Cell Culture Supplements Market Size and Growth 2026 to 2035
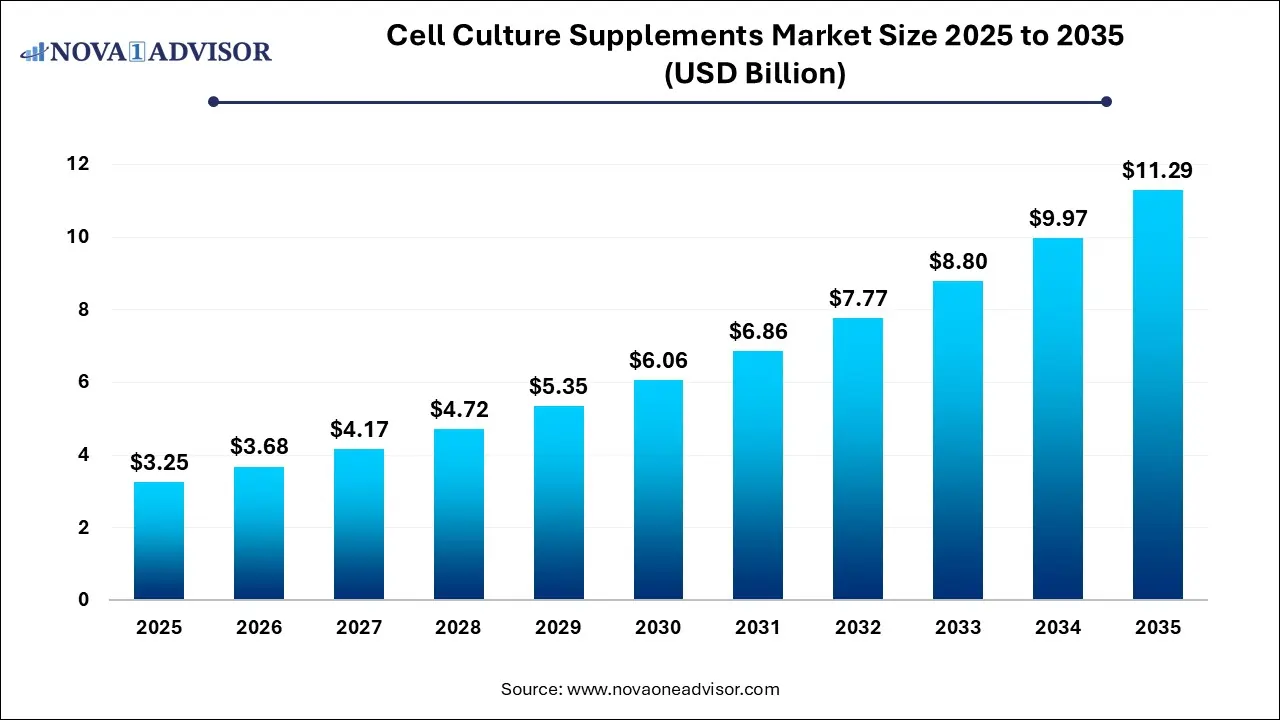
The global cell culture supplements market size was estimated at USD 3.25 billion in 2025 and is predicted to increase from USD 3.68 billion in 2026 to approximately USD 11.29 billion by 2035, expanding at a CAGR of 13.26% from 2026 to 2035. The cell culture supplements market is driven by the increasing prevalence of chronic diseases, escalated demand for biologics, requiring high-volume manufacturing, more supplements for optimization, rising cell and gene therapies, and innovation in cell culture technologies.
Key Takeaways
- By region, North America dominated the global cell culture supplements market with a share of 40% in 2025.
- By region, the Asia Pacific is expected to grow at the fastest rate in the market during the forecast period.
- By product, the serum-based supplements segment held a dominant position in the market with a share of 30% in 2025.
- By product, the chemically defined supplements segment is the fastest growing between 2026 and 2035.
