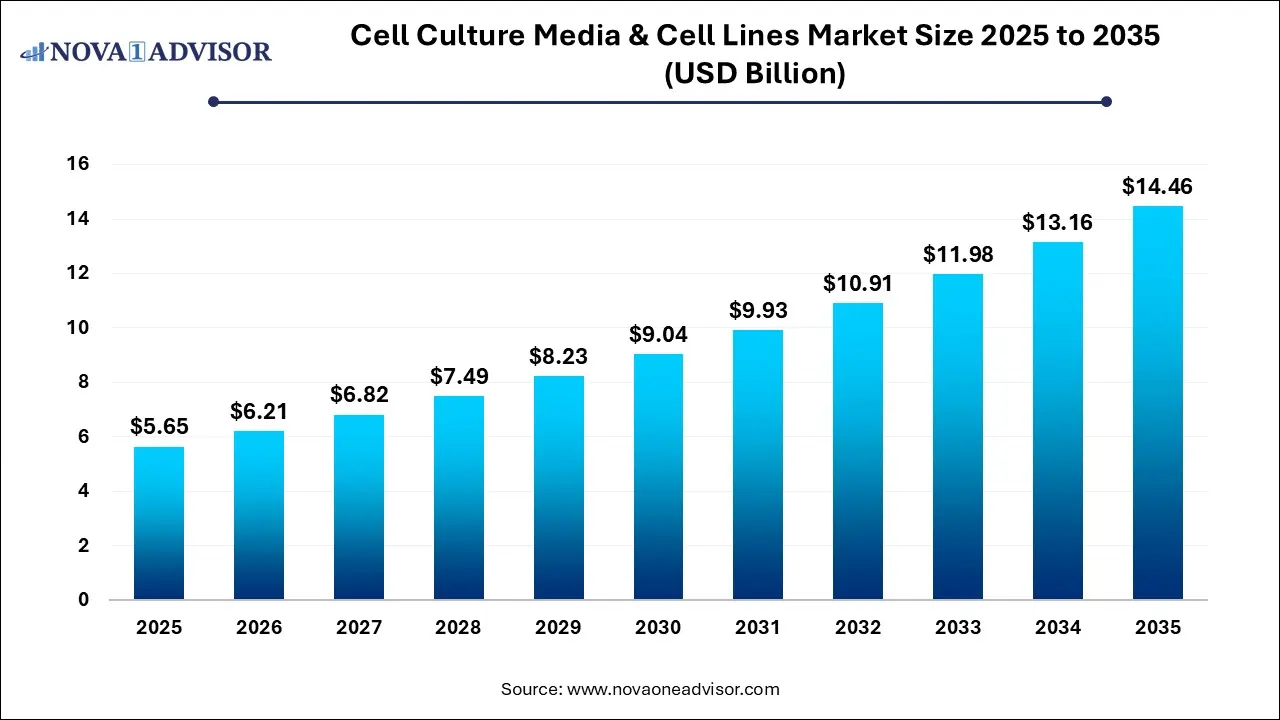
The global cell culture media & cell lines market was estimated at USD 5.65 billion in 2025 and is projected to reach USD 14.46 billion by 2035, growing at a CAGR of 9.65% over the forecast period 2026-2035. Market growth is driven by the rising production of biologics and biosimilars, which requires extensive use of specialized media to grow cells at scale, development of targeted therapies and antibodies, and the industry's shift toward serum-free media, automated cell culture systems, and single-use technologies, enhancing efficiency and decreasing contamination.
