Autoimmune Disease Diagnosis Market Size, Growth and Trends 2026 to 2035
The global autoimmune disease diagnosis market size was reached at USD 6.30 billion in 2025 and is expected to be worth around USD 13.74 billion by 2035 with a CAGR of 8.11% from 2026 to 2035.
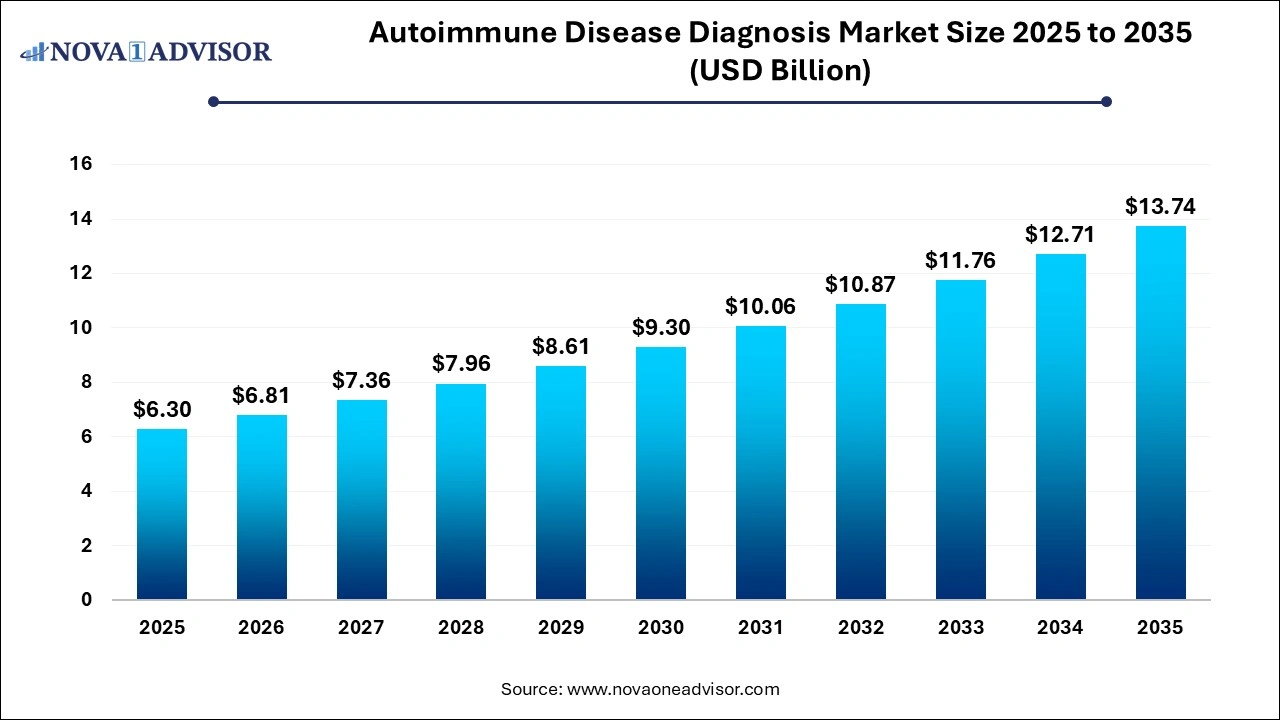
Autoimmune Disease Diagnosis Market Outlook
- Market Growth Overview: The autoimmune disease diagnosis market is expected to grow significantly between 2026 and 2035, driven by the rising prevalence of autoimmune diseases, adoption of technological advancements in diagnostics, and growing public awareness and screening.
- Sustainability Trends: Sustainability trends involve automation and efficiency, technological integration, and telemedicine and remote care.
- Major Investors: Major investors in the market include Thermo Fisher Scientific, Abbott Laboratories, Siemens Healthineers, F. Hoffmann-La Roche, and Bio-Rad Laboratories.
Report Scope of Autoimmune Disease Diagnosis Market
| Report Coverage |
Details |
| Market Size in 2026 |
USD 6.81 Billion |
| Market Size by 2035 |
USD 13.74 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 8.11% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product, By Test Type, By Disease, By End User |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Covered |
North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled |
Thermo Fisher Scientific, Siemens Healthineers, Abbott, Bio-Rad Laboratories |
Autoimmune Disease Diagnosis Market Segment Insights
By Product Insights
How did the Consumables and Assay Kits Segment Dominate the Autoimmune Disease Diagnosis Market?
The consumables & assay kits segment is driven by the rising prevalence of complex conditions, such as lupus and rheumatoid artirities rises, and the necessity for specialized, tailored assays ensures a consistent and expanding revenue system that outpaces one-time instrument sales. The integration of multiplex technology into rapid diagnostic kits has transformed these consumables from simple reagents into high-precision tools for immediate clinical decision-making.