- By application, the biopharmaceutical manufacturing segment accounted for a considerable revenue share of 40% in the market in 2025.
- By end-use, the cell culture media manufacturers segment is the dominant in the global cell culture supplements market with a share of 35% in 2025.
Cell culture supplements, which comprise recombinant proteins, growth factors, and serum replacements, are essential additives used to enhance cell growth, viability, and productivity in both laboratory research and industrial-scale biopharmaceutical production. The improved lot-to-lot consistency, reduced contamination risks, and optimized cell proliferation for applications, such as monoclonal antibody production, vaccine development, and stem cell research. Market growth is driven by increasing investment in cell-based therapies, the shift toward animal-free, chemically defined media, and the expansion of 3D cell culture organoid technologies.
Cell Culture Supplements Market Trends
- Shift Toward Serum-Free and Chemically Defined Media
The industrial trend toward animal-derived serum, such as fetal bovine serum, is toward chemically defined, animal-free supplements. These alternatives offer higher batch-to-batch consistency, reduce the risk of adventitious agent contamination, and improve reproducibility, which is critical for strict regulatory standards.
- Rapid Adoption of Recombinant Proteins and Growth Factors
The growing demand for recombinant growth factors, cytokines, and proteins produced via genetic engineering. These high-purity components provide specialized nutrition for complex cell lines and stem cells, increasing productivity and supporting GMP-compliant biopharmaceutical manufacturing.
- Integration in 3D Cell Culture and Organoid Specialized Supplements
The industry shift from 2D monolayers to 3D culture models and organoids, which better mimic human tissue physiology, is driving the need for specialized supplements, extracellular matrix proteins, and nutrient-rich media. These additives are crucial for maintaining viability and differentiation in advanced disease modeling and regenerative medicine applications.
Key AI Integration in the Cell Culture Supplements Market
According to Sartorius, AI and ML are transforming the cell culture supplements industry by enabling the shift from labour-intensive trial-and-error methods to data-driven, precise media optimization. Advanced ML algorithms, such as Bayesian optimization and deep learning, analyse vast datasets to identify optimal combinations of growth factor and nutrients, accelerating the development of custom-tailored media with higher protein titers. AI facilitates the development of sustainable, serum-free, and animal-free supplements by predicting how cells behave in complex, non-traditional formulations. In manufacturing, AI-driven automation and real-time monitoring of critical environmental parameters (pH, nutrient levels) enhance consistency and reduce batch variability.
Cell Culture Supplements Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 3.68 Billion |
| Market Size by 2035 |
USD 11.29 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 14.26% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
Product, Application, End use, Region |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
Merck KGaA; Thermo Fisher Scientific Inc.; HiMedia Laboratories; Danaher; Sartorius AG; Corning Inc.; R&D Systems (Bio-Techne); STEMCELL Technologies; Repligen Corporation; Proteintech Group, Inc. |
Market Dynamics
Driver
Rising Demand for Biologics and Biosimilars
The growing need for cost-effective treatments for chronic diseases, for the cell culture supplements market. As patents expire for blockbuster biologics, the increased production of biosimilars necessitates high-performance, consistent media components to ensure structural and functional similarity to the reference products. Manufacturers are increasingly shifting toward chemically defined and animal-free supplements to ensure batch-to-batch consistency and regulatory compliance. The expansion of biologics, particularly monoclonal antibodies and cell therapies, demands higher cell densities and productivity, which is fulfilled by specialized nutrients and growth factors, thus boosting the supplements market.
Restraint
High Cost of Product
The market growth is hindered by limiting widespread adoption among budget-constrained academic and early-stage biotechnology laboratories. These high prices, which stem from complex manufacturing, specialized raw materials, and strict quality compliance, increase overall research and production expenses. Many researchers are prompted to cut usage or opt for lower-grade alternatives, which affects experimental reproducibility and product performance. This economic burden is a primary restraint on adopting advanced serum-free media, especially in developing regions where high-cost imports are not feasible.
