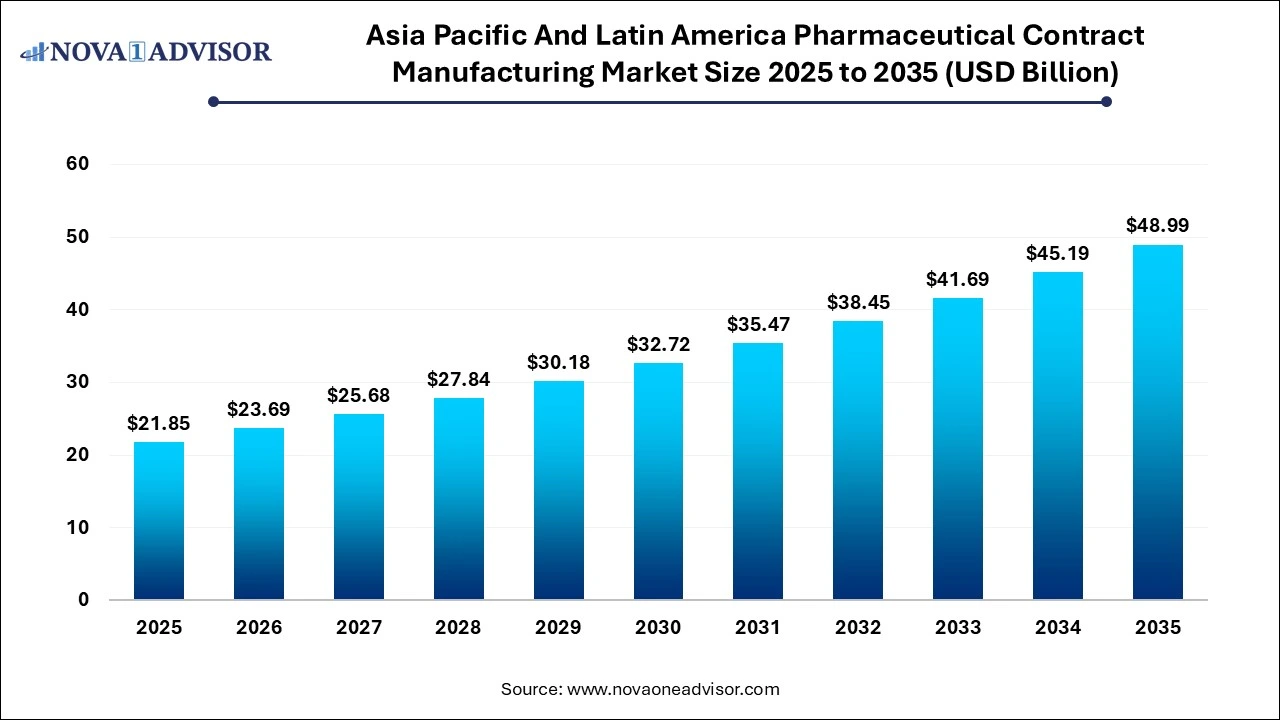
The Asia Pacific and Latin America pharmaceutical contract manufacturing market size was estimated at USD 21.85 billion in 2025 and is projected to increase from USD 23.69 billion in 2026 to approximately USD 48.99 billion by 2035, growing at a CAGR of 8.41% from 2026 to 2035. The Asia Pacific and Latin America pharmaceutical contract manufacturing market is driven by the increasing demand for complex medicines that require specialized manufacturing capabilities that CDMOs provide, growing investment in pharma hubs and supportive policies encourage manufacturing expansion, and rising demand for generic drugs and new drug applications boost the need for third-party production.
