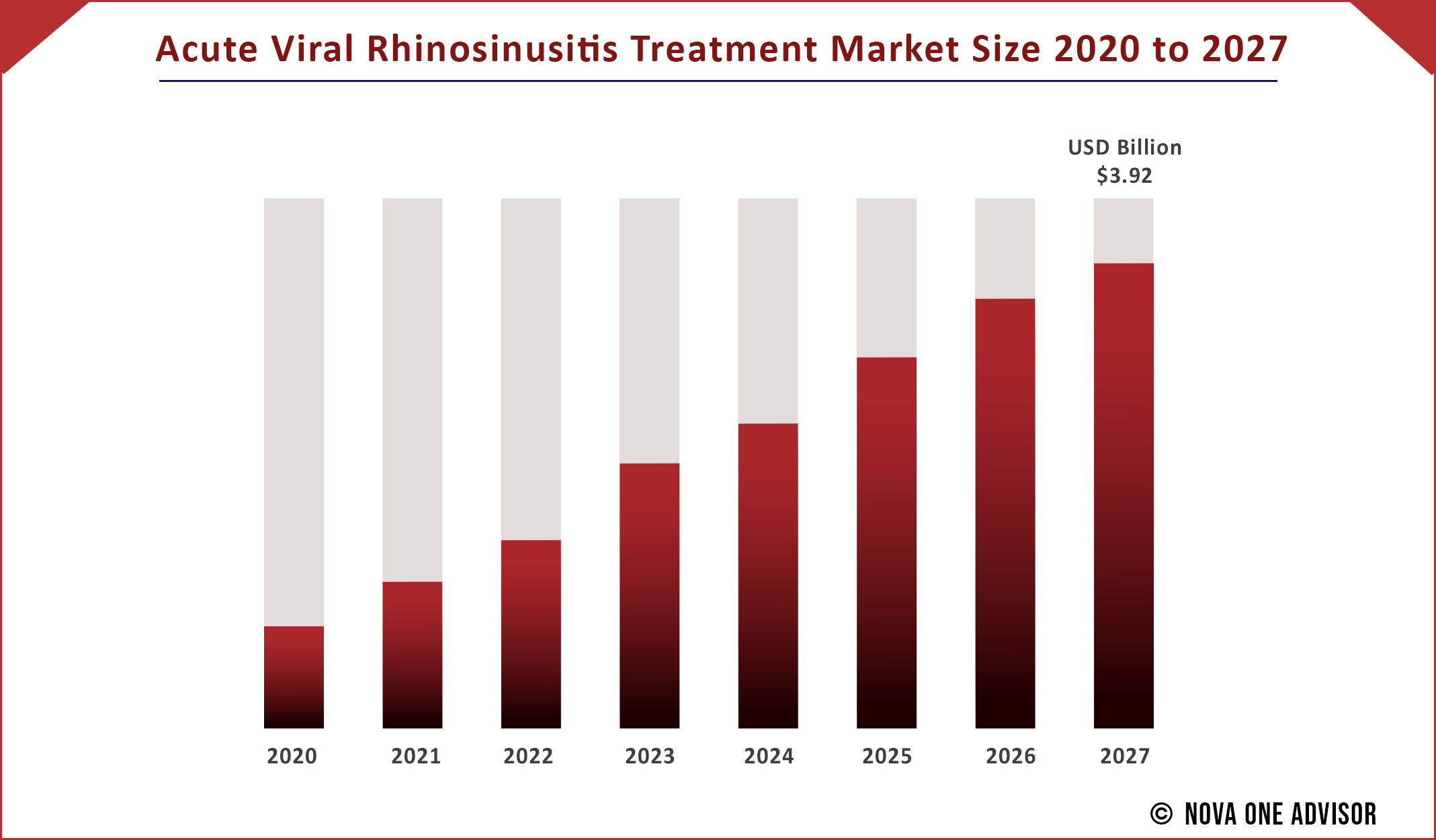
The global Acute Viral Rhinosinusitis Treatment market gathered revenue around USD 2.0 Billion in 2020 and market is set to grow USD 3.92 Billion by the end of 2027 and is estimated to expand at a modest CAGR of 5.0% during the prediction period 2021 to 2027.
