Cardiac Biomarkers Market Size and Growth 2026 to 2035
The cardiac biomarkers market size was estimated at USD 22.09 billion in 2022 and is expected to surpass around USD 87.82 billion by 2035 and poised to grow at a compound annual growth rate (CAGR) of 14.8% during the forecast period 2026 to 2035.
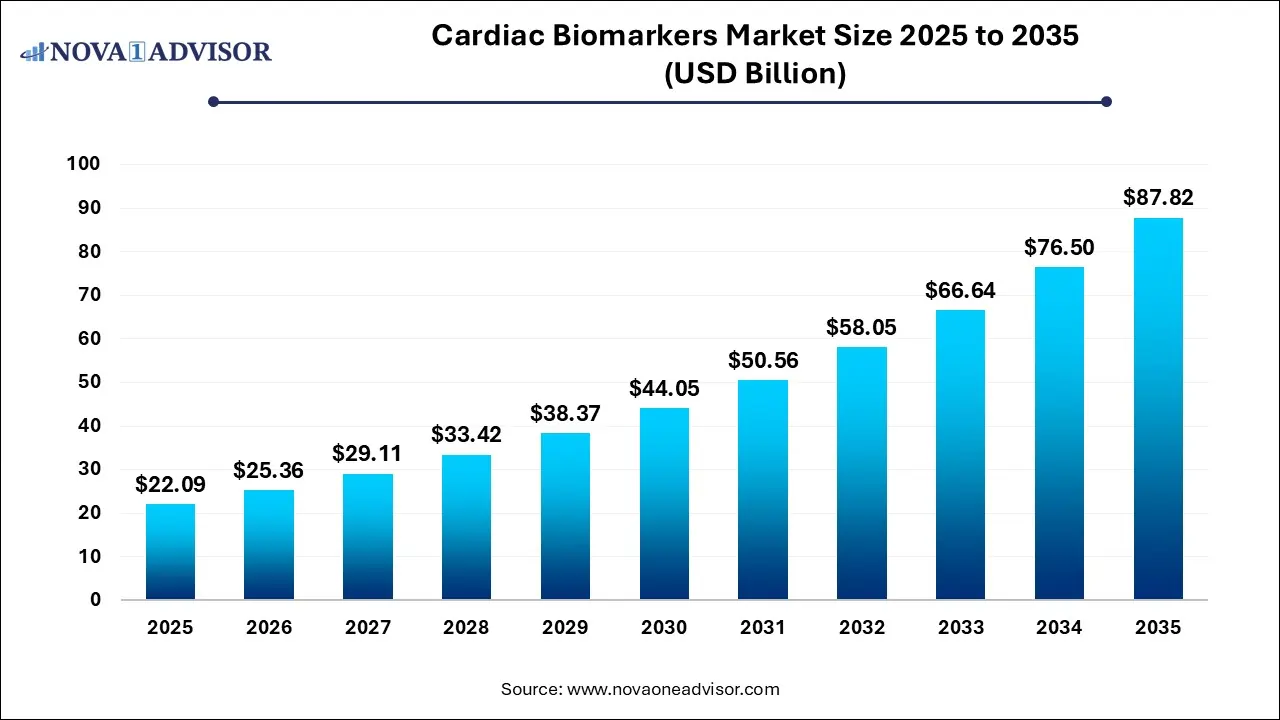
Key Takeaways:
- North America dominated the global market with a share of over 42.0% in 2025.
- Asia Pacific is expected to expand at a significant rate during the forecast period
- The troponin segment held the largest revenue share of over 33.0% in 2025 and is expected to witness the fastest growth during the forecast period.
- Acute coronary syndrome held the largest revenue share of over 53.0% in 2025.
- The laboratory testing segment held the largest revenue share of over 67.0% in 2025.
How AI is Impacting the Cardiac Biomarkers Market
AI and ML are transforming the cardiac biomarkers industry by shifting diagnostics from reactive testing to proactive, predictive risk stratification. Algorithms are being integrated into point-of-care (POC) devices, analyzing high-sensitivity troponin and novel biomarkers to enable rapid, precise diagnosis within 10-20 minutes. AI-enabled predictive analytics enhance clinical decision-making by integrating biomarker data with electronic health records (EHR) and imaging, improving the ability to predict major adverse cardiac events up to a year in advance.