Key Takeaways
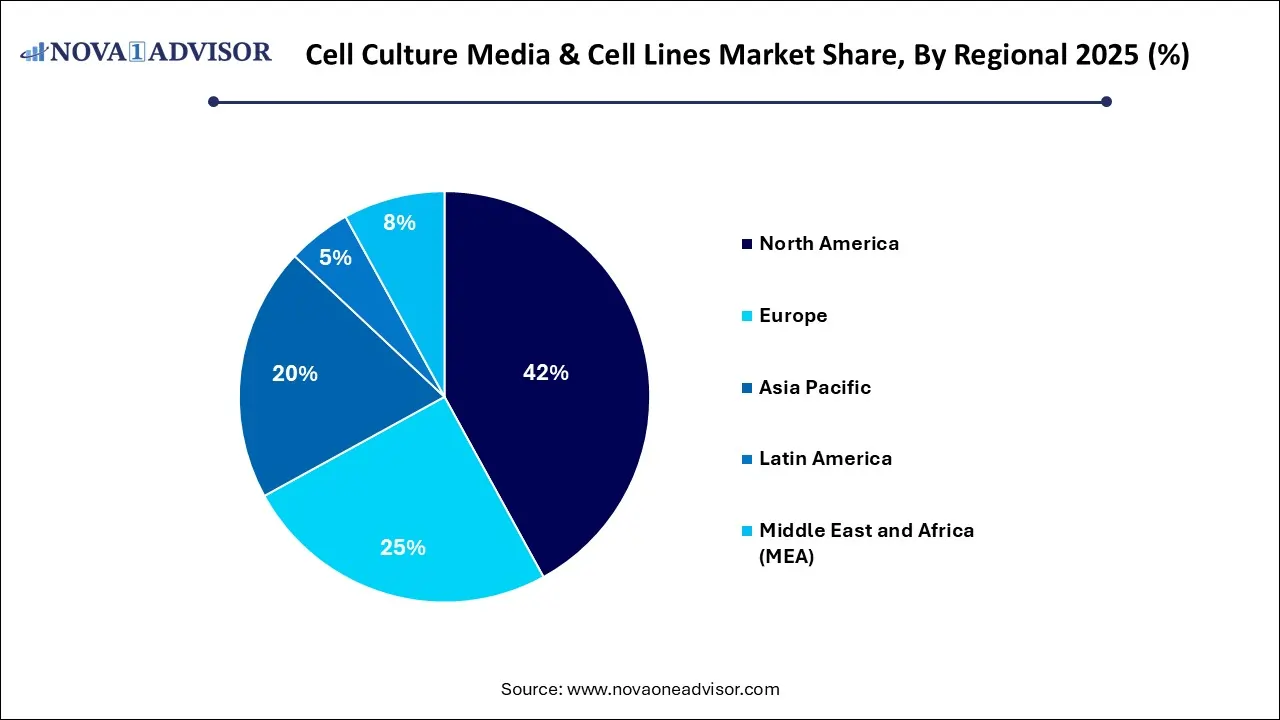
- By region, North America held the largest share of the cell culture media & cell lines market in 2025.
- By region, Asia Pacific is expected to experience the fastest growth between 2025 and 2034.
- By product, the specialty media products segment led the market in 2025.
- By product, the stem cells & blood-derived cells segment is expected to expand at the highest CAGR over the projected timeframe.
- By application, the biopharmaceutical production segment led the market in 2025.
- By application, the tissue engineering and regenerative segment is the fastest growing in the cell culture media & cell lines market.
- By end-use, the large biopharmaceutical companies segment dominated the cell culture media & cell lines market in 2025.
According to the National Institute of Health, AI and ML are transforming the cell culture media and cell lines industry by accelerating formulation development and improving process efficiency, thereby shifting workflows from trial-and-error to data-driven decision-making. ML models analyze complex, multidimensional data to predict optimal nutrient compositions, reducing media development time and costs. In cell line development, AI automates imaging and analysis to rapidly identify and select high-producing clones, which can increase titers compared with traditional methods. ML enables real-time monitoring and automation of bioreactors, ensuring consistent quality, improved scalability, and reduced batch-to-batch variability, particularly for serum-free and chemically defined media.
The cell culture media & cell lines market comprises a nutrient-rich solution, commonly serum free or chemically defined, that provide essential amino acids, vitamins, and salts to support in vitro cell growth, while cell line are immortalized cells used for consistent, to support in vitro cell growth, while cell lines are immortalized cells used for consistent, reproducible experiments. These technologies offer major benefits in biopharmaceutical production by ensuring high-yield, reproducible, and safe manufacturing of therapeutic proteins and vaccines. In drug discovery, cell lines act as essential models for toxicity testing and screening, reducing the reliance on animal testing.