By Test Type Insights
How did the Autoantibody and Immunologic Tests Segment Account for the Largest Share in the Autoimmune Disease Diagnosis Market?
The autoantibody and immunologic tests segment is driven by the widespread utilization of antinuclear antibody (ANA) tests as a primary screening tool, combined with the integration of automated ELISA platforms, which has significantly improved diagnostic efficiency and standardization. As the rising global disease prevalence rises, these tests remain indispensable for identifying specific immune markers, often before physical symptoms fully manifest.
How did the RoYutine Laboratory Tests Segment Expect to Hold the Fastest-Growing Autoimmune Disease Diagnosis Market in the Coming years?
The routine laboratory tests segment is driven by the wide clinical utility of ANA and CRP testing, which provides a reliable, cost-effective foundation for early detection and long-term disease monitoring. The integration of modernized, automated lab techniques is significantly increasing the throughput and accuracy of these fundamental diagnostic tools. The synergy between rising disease prevalence and enhanced accessibility ensures that routine testing remains the highest-volume segment within the autoimmune diagnostic landscape.
By Disease Insights
How did the Systemic Autoimmune Diseases Segment Account for the Largest Share in the Autoimmune Disease Diagnosis Market?
The systemic autoimmune diseases segment is driven by its inherent complexity and the high-volume, multi-organ testing they require for accurate management. The clinical reliance on ANA testing as a primary diagnostic tool, combined with the integration of multiplex assays, allows for the comprehensive monitoring of chronic conditions like Lupus and Crohn's disease. As global awareness shifts toward early detection to prevent irreversible organ damage, the demand for these sophisticated antibody panels continues to escalate.
How did the Rheumatoid Arthritis Segment Expect to Hold the Fastest-Growing Autoimmune Disease Diagnosis Market in the Coming Years?
The rheumatoid arthritis segment is driven by the development of highly specific biomarkers, such as ACPA, combined with the transition to automated immunoassays, which has drastically improved detection accuracy and patient outcomes. The strategic market moves like LabCorp’s acquisition of the Vectra RA test underscore the massive commercial push to dominate this high-volume landscape.
By End User Insights
How did the Hospitals Segment Account for the Largest Share in the Autoimmune Disease Diagnosis Market?
The hospitals segment is driven by serving as the primary hubs for high-volume, complex diagnostic workups required for severe autoimmune conditions. The integration of advanced infrastructure, such as high-throughput autoantibody panels and flow cytometry, allows for rapid, in-house verification of life-altering diagnoses. The presence of multidisciplinary specialist teams ensures that diagnostic insights are immediately converted into integrated treatment plans.
How did the Clinical Laboratories Segment Expect to Hold the Fastest-Growing Autoimmune Disease Diagnosis Market in the Coming Years?
The clinical laboratories segment is driven by leveraging a specialized focus on high-throughput automation and complex molecular diagnostics. The integration of next-generation instruments, such as Revvity's EUROIMMUN UNIQO 160, allows these labs to deliver superior precision and speed that traditional hospital settings often struggle to match. The growing trend of hospital outsourcing for specialized multiplex immunoassays has created a massive, recurring revenue stream for independent diagnostic providers.
Key Players in the Autoimmune Disease Diagnosis Market
- Thermo Fisher Scientific: Thermo Fisher Scientific provides a broad portfolio of autoimmune diagnostic tests through its EliA autoimmunity solutions, which offer high specificity and sensitivity for detecting autoantibodies.
- Siemens Healthineers: Siemens Healthineers offers a comprehensive range of immunoassay systems (such as ADVIA Centaur and IMMULITE) to detect and monitor autoimmune diseases, including thyroid disorders, rheumatoid arthritis, and SLE.
- Abbott: Abbott Laboratories contributes to the market with a wide range of diagnostic instruments and consumables designed for high-throughput testing in laboratories.
- Bio-Rad Laboratories: Bio-Rad Laboratories provides a diverse portfolio of autoimmune diagnostic tools, including BioPlex multiplex immunoassays that allow for the simultaneous detection of multiple autoantibodies.
Autoimmune Disease Diagnosis Market Segments Covered
By Product
- Consumables & Assay Kits
- Instruments
By Test Type
- Routine Laboratory Tests
- Inflammatory Markers
- Autoantibodies & Immunologic Test
- Other Tests
By Disease
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Sjögren’s Syndrome
- Thyroiditis
- Scleroderma
- Other Diseases
By End User
- Clinical Laboratories
- Hospitals
- Other End Users
By Region
- North America
- Europe
- Germany
- France
- UK
- Italy
- Spain
- RoE
- Asia Pacific
- China
- Japan
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Middle East & Africa