Market Outlook
- Market Growth Overview: The cardiac biomarkers market is expected to grow significantly between 2025 and 2034, driven by the rising burden of cardiovascular diseases, the growing geriatric population, and increasing adoption of point-of-care testing.
- Sustainability Trends: Sustainability trends involve the development of eco-friendly products, energy efficient compacts equipment, and optimized supply chains and green logistics.
- Major Investors: Major investors in the market include Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific, and Danaher Corporation.
Cardiac Biomarkers Market Report Scope
| Report Attribute |
Details |
| Market Size in 2026 |
USD 25.36 Billion |
| Market Size by 2035 |
USD 87.82 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 14.8% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
Type, Application, End-use, Region |
| Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
| Report Coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Key Companies Profiled |
Abbott; Quidel Corporation; Siemens Healthineers; F. Hoffmann-La Roche Ltd.; Danaher Corporation; Biomérieux SA; Bio-Rad Laboratories, Inc.; Randox Laboratories Ltd.; Creative Diagnostics; Life Diagnostics |
By Type Insights
The troponin segment held the largest revenue share of over 33.0% in 2025 and is expected to witness the fastest growth during the forecast period. The exponential growth can be attributed to factors such as diagnostic efficiency, specificity, and accurate predictive detection of cardiac events when compared to other tests. The growing prevalence of myocardial infarction and stroke across the globe further boosts the market growth.
The BNP and NT-proBNP segment is expected to witness significant growth owing to its usage in the detection of heart damage and stress. New players are entering the market with the test in order to generate lucrative revenues. For instance, in June 2022, LumiraDx Limited announced the diversification of its portfolio with CE Mark for its D-Dimer test and NT-proBNP test. The commercialization of the latter is projected by the end of 2025, which is expected to facilitate clinical decision-making.
By Application Insights
Acute coronary syndrome held the largest revenue share of over 53.0% in 2025 as the disease burden is rapidly rising in low- and middle-income countries when compared to high-income countries. Factors such as rapid urbanization and an increase in jobs that are sedentary are driving the disease incidence. Cardiac troponin T and I biomarkers tests have emerged as the prime diagnostic methods employed for the diagnosis of the disease.
Myocardial infarction is anticipated to witness lucrative growth owing to its rising incidence globally. The enhanced focus of researchers on myocardial infarction is expected to create lucrative opportunities in the segment. For instance, as per the Science Journal, in 2025, researchers identified 27 novel proteins for personalized prediction of myocardial infarction risk.
By End-use Insights
The laboratory testing segment held the largest revenue share of over 67.0% in 2022. This can be attributed to the advantages offered by laboratory testing such as high sensitivity, specificity, scalability, and cost-efficiency. Furthermore, these laboratories facilitate the testing of all types of samples. Diagnostic laboratory services are crucial in clinical decision-making.
The point of care (POC) testing segment is demonstrating exponential growth due to the increasing use and adoption of highly sensitive and easy-to-use POC Troponin tests. The rising emphasis on providing effective, value-based healthcare services in the face of tight budgets is enhancing the importance of POC tests. In addition, these tests are the primary choice of medical professionals during emergencies for faster diagnosis of the patients. The demand for POC test kits for use in nursing homes and home healthcare is driven by the global phenomenon of the aging population, which is on the rise.
By Regional Insights
North America dominated the global market with a share of over 42.0% in 2025. This can be attributed to the developed healthcare system and high adoption of cardiac biomarkers as a tool for diagnosis and predictive diagnosis of conditions. The rising geriatric population requires the use of biomarker testing for the identification of conditions such as acute myocardial infarction, which is driving the demand for testing products. The presence of key players such as Quidel Corporation and Danaher Corporation in the region is another factor adding to the growth of this market.