The term acute rhinosinusitis (ARS) refers to a symptomatic inflammation of the nasal cavity and paranasal sinuses that lasts less than four weeks. As inflammation of the sinuses seldom occurs without infection of the nasal mucosa, the name rhinosinusitis is favored over sinusitis. A viral infection is the most prevalent cause of ARS. The global acute viral rhinosinusitis treatment market is anticipated to be driven by increase in rhinosinusitis cases.
North America dominated the global acute viral rhinosinusitis treatment market in 2020, and the trend is anticipated to continue during the forecast period. Acute viral rhinosinusitis is characterized by rapid exacerbation or emergence of symptoms in a patient with chronic sinusitis, which is usually accompanied with purulence draining from the sinuses as shown on nasal endoscopy.
Surge in healthcare expenditure and rise in awareness about sinusitis are expected to fuel the growth of the market in Asia Pacific. The market in the region is likely to be driven by increase in investment in R&D of pharmaceutical drugs.
The market in Europe is projected to be driven by increase in demand for drugs for the treatment of acute viral rhinosinusitis. Additionally, increase in awareness about acute viral rhinosinusitis and focus on discovering novel treatment drugs are anticipated to propel the market in the region.
This research report purposes at stressing the most lucrative growth prospects. The aim of the research report is to provide an inclusive valuation of the Acute Viral Rhinosinusitis Treatment market and it encompasses thoughtful visions, actualities, industry-validated market findings, historic data, and prognoses by means of appropriate set of assumptions and practice. Global Acute Viral Rhinosinusitis Treatment market report aids in comprehending market structure and dynamics by recognizing and scrutinizing the market sectors and predicted the global market outlook.
COVID-19 Impact Assessment on Market Landscape
The report comprises the scrutiny of COVID-19 lock-down impact on the income of market leaders, disrupters and followers. Since lock down was instigated differently in diverse regions and nations, influence of same is also dissimilar across various industry verticals. The research report offers present short-term and long-term influence on the market to assist market participants across value chain makers to formulate the framework for short term and long-lasting tactics for recovery and by region.
Acute Viral Rhinosinusitis Treatment market report empowers readers with all-inclusive market intelligence and offers a granular outline of the market they are operational in. Further this research study delivers exceptional combination of tangible perceptions and qualitative scrutiny to aid companies accomplishes sustainable growth. This report employs industry-leading research practices and tools to assemble all-inclusive market studies, intermingled with pertinent data. Additionally, this report also emphases on the competitive examination of crucial players by analyzing their product portfolio, pricing, gross margins, financial position, growth approaches, and regional occurrence.
Scope of the Report
This report provides an all-inclusive environment of the analysis for the Acute Viral Rhinosinusitis Treatment Market. The market estimates provided in the report are the result of in-depth secondary research, primary interviews and in-house expert reviews. These market estimates have been considered by studying the impact of various social, political and economic factors along with the current market dynamics affecting the Acute Viral Rhinosinusitis Treatment Market growth
Along with the market overview, which comprises of the market dynamics the chapter includes a Porter’s Five Forces analysis which explains the five forces: namely buyers bargaining power, suppliers bargaining power, threat of new entrants, threat of substitutes, and degree of competition in the Acute Viral Rhinosinusitis Treatment Market. It explains the various participants, such as system integrators, intermediaries and end-users within the ecosystem of the market. The report also focuses on the competitive landscape of the Acute Viral Rhinosinusitis Treatment Market.