Market Outlook
- Market Growth Overview: The cell culture media & cell lines market is expected to grow significantly between 2025 and 2034, driven by the increasing prevalence of chronic diseases and infections has surged the demand for monoclonal antibodies, recombinant proteins, and vaccines necessitating high-ield cell culture systems, expansion of cell and gene therapy pipelines, and growing adoption of 3D cell clture along with automated, high-throughput systems, has improved production efficiency and precision.
- Sustainability Trends: Sustainability trends are industry is moving away from fetal bovine serum to chemically defined, plant-based, and synthetic alternatives, reducing ethical concerns, batch-to-batch variation, and contamination risk. Adoption of single-use technology, implementing green manufacturing processes, and efforts to reduce raw materials.
- Major Investors: Major investors in the market include Thermo Fisher Scientific, Merck KGaA, Danaher Corporation, Sartorius AG, Lonza Group, and Fujifilm Holdings. They are accelerated by extensive product portfolios and heavy investment in manufacturing capacity for bioprocessing, investing in partnerships to optimize synthetic biology and cell growth efficiency, and growing investments in specialized cell lines and media designed for stem cell and bioprocessing applications.
- Startup Economy: The startup economy in the market consists of advanced therapies, such as cell therapies, stem cells, and gene therapies, which require customized media formulations that standard providers do not offer. Advancing serum-free technologies, growing digital tools to speed up the discovery of new media formulations and automate the production process, enhancing efficiency and lowering costs for therapeutic manufacturing.
| Report Coverage |
Details |
| Market Size in 2026 |
USD 6.21 Billion |
| Market Size by 2035 |
USD 14.46 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 9.65% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product, By Application, By End-use, By Region |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
Sartorius AG; Danaher; Merck KGaA; Thermo Fisher Scientific, Inc.; FUJIFILM Corporation; Lonza; STEMCELL Technologies; PromoCell GmbH; ATCC; AllCells |
Market Dynamics
Drivers
Surging Demand for Biopharmaceutical
With the rapid expansion of the cell culture media and cell lines industry, as biotechnology and pharmaceutical companies scale up production to treat chronic and infectious diseases, there is a heightened need for high-quality, consistent, and customized cell culture products to ensure efficient, high-yield manufacturing. This surge drives the widespread adoption of specialized, serum-free, and chemically defined media, which offer better reproducibility and lower contamination risks compared to traditional methods. The growing focus on personalized medicine and regenerative medicine necessitates advanced cell lines and nutrient-rich media to maintain cell viability, directly accelerating market growth.
- In January 2024, AbbVie announced a $223 million investment to expand its biologics manufacturing capacity in Singapore by 24,000 liters to support its immunology and oncology pipeline.
Rising Growth in Cell and Gene Therapies
The rapid expansion of the cell and gene therapy sector, driven by increasing clinical trials and regulatory approvals, is significantly boosting the demand for high-quality cell culture media and specialized cell lines. CGT manufacturing involves complex processes, such as viral vector production and genetic modification of cells. There is a surge in demand for specialized, serum-free, and chemically defined media that ensure process consistency, high yield, and regulatory compliance. The need to produce customized, patient-specific cells for therapies, along with the scaling up of commercial production, requires a constant supply of validated cell lines and media.
Restraint
High Cost of Specialized Media
The market growth is hindered by the restricted adoption among smaller laboratories and academic research institutions with limited budgets. These advanced media types often account for a substantial portion of the total production cost, sometimes up to 55%–95%, making large-scale biomanufacturing financially challenging. The high expenditures on these products limit the ability of emerging biotech firms to invest in R&D, thus reducing the overall market volume. The need for specialized media to support sensitive cell lines like stem cells increases the cost of regenerative medicine research, hindering the expansion of these promising, high-growth sectors.