Opportunity
Development of Novel Animal-Free Supplements
The addressing critical demands for safety, consistency, and ethical sourcing in biopharmaceutical production. By replacing traditional, variable animal-derived components, such as Fetal Bovine Serum, with chemically defined plant-based or recombinant alternatives, manufacturers can eliminate the risk of adventitious agents and contamination, boosting market confidence. These innovative, high-performance supplements improve cell growth rates and product yields, offering superior reproducibility, which is essential for regulatory compliance in clinical and commercial applications.
Segment Insights
By Product Insights
Why Did the Serum-Based Supplements Segment Hold a 30% Share for the Cell Culture Supplements Market?
The serum-based supplements segment held a dominant position in the market with a share of 30% in 2025, owing to the widespread use of serum-based products in cell cultures for their rich content of growth factors. The rising interest in serum-free alternatives in biopharma, the high demand in regenerative medicine, and the widespread use in various applications.
The protein-based & recombinant supplements segment held a 26% share of the market in 2025. Due to the integration supplements being protein-based, including recombinant proteins and peptides for cell culture. The strategic transition to animal-origin-free and chemically defined media is critical for mitigating contamination risks and ensuring the
The chemically defined supplements segment held a 20% share of the market in 2025, driven by the strategic shift toward animal-origin-free and chemically defined media, which is essential for ensuring the high-purity and safety standards mandated by global regulatory bodies. The supplements with precise chemical compositions are ideal for controlled cell culture environments.
The others segment held a 24% share of the market in 2025, driven by the industry's transition toward chemically defined and animals-free system, the need to eliminate contamination risks, and meet the rigorous safety mandates of the EDA and EMA. The variety of niche applications across different industries fuels the market growth.
By Application Insights
Why Did the Biopharmaceutical Manufacturing Segment Lead the Cell Culture Supplements Market?
The biopharmaceutical manufacturing segment registered its dominance over the global market with a share of 40% in 2025, owing to the large-scale demand for biologics and vaccine production in the pharma industry. Growing investment in manufacturing capacity by contract development and manufacturing organizations and vaccine manufactures accelerate the need for these supplements, with a rapid rise in advanced therapeutics.
The cell & gene therapy segment held the 25% share of the market in 2025, due to the rapid advancements and demand for novel therapies, growing expansion of clinical trials, and commercial approvals for cell and gene therapies create a massive need for GMP-compliant reagents, supplements, and serum. Massive funding from investment and research, and a shift towards serum-free media.
The drug discovery segment held a 20% share of the market in 2025 because of the cell culture's major role in testing and discovering new drugs and therapies. The strategic adoption of 3D models and organoids provides the physiological relevance necessary for advanced drug discovery and toxicity testing.
The others segment held a 15% share of the market in 2025 due to the applications outside biopharma, cell therapy, and drug discovery. The shift from 2D to 3D cell culture systems requires specialized extracellular matrix proteins and defined additives, expansion of serum-free alternatives.
By End-Use Insights
Why Did the Cell Culture Media Manufacturers Segment Hold a 35% Share for the Cell Culture Supplements Market?
The cell culture media manufacturers segment led the global market with a share of 35% in 2025, due to the manufacturing companies that produce the media for cell culture in laboratories and manufacturing environments. The massive biopharmaceutical manufacturing expansion and rise in drug developments are increasingly outsourcing media requirements, fuelling growth among specialized suppliers.