Asia Pacific and Latin America pharmaceutical contract manufacturing comprises outsourcing drug development and production to specialized third-party organizations. Asia Pacific offers significant cost-effective API production and high-volume manufacturing, with China and India acting as dominant hubs benefiting from skilled labor and massive FDA-approved facilities. Latin America serves as a crucial nearshoring production hub, offering expedited regulatory approvals and growing capabilities in biologics/biosimilar manufacturing. The market growth is driven by the increasing complexity of drugs requiring specialized expertise and strategic investments in advanced manufacturing technology, such as continuous manufacturing.
The shift from small molecules to high-value, complex large molecules, including monoclonal antibodies, vaccines, and cell and gene therapies, requires CDMOs to invest in specialized aseptic processing. South Korea, Singapore, and China are increasing hubs for biopharmaceutical production to meet the rising demand for biosimilars.
Pharmaceutical companies are increasingly seeking partners that offer integrated services, from early-stage formulation development to commercial-scale manufacturing and packaging, rather than relying on multiple providers. This one-stop-shop approach helps reduce drug development times and optimize logistical costs.
APAC-based CDMOs are aggressively integrating Industry 4.0 paradigms, leveraging artificial intelligence to drive automated quality assurance and predictive maintenance protocols. The strategic deployment of single-use bioreactor technologies is enhancing manufacturing agility, allowing for rapid transition between complex product lines. These high-tech advancements are mission-critical for ensuring operational alignment with the stringent regulatory benchmarks established by the U.S. FDA and the European Medicines Agency.
AI and ML are profoundly transforming the pharmaceutical contract manufacturing industry in the Asia-Pacific and Latin America region by enabling advanced process optimization, predictive maintenance, and smarter quality control. In APAC, leading hubs, such as China, India, and South Korea, are integrating AI-driven systems into manufacturing lines to minimize human error and drastically reduce operational costs. AI adoption is enhancing API production and accelerating clinical batch manufacturing, allowing manufacturers to meet growing demand and strengthen regulatory compliance. Key applications include AI-powered machine vision for instant defect detection, and predictive models that improve production yields in sterile injectables and biologics manufacturing.
| Report Coverage | Details |
| Market Size in 2026 | USD 23.69 Billion |
| Market Size by 2035 | USD 48.99 Billion |
| Growth Rate From 2026 to 2035 | CAGR of 8.41% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Type, Molecule Type, Services, Therapeutic Area, Region |
| Market Analysis (Terms Used) | Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope | North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled | WuXi STA; Piramal Pharma Solutions; Syngene International; Samsung Biologics; Jubilant Pharmova; Celltrion; Ajinomoto Bio-Pharma Services; Almac Group; Rubicon Research Pvt. Ltd.; Aurigene Pharmaceutical Services; Eurofarma Laboratórios S.A; Unither Pharmaceuticals; Fujifilm Diosynth Biotechnologies; Patheon (Thermo Fisher Scientific); Pfizer |
Driver
Rising Generic and Biosimilar Demand
The surge in patient expiries for blockbuster biologic drug has created a patent cliff opportunity, leading to a high demand for cost-effective manufacturing solutions that can produce biosimilars at a fraction of the cost of originators. The government policies encouraging affordable healthcare and local production in both regions are pushing for local CMOs to adopt specialized manufacturing for biologics and sterile injectables, speeding up market entry.
Infrastructure and Quality Constraints
The market growth is hindered by the high capital investments needed for advanced infrastructure, coupled with stringent, varying regulatory standards, which restrict expansion for smaller players, while chronic shortages of specialized skilled labor impact quality control. Latin American manufacturers face technological gaps, with a significant reliance on imported active pharmaceutical ingredients (APIs) and limited specialized facilities for complex biologics.
Expansion of Biologics/CDMO Services
The shifting focus from traditional small molecules to complex biopharmaceuticals, such as monoclonal antibodies and vaccines. The rising investment in biologics infrastructure by CDMOs in China, India, and South Korea, supported by government initiatives, is creating a robust ecosystem for end-to-end outsourcing. The need for cost-effective manufacturing and increased local production of biosimilars is propelling rapid market expansion.