Opportunities
3D Cell Culture and Organoids
The expansion of cell culture media & cell lines creates a significant opportunity by providing more physiologically relevant models than traditional 2D systems, directly creating a high demand for specialized, complex cell culture media and supplements. 3D models require specialized, optimized media to sustain long-term growth, differentiation, and complex cell interactions. Companies are launching highly customized, serum-free, and chemically defined media formulations to replace less consistent options. The need for patient-derived organoids and stem cell research necessitates high-quality, sustainable cell lines to ensure reproducibility and therapeutic efficiency in drug development screening.
Custom Media Formulation Services
The market is creating immersive opportunities by providing tailored, optimized nutrient solutions that enhance cell growth, viability, and product yield for specialized, fastidious cell lines. These services enable biopharmaceutical companies to transition from generic, off-the-shelf media to chemically defined, animal-component-free formulations that ensure batch-to-batch consistency and comply with strict regulatory standards (GMP). By addressing specific metabolic needs, custom media accelerates the development timeline for novel therapeutics and vaccines, reducing the cost and time involved in process optimization and scaling up from the laboratory to commercial production.
Economic Growth and GDP
Economic growth and rising GDP generally lead to positive growth. By increasing investment in pharmaceutical R&D, biopharmaceutical production, and healthcare infrastructure. High GDP growth allows for greater funding in biotechnology startups and academic research, accelerating the demand for specialized, high-quality culture products used in drug development and regenerative medicine.
Inflation & Drug Pricing Pressures
It can negatively affect the growth of the cell culture media & cell lines market by increasing raw material costs, energy expenses, and reducing profit margins for biopharmaceutical manufacturers. These resources also drive a long-term market shift towards higher efficiency, as companies increase investments in advanced, high-productivity media and automated technologies to offset higher production costs.
Exchange Rates
Exchange rate fluctuations can negatively affect, creating volatility in procurement costs for raw materials, which complicates pricing and margins for international manufacturers. While depreciation of a local currency can make exports cheaper, sharp fluctuations or appreciation in the currency of importing countries often increases the cost of high-quality cell culture consumables, limiting adoption for smaller, price-sensitive organizations.
Segment Outlook
Product Insights
Why did the Specialty Media Products Segment Dominate the Cell Culture Media & Cell Lines Market in 2025?
The specialty media products segment dominated the market, accounting for the largest share in 2025. The transition toward high-yield, serum-free, and chemically defined media for manufacturing biologics like monoclonal antibodies and vaccines. Massive investment in cancer research and regenerative medicine is simultaneously fueling the need for specialized, nutrition-rich environments tailored for sensitive cell and gene therapies. Stringent regulatory requirements and the push for batch consistency have further accelerated the adoption of animal-component-free formulations to ensure patient safety.
The stem cells & blood-derived cells segment is expected to grow at the fastest CAGR during the projection period, driven by the increased government and private funding, accelerating the transition from lab-scale research to large-scale commercialization of stem cell-based treatments. The expansion of CROs and CMOs has created a standardized infrastructure for these complex therapies, driving a consistent, high-volume need for customized media formulations. The focus intensifies on chronic and neurodegenerative diseases, and the cell culture market is evolving into a high-value ecosystem centered on precision nutrients and advanced stem cell platforms.
Application Insights
How Does the Biopharmaceutical Production Segment Lead the Cell Culture Media & Cell Lines Market in 2025?
The biopharmaceutical production segment led the cell culture media & cell lines market in 2025 due to massive consumption of specialized cell lines and high-volume media to support large-scale therapeutic protein production. The biopharma facility expansion and the global rise of CDMOs, which provide the infrastructure needed for flexible, high-quality manufacturing. The transition to single-use systems and cell-based vaccine production further accelerates the market by improving operational efficiency and reducing contamination risks. The industry’s shift toward chemically defined and serum-free media ensures the safety and consistency required to meet strict global regulatory standards.
The tissue engineering and regenerative medicine segment is projected to grow at the highest CAGR in the coming years. This is mainly because of the critical need for high-performance, chemically defined media to maintain the viability of complex engineered tissues. The biopharmaceutical R&D surge, where increased funding and regulatory support for stem cell and personalized therapies necessitate robust, specialized cell line development. The market is also seeing a decisive shift toward serum-free and custom formulations, which enhance the safety and reproducibility required for repairing or replacing damaged organs.