| Report Highlights |
Details |
| Market Size |
US$ 3.92 Billion by 2027 |
| Growth Rate |
CAGR of 5.0% From 2021 to 2027 |
| Base Year |
2020 |
| Historic Data |
2017 to 2020 |
| Forecast Period |
2021 to 2027 |
| Segments Covered |
Treatment,,Route of Administration,Distribution Channel,Infection Type,Region |
| Regional Scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa (MEA) |
| Companies Mentioned |
Sun Pharmaceutical Industries, Inc., Pfizer, Inc., Janssen Pharmaceuticals, Inc. (Johnson & Johnson), Fresenius Kabi USA, LLC, Dr. Reddy's Laboratories, Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Sanofi, Abbott Laboratories, Bayer AG, Eli Lilly and Company, Bristol-Myers Squibb, Merck & Co, Inc. |
Competitive Rivalry
Foremost players in the market are attentive on adopting corporation strategies to enhance their market share. Some of the prominent tactics undertaken by leading market participants in order to sustain the fierce market completion include collaborations, acquisitions, substantial spending in R&D and the improvement of new-fangled products or reforms among others.
Major manufacturers & their revenues, percentage splits, market shares, growth rates and breakdowns of the product markets are determined through secondary sources and verified through the primary sources.
- Company Overview
- Company Market Share/Positioning Analysis
- Product Offerings
- Financial Performance
- Recent Initiatives
- Key Strategies Adopted by Players
- Vendor Landscape
- List of Suppliers
- List of Buyers
List of key Players in Acute Viral Rhinosinusitis Treatment Market Report are:
Sun Pharmaceutical Industries, Inc., Pfizer, Inc., Janssen Pharmaceuticals, Inc. (Johnson & Johnson), Fresenius Kabi USA, LLC, Dr. Reddy's Laboratories, Inc., Novartis AG, Teva Pharmaceutical Industries Ltd., Sanofi, Abbott Laboratories, Bayer AG, Eli Lilly and Company, Bristol-Myers Squibb, Merck & Co, Inc., AstraZeneca plc, Amneal Pharmaceuticals LLC, GlaxoSmithKline plc.
Unravelling the Critical Segments
This research report offers market revenue, sales volume, production assessment and prognoses by classifying it on the basis of various aspects including product type, application/end-user, and region. Further, this research study investigates market size, production, consumption and its development trends at global, regional, and country level for period 2017 to 2027 and covers subsequent region in its scope:
Global Acute Viral Rhinosinusitis Treatment Market: Segmentation
- Acute Viral Rhinosinusitis Treatment Market, by Treatment
- Saline Nasal Spray
- Nasal Corticosteroids
- Fluticasone
- Budesonide
- Mometasone
- Others
- Decongestants
- Topical Decongestants
- Systemic Decongestants
- NSAIDs
- Acetaminophen
- Ibuprofen
- Aspirin
- Others
- Others
- Acute Viral Rhinosinusitis Treatment Market, by Route of Administration
- Topical
- Nasal
- Oral
- Injectable
- Global Acute Viral Rhinosinusitis Treatment Market, by Distribution Channel
- Online Pharmacies
- Retail Pharmacies
- Hospital Pharmacies
- Acute Viral Rhinosinusitis Treatment Market, by Infection Type
- Rhinovirus (Picornavirus)
- Non-Rhinovirus
- Influenza
- Enterovirus
- Adenovirus
- Others
By Geography
North America
Europe
- Germany
- France
- United Kingdom
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
Latin America
- Brazil
- Rest of Latin America
Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
Highlights of the Report:
- Market Penetration: Comprehensive information on the product portfolios of the top players in the market.
- Product Development/Innovation: Detailed insights on the upcoming technologies, R&D activities, and product launches in the market
- Competitive Assessment: In-depth assessment of the market strategies, geographic and business segments of the leading players in the market
- Market Development: Comprehensive information about emerging markets. This report analyzes the market for various segments across geographies
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the market
Research Methodology
A unique research methodology has been utilized by Nova one advisor to conduct comprehensive research on the growth of the global Acute Viral Rhinosinusitis Treatment market and arrive at conclusions on its growth prospects. This research methodology is a combination of primary and secondary research, which helps analysts warrant the accuracy and reliability of the drawn conclusions.