Why Did the Pre-Clinical Segment Hold a 63% Share for the Asia Pacific and Latin America Pharmaceutical Contract Manufacturing Market?
The pre-clinical segment held a dominant position in the market with a share of 63% in 2025, owing to the pharmaceutical firms' utilization of CROs to manage high research and development costs and enhance operational efficiency. The increasing number of small-molecule and early-stage drug candidates requires robust in vitro and in vivo studies, which are often outsourced to local contract service providers. This stage is critical for safety and efficacy testing, and cost-efficient outsourcing.
The clinical segment held a 27% share of the market in 2025. The increasing number of drug development programs and specialized clinical trials, particularly for oncology and chronic diseases, drives demand for clinical-phase manufacturing. It reflects the ongoing importance of pre-clinical stages, a large patient population, and efficient recruitment.
The drug discovery segment holds a 10% share of the market in 2025, with the increasing investment in advanced cell and gene therapy manufacturing, attracting research and development outsourcing. The government supports local pharmaceutical manufacturing and improvements in healthcare. The growing focus on later-stage development and high-volume geriatric demand.
Why Did the Large Molecules Segment Lead the Asia Pacific And Latin America Pharmaceutical Contract Manufacturing Market?
The large molecules segment registered its dominance over the market with a share of 53% in 2025, owing to the increasing reliance on biologics for treating complex diseases. The significant investment in biomanufacturing technology and favorable government policies for pharmaceutical outsourcing have accelerated the development of these systems.
The small molecules segment held the 47% share of the market in 2025, due to a surge in diseases requiring small molecule treatments, which drives the need for local production, low-cost manufacturing, substantial GMP-compliant infrastructure, and a skilled workforce, fueling market growth. The gain in traction through increased clinical research and government initiatives fostering domestic pharmaceutical manufacturing.
Why Did the Project Management/Clinical Supply Management Segment Lead the Asia Pacific And Latin America Pharmaceutical Contract Manufacturing Market?
The project management/clinical supply management segment registered its dominance over the market with a share of 34% in 2025, owing to the importance of efficient drug development, the increasing number of clinical trials, and the growing need for expert clinical supply management. The complexity of managing, shipping, and distributing trial materials in a diverse geographic area has driven the demand for specialized project management services.
The investigator payments segment held the 23% share of the market in 2025, with the surge in regional clinical trial activities requiring extensive management of trial management, sites, and investigators, elevating the need for organized payment systems. Growing the need for robust financial management in clinical trials.
The technology segment held 10% share of the market in 2025, due to the shift toward biologics, high-potency APIs, and personalized medicine necessitates advanced manufacturing that CMOs in these regions are investing. Adoption of advanced technology reduces production costs and enhances capacity, attracting multinational firms.
The patient and site recruitment segment held a 4% share of the market in 2025. With the rising complexity in trials and the need to manage study sites and patient retention in decentralized, multi-center trials increases reliance on specialized recruitment services. It is improving regulatory harmonization and increasing retention in decentralized, multi-center trials, and increasing reliance on specialized recruitment services.
The laboratory segment held 6% share of the market in 2025, because it plays a major role in development, expansion of academic and preclinical research, particularly in antibody engineering, immunology, and therapeutic discovery, which increased reliance on external laboratories.
The bio-statistics segment held 4% share of the market in 2025, with the shift from traditional small-molecule drugs to complex biologics and biosimilars necessitating advanced analytical and statistical capabilities. It offers cost-effective, high-quality analytical and manufacturing services.
The quality management/assurance segment held 6% share of the market in 2025, due to the advancement in manufacturing requiring sophisticated, integrated quality assurance, boosting value, production’s shift toward more complex, sensitive biologics, and stringent quality control.
The clinical monitoring segment held 5% share of the market in 2025, because the rising complex trials for cancer and rare diseases necessitate specialized monitoring. A surge in clinical trials and government efforts to strengthen local production has increased demand for local monitoring and trial management.