End-use Insights
How Does the Large Biopharmaceutical Companies Segment Lead the Cell Culture Media & Cell Lines Market in 2025?
The large biopharmaceutical companies segment led the cell culture media & cell lines market in 2025 due to a global push for high-volume manufacturing of vaccines and recombinant proteins, a massive need for stable, high-quality cell culture media, and optimized cell lines. The strategic multi-billion dollar investment from industry leaders, such as Danaher, to expand production capacity and meet the growing bioprocessing infrastructure in North America and the Asia Pacific. Technological breakthroughs in CHO K-1 cell lines and the shift towards serum-free, cGMP-certified media are setting new benchmarks for high protein expression and regulatory safety.
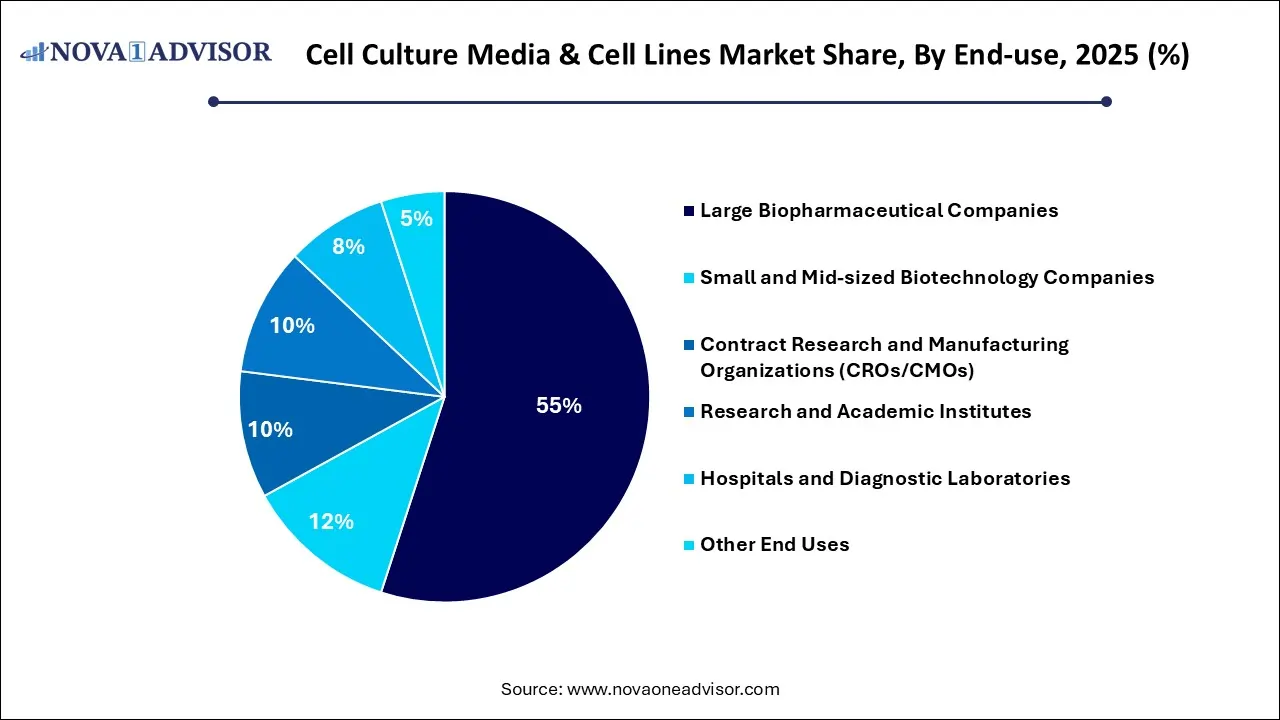
The contract research and manufacturing organizations (CROs/CMOs) segment is projected to grow at the highest CAGR in the coming years. This is mainly because the leveraging external expertise in custom media formulations and high-performance cell line development. By utilizing the advanced infrastructure of these partners, companies can significantly reduce the high capital costs of maintaining in-house, compliant manufacturing facilities while ensuring rapid scalability. The reliance on CRO/CMO partnerships is particularly vital for startups and mid-sized firms, providing them access to automated, high-throughput technologies that accelerate regulatory approval.