Nova one advisor employs comprehensive and iterative research methodology focused on minimizing deviance in order to provide the most accurate estimates and forecast possible. The company utilizes a combination of bottom-up and top-down approaches for segmenting and estimating quantitative aspects of the market. In Addition, a recurring theme prevalent across all our research reports is data triangulation that looks market from three different perspectives. Critical elements of methodology employed for all our studies include:
Preliminary data mining
Raw market data is obtained and collated on a broad front. Data is continuously filtered to ensure that only validated and authenticated sources are considered. In addition, data is also mined from a host of reports in our repository, as well as a number of reputed paid databases. For comprehensive understanding of the market, it is essential to understand the complete value chain and in order to facilitate this; we collect data from raw material suppliers, distributors as well as buyers.
Technical issues and trends are obtained from surveys, technical symposia and trade journals. Technical data is also gathered from intellectual property perspective, focusing on white space and freedom of movement. Industry dynamics with respect to drivers, restraints, pricing trends are also gathered. As a result, the material developed contains a wide range of original data that is then further cross-validated and authenticated with published sources.
Statistical model
Our market estimates and forecasts are derived through simulation models. A unique model is created customized for each study. Gathered information for market dynamics, technology landscape, application development and pricing trends is fed into the model and analyzed simultaneously. These factors are studied on a comparative basis, and their impact over the forecast period is quantified with the help of correlation, regression and time series analysis. Market forecasting is performed via a combination of economic tools, technological analysis, and industry experience and domain expertise.
Econometric models are generally used for short-term forecasting, while technological market models are used for long-term forecasting. These are based on an amalgamation of technology landscape, regulatory frameworks, economic outlook and business principles. A bottom-up approach to market estimation is preferred, with key regional markets analyzed as separate entities and integration of data to obtain global estimates. This is critical for a deep understanding of the industry as well as ensuring minimal errors. Some of the parameters considered for forecasting include:
- Market drivers and restrains, along with their current and expected impact
- Raw material scenario and supply v/s price trends
- Regulatory scenario and expected developments
- Current capacity and expected capacity additions up to 2027
We assign weights to these parameters and quantify their market impact using weighted average analysis, to derive an expected market growth rate.
Primary validation
This is the final step in estimating and forecasting for our reports. Exhaustive primary interviews are conducted, on face to face as well as over the phone to validate our findings and assumptions used to obtain them. Interviewees are approached from leading companies across the value chain including suppliers, technology providers, domain experts and buyers so as to ensure a holistic and unbiased picture of the market. These interviews are conducted across the globe, with language barriers overcome with the aid of local staff and interpreters. Primary interviews not only help in data validation, but also provide critical insights into the market, current business scenario and future expectations and enhance the quality of our reports. All our estimates and forecast are verified through exhaustive primary research with Key Industry Participants (KIPs) which typically include:
- Market leading companies
- Raw material suppliers
- Product distributors
- Buyers
The key objectives of primary research are as follows:
- To validate our data in terms of accuracy and acceptability
- To gain an insight in to the current market and future expectations
Secondary Validation
Secondary research sources referred to by analysts during the production of the global Acute Viral Rhinosinusitis Treatment market report include statistics from company annual reports, SEC filings, company websites, investor presentations, regulatory databases, government publications, and industry white papers. Analysts have also interviewed senior managers, product portfolio managers, CEOs, VPs, and market intelligence managers, who contributed to the production of Nova one advisor’s study on the Acute Viral Rhinosinusitis Treatment market as primary methods.
The study objectives of this report are:
- To analyze and study the global market capacity, production, value, consumption, status (2017-2020) and forecast (2021-2027);
- Focuses on the key manufacturers, to study the capacity, production, value, market share and development plans in future.
- Comprehensive company profiles covering the product offerings, key financial information, recent developments, SWOT analysis, and strategies employed by the major market players
- To define, describe and forecast the market by type, application and region.
- To analyze the global and key regions market potential and advantage, opportunity and challenge, restraints and risks.
- To identify significant trends and factors driving or inhibiting the market growth.
- To analyze the opportunities in the market for stakeholders by identifying the high growth segments.
- To strategically analyze each submarket with respect to individual growth trend and their contribution to the market
- To analyze competitive developments such as expansions, agreements, new product launches, and acquisitions in the market
- To strategically profile the key players and comprehensively analyze their growth strategies.