The medical writing segment held 1% share of the market in 2025, because of the rising pharmaceutical research and developments, clinical trials, and drug approvals in the region, high quality documentation for submission to various regulatory bodies. Increasing investment in complex products and advanced theories, and a rising need for cost-effective solutions.
The regulatory/medical affairs segment held 3% share of the market in 2025, because the growth of complex, biologics, biosimilars, and generics requires rigorous documentation, clinical trial approvals, and post-market surveillance. The shift toward outsourcing to specialized regulatory affairs providers to minimize in-house costs associated with maintaining regulatory teams.
The data management segment held 1% share of the market in 2025, because the increasing adoption of AI, machine learning, and data analytics tools helps optimize production and manage in, out flow of data, improving quality control. Rising complexity of biologics, expansion of end-to-end service, and rapid expansion in API and drug manufacturing.
The other segment held 2% share of the market in 2025, due to the broad nature of other services. Increased focus on high-potency drugs, advanced drug delivery systems, and sterile manufacturing services, and expansion of specialized services.
Why Did the Cardiovascular Disease Segment Lead the Asia Pacific And Latin America Pharmaceutical Contract Manufacturing Market?
The cardiovascular disease segment registered its dominance over the market with a share of 24% in 2025, owing to the strong focus on cardiovascular health as a major global health issue. The growing middle class and improved healthcare access, high-volume output, align with manufacturing services within the pharmaceutical contract market. Increasing sedentary lifestyles and urbanized populations contribute to a greater prevalence.
The respiratory diseases segment held the 24% share of the market in 2025, with the rising respiratory conditions globally, especially post-pandemic. Significant investment in healthcare infrastructure and increased access to treatment have elevated the demand for locally produced, affordable generics and respiratory medicines.
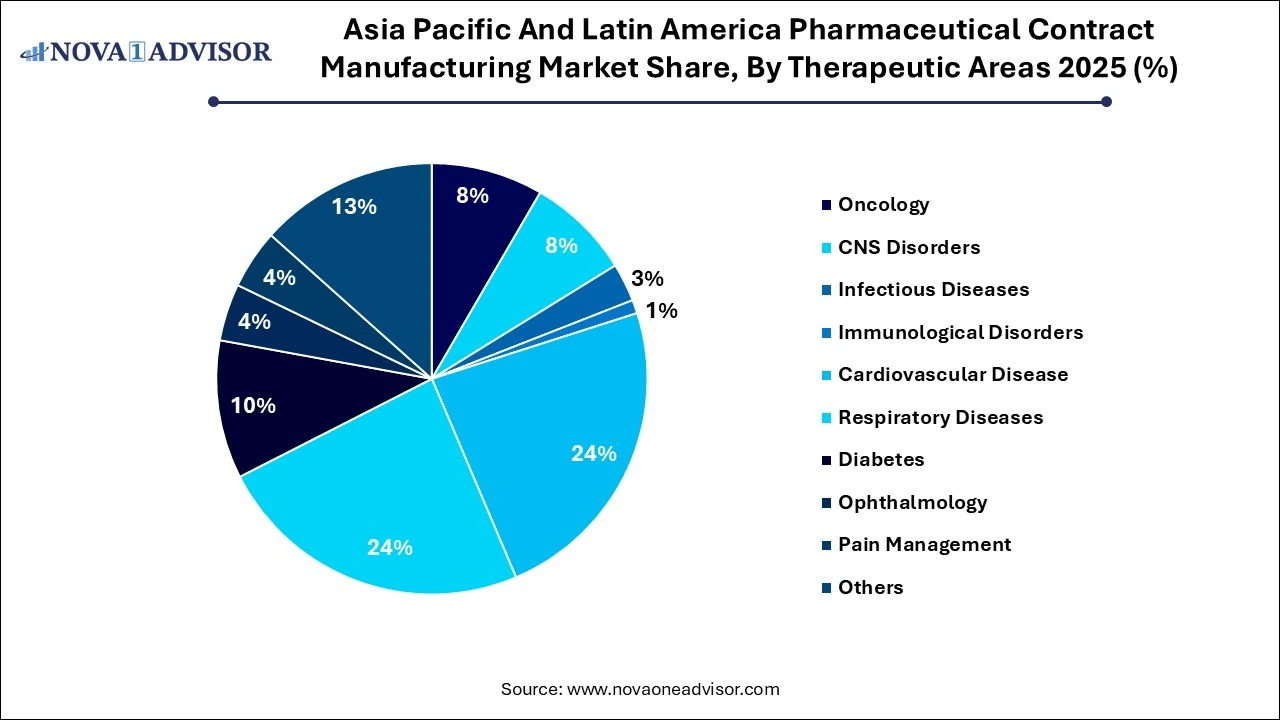
The oncology segment held 8% share of the market in 2025, due to the rising prevalence of various cancers, increased production of chemotherapy, and targeted therapies. Growing research and development spending on biologics, monoclonal antibodies, and antibody-drug conjugations contribute to the strong demand.