Regional Analysis
What Made North America the Dominant Region in the Cell Culture Media & Cell Lines Market?
North America maintained dominance in the cell culture media & cell lines market while holding the largest share in 2025. The region’s dominance is attributed to its substantial R&D funding from both government and private sectors, fueling innovation in high-growth areas, such as cancer biology and regenerative medicine. The early adoption of automated 3D culture systems and bioreactors, alongside a supportive FDA regulatory environment, ensures that these advanced therapies reach the market with high efficiency and safety. The urgent need for personalized treatments for chronic diseases is transforming the market into a high-value sector focused on precision medicine and advanced cell-based assays.
U.S. Cell Culture Media & Cell Lines Market Trends
The U.S. is a major contributor to the market in North America due to the breakthroughs in CAR-T and iPSC therapies, which require specialized, GMP-compliant nutrients to ensure the safety and potency of living drug products. The industry-wide transition toward serum-free and chemically defined formulations is critical for meeting strict regulatory standards and achieving the batch-to-batch consistency needed for commercial success. The integration of 3D organoids and automated bioreactors is revolutionizing the market by providing more accurate human models and streamlining the path from drug discovery to high-throughput production.
What Makes Asia Pacific the Fastest-Growing Area in the Market?
Asia Pacific is expected to grow at the fastest rate in the coming years. This is due to the rising prevalence of chronic and rare diseases, which necessitates the large-scale production of complex biologics, vaccines, and personalized therapies through advanced outsourcing models like CDMOs and CROs. Innovation in specialized research areas, such as 3D organoid cultures and regenerative medicine, is simultaneously driving the need for sophisticated, high-performance media tailored to mimic human physiological environments. The industry’s decisive shift toward serum-free and chemically defined media ensures the safety, yield, and reproducibility required to meet stringent global standards for next-generation drug development.
China Cell Culture Media & Cell Lines Market Trends
China is a key player in the Asia Pacific cell culture media & cell lines market due to the dominance of recombinant cell lines, such as CHO K-1, which are essential for high-yield monoclonal antibody manufacturing. Strategic government initiatives, such as Healthy China 2030, are accelerating these advancements by funding biotech infrastructure and localizing raw material supplies to reduce import reliance. The rise of 3D cell culture and organoids is opening new high-value opportunities by providing more accurate physiological models for drug toxicity screening and personalized medicine.
How is the Opportunistic Rise of Europe in the Cell Culture Media & Cell Lines Market?
Europe is experiencing a strategic growth in the market due to the increasing prevalence of cancer and infectious diseases, necessitating the mass production of biologics and vaccines. Strategic investments in stem cell research and immunotherapy have further catalyzed the market, creating a high-volume demand for specialized media and advanced cell line development. Major industry players like Fujifilm, Irvine Scientific, and Merck KGaA are securing these supply chains through localized manufacturing expansions in Europe to ensure stability and rapid delivery.
United Kingdom Cell Culture Media & Cell Lines Market Trends
The United Kingdom is leading the market in Europe due to the shift toward reproducibility and safety required for large-scale regulatory compliance in manufacturing. The rapid expansion of biologics and cell therapies necessitates high-quality, specialized media to maintain the potency of living drug products. Significant investment in iPSC research and 3D organ-on-chip technology is creating high-value opportunities for tailored formulations that accurately mimic human physiology for drug discovery.
- Raw Material Sourcing & Ingredient Supply
This stage involves the procurement of high-quality components necessary for media formulation, including amino acids, vitamins, salts, growth factors, and serum or animal-derived component alternatives.
Key Players: Merck KGaA (Sigma-Aldrich), Thermo Fisher Scientific, Avantor, FUJIFILM Irvine Scientific, Ajinomoto Co., Inc.