Asia Pacific is expected to expand at a significant rate during the forecast period owing to rising affordability for advanced cardiac tests, modernization in the countries, and increasing disposable income. The growing incidence of cardiovascular diseases (CVDs) in this region has resulted in a rise in the demand for improved diagnostics, government funding for target disease and other research, and healthcare expenditure. India has a higher prevalence of CVDs when compared to other developing countries. This high disease burden in the region is expected to drive the demand for cardiac biomarker diagnostics for facilitating the diagnosis, evaluation, and management of CVDs.
U.S. Cardiac Biomarkers Market Trends
The U.S.’s strategic shift toward point-of-care (POC) testing, which allows for decentralized, rapid diagnostics in emergency rooms and ambulances, aims to improve patient outcomes. The integration of AI-driven predictive analytics is transforming these biomarkers from simple diagnostic tools into sophisticated risk-assessment systems for remote and outpatient monitoring. The expansion of biomarker applications into sports medicine and corporate wellness is diversifying the market footprint beyond traditional hospital settings.
China Cardiac Biomarkers Market Trends
China’s government’s volume-based procurement (VBP) policy is significantly increasing testing volumes by lowering costs and incentivizing the adoption of high-quality domestic medical devices. The synergy of aging demographics and aggressive healthcare infrastructure reform is cementing China as a global leader in cardiac biomarker consumption.
Supply Chain Analysis of the Cardiac Biomarkers Market
- Research & Development (R&D) and Novel Marker Identification
This initial stage involves the discovery of novel biomarkers and the enhancement of existing high-sensitivity troponin assays to improve diagnostic accuracy and early rule-out capabilities. R&D focuses on improving sensitivity for detecting minor cardiac injuries and widening the pipeline for personalized cardiac therapeutics.
Key Players: F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific, Inc., bioMérieux SA, and Academic research institutes.
- Manufacturing and Production
This stage includes the high-volume manufacturing of diagnostic kits, reagents, and analytical instruments such as immuno-fluorescence analyzers (IFA) and point-of-care (POC) devices.
Key Players: Abbott Laboratories, Siemens Healthineers AG, Danaher Corporation, Bio-Rad Laboratories, Inc., and Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- Regulatory Approval and Clinical Validation
Before entering the market, cardiac biomarker products must undergo rigorous testing to obtain regulatory clearances (e.g., FDA, CE-mark), ensuring safety and efficacy. This phase is crucial for establishing the clinical utility of new assays, particularly in high-sensitivity troponin testing and quick-turnaround POC systems.
Key Players: Regulatory bodies (FDA, NMPA, PMDA), major manufacturers like Roche and Abbott.
Key Players in the Cardiac Biomarkers Market
Abbott contributes to the cardiac biomarkers market by providing high-sensitivity troponin-I tests, such as the ARCHITECT and Alinity i platforms, which enable rapid and precise diagnosis of heart attacks.
QuidelOrtho is a major player in the cardiac POC market, known for developing the Triage® System, which is recognized as the world's first point-of-care cardiac biomarker testing system.
Siemens Healthineers contributes with their Atellica Laboratory Diagnostics and POC systems, which offer fast, 10-minute high-sensitivity troponin I results to accelerate the management of cardiovascular diseases.
- F. Hoffmann-La Roche Ltd.
Roche is a market leader providing the Elecsys platform, which uses highly sensitive troponin T (cTnT-hs) and NT-proBNP assays for early detection and risk stratification of heart failure and AMI. Their contributions focus on reducing the time to diagnosis with rapid, automated tests that aid clinicians in cardiac risk assessment.
Danaher contributes through its diagnostics subsidiary, Beckman Coulter, which specializes in automated immunoassay systems for cardiac markers like BNP and troponin, with a strong focus on rapid point-of-care testing.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the Cardiac Biomarkers market.
By Type
- Troponin
- CK-MB
- Myoglobin
- BNP and NT-proBNP
- Others
By Application
- Acute Coronary Syndrome
- Myocardial Infarction
- Congestive Heart Failure
- Others
By End-use
- Laboratory Testing
- Point of Care Testing
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa (MEA)