The CNS disorders segment held a 8% share of the market in 2025. The increase in neurological disorders and higher production volumes for treatments, specifically in aging populations. It involves complex molecules, leading firms to rely on specialized contract development and manufacturing organizations for manufacturing and clinical trials.
The infectious diseases segment held a 3% share of the market in 2025. Due to the growing shift towards chronic diseases and personalized medicine. The rising prevalence of infectious diseases and increasing focus on novel antimicrobials and advanced therapeutics necessitate outsourcing to specialized CMOs in the region.
The immunological disorders segment held a 1% share of the market in 2025. The expansion of patents for biologics used in immunology in biosimilar production requires robust contract manufacturing. The shift toward targeted therapies and advanced bioprocessing techniques contributes to the reliance on CDMOs for specialized immunological drug production.
The diabetes segment held a 10% share of the market in 2025. The convergence of escalating chronic disease prevalence and the strategic pivot toward regionalized CDMO partnerships is fundamentally reshaping the global pharmaceutical landscape. By integrating advanced manufacturing infrastructure with cost-optimized production models, Asia-Pacific and Latin American hubs are evolving into indispensable components of the global drug supply chain.
The ophthalmology segment held a 4% share of the market in 2025, with the increasing cases of macular degeneration, diabetic retinopathy, and dry eye syndrome, particularly with the aging population. Increasing outsourcing to specialized CDMOs and cost-effective manufacturing. The industry's shift toward preventative-free, unit-dose packaging and specialized, high-margin.
The pain management segment held a 4% share of the market in 2025 because of the increasing focus on developing innovative drug delivery systems, which drives contract manufacturing growth for specialized topical and sustained-release pain treatments. A high prevalence of chronic disease burden and cost-efficient outsourcing.
The others segment held a 13% share of the market in 2025, with the increasing diversification in drug development. The expansion of regional packaging hubs and the booming generic drug necessitates comprehensive contract manufacturing, spanning from API to the final packaging, and diversified service providers.
Why Did the Latin America Region Lead the Asia Pacific and North American Laboratory Animal Medicine Market?
Latin America registered its dominance over the market with a share of 80% in 2025, owing to the strong pharmaceutical manufacturing base and strategic partnerships within the region. The combination of favorable demographic tailwinds and a strengthening regulatory landscape is attracting significant foreign direct investment from global life sciences firms.
The Asia Pacific region held the 20% share of the market in 2025, with a rising incidence of diseases originating from animals has increased the demand for surveillance and preventative treatments, boosting market growth. Rapid urbanization and increased disposable incomes, advanced veterinary care, sophisticated diagnostics, and medications.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the Asia Pacific and Latin America pharmaceutical contract manufacturing market.
By Type
By Molecule Type
By Services
By Therapeutic Areas
By Regional