- Media Formulation, Cell Line Development & R&D
Companies develop specialized media formulations tailored for specific cell lines, such as CHO, HEK 293, or stem cells, and engineer cell lines to maximize productivity for biologics and vaccine production.
Key Players: Corning Inc., Lonza Group, Danaher (Cytiva), FUJIFILM Diosynth Biotechnologies, ATCC, WuXi Biologics.
This stage includes the packaging of cell culture media in sterile containers (bottles, bags) and specialized logistics to manage the temperature-sensitive nature of products, particularly serum-based formulations. Efficient cold-chain distribution ensures product stability and timely delivery to pharmaceutical firms and research laboratories globally.
Key Players: Avantor Inc., Thermo Fisher Scientific, Corning Inc., Danaher, Bio-Techne.
- Sartorius AG; Sartorius provides a broad portfolio of cell culture media, including specialty, chemically defined, and serum-free formulations, while offering advanced automated microbioreactor systems (Ambr®) to accelerate cell line development.
- Danaher (Cytiva & Pall); Operating heavily through Cytiva and Pall, Danaher contributes with a strong portfolio of cell culture media and supplements, particularly within the HyClone brand for bioprocessing and industrial-scale applications.
- Merck KGaA; Merck (Merck Millipore) offers a comprehensive line of customized cell culture media, including the Cellvento® brand, designed to meet specific cell-line requirements and optimize cell growth and protein expression. Through its Life Science business, it supports both early-stage research and large-scale manufacturing, including specialized media for stem cell research and viral vector production.
- Thermo Fisher Scientific, Inc.: As a market leader, Thermo Fisher leverages its Gibco portfolio to provide a wide array of cell culture media, serum, reagents, and supplements supporting various cell types in research and commercial manufacturing.
- FUJIFILM Corporation: FUJIFILM contributes to the market through specialized, animal-component-free cell culture media for vaccines, biologics, and cell/gene therapies, aiming to enhance product yield.
Recent Developments
- In February 2025, Cellistic, a leader in iPSC-based cell therapy, officially launched Allo Chassis™, a platform of ready-to-use, immune-cloaked induced pluripotent stem cell (iPSC) lines designed to drastically reduce the cost and time required for cell therapy development. This involves cell lines that represent a substantial leap forward in cell line development, effectively reducing both timelines and costs.
- In April 2025, Thermo Fisher Scientific announced a major enhancement to its biologics development platform, introducing a high-performance CHO K-1 cell line and integrated "Gene to Patient" services. This launch specifically targets the logistical bottlenecks in pre-clinical drug development to get therapies to patients faster.
- My 2025, German life science leader PL BioScience and Korean biotech innovator DewCell Biotherapeutics signed a Letter of Intent (LOI) to co-develop the world’s first artificial human platelet lysate (HPL). This partnership aims to revolutionize the market by providing a high-performance, completely animal-free alternative to traditional cell culture media.
Exclusive Analysis on the Cell Culture Media & Cell Lines Market
The global cell culture media & cell lines market is leveraged by the surging demand for biopharmaceuticals (monoclonal antibodies and vaccines), rising cell/gene therapy clinical trials, and advanced regenerative medicine research. Key market drivers include the urgent need for consistent, scalable, and contamination-free production, leading to a shift toward serum-free and chemically defined media to meet stringent GMP standards. Segmentally, liquid media dominate in form due to convenience in large-scale production, while stem cell culture media is growing at the highest CAGR. The biopharmaceutical segment leads in application, supported by increased R&D funding for chronic disease therapeutics.
Segments Covered in the Report
By Product
- Traditional Cell Lines
- Classical Media
- Stem Cells & Blood-derived Cells
- Specialty Media Products
By Application
- Biopharmaceutical Production
- Diagnostics
- Drug Screening and Development
- Tissue Engineering and Regenerative Medicine
- Other Applications
By End-use
- Large Biopharmaceutical Companies
- Small and Mid-sized Biotechnology Companies
- Contract Research and Manufacturing Organizations (CROs/CMOs)
- Research and Academic Institutes
- Hospitals and Diagnostic Laboratories
- Other End Uses
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa