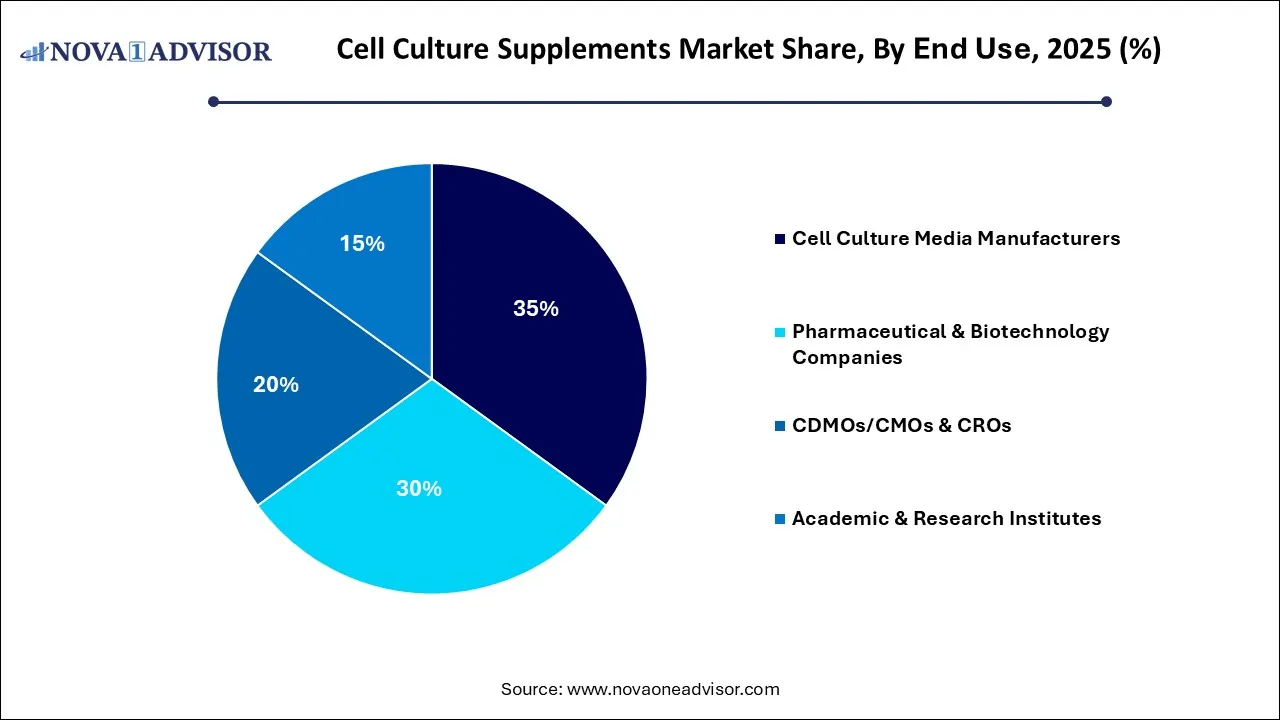
The pharmaceutical & biotechnology companies segment held a 30% share of the market in 2025, with the firms that use cell culture for research, development, and manufacturing of drugs and biologics. The demand for high-performance, scalable cell culture supplements is increasing to ensure commercial-grade yields. The specialized additives provide the critical infrastructure for the next generation of precision medicine and large-scale biopharmaceutical manufacturing.
The CDMOs/CMOs & CROs segment held a 20% share of the market in 2025, due to growing outsourcing trends to contract service providers, rising need for specialized expertise, and technology. CDMO ensures regulatory alignment with FDA/EMA standards, enabling faster product launches and managing the high-purity requirements for cell culture products.
The academic & research institutes segment held a 15% share of the market in 2025, due to academic institutions and research centers utilizing cell culture for scientific studies. Growing research into cancer and other chronic diseases requires specialized media components, accelerating the consumption of high-quality supplements in research settings. Rising use of specialized cells and expansion of research and development.
By Region Insights
Why Did North America Lead the Global Cell Culture Supplements Market?
North America dominated the global cell culture supplements market with a share of 40% in 2025, supported by the vast number of pharmaceutical companies and cutting-edge research, securing the market. The robust expansion of advanced biopharmaceutical manufacturing, particularly for CAR-T cell therapies, has led to the adoption of high-performance cell culture supplements. The rising trend of outsourcing to CDMOs further amplifies the demand for these specialized, clinical-grade components to support scalable, high-volume production.
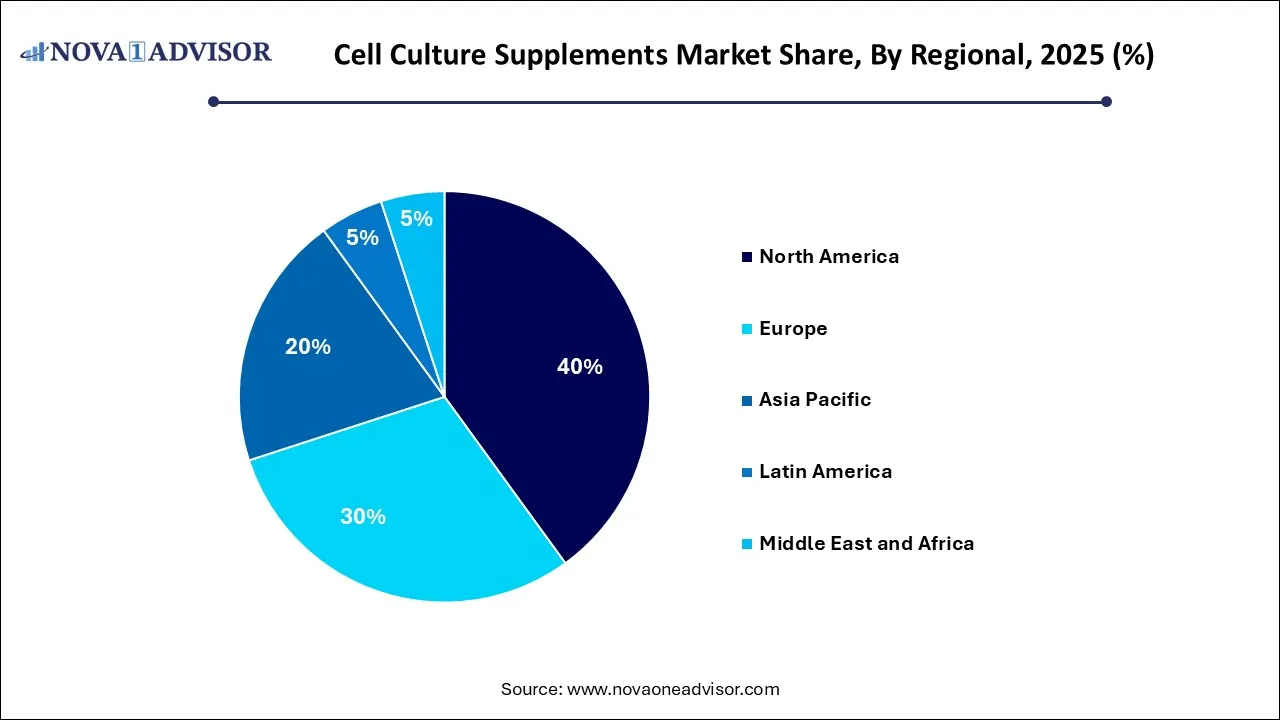
Why is Asia Pacific undergoing the Fastest Growth in the Cell Culture Supplements Market?
Asia Pacific is expected to be the fastest-growing region during the forecast period, due to rapid growth in the biotech and healthcare sectors, emerging as a key player in drug production and biotech. The region’s combination of cost-effective production and local-global collaborations has established it as a premier hub for the scalable, reliable manufacturing of next-generation biopharmaceuticals. To ensure global regulatory compliance and therapeutic consistency, manufacturers are aggressively transitioning to chemically defined and animal-free media, eliminating the contamination risks of traditional serum.
How Is Europe Driving Growth in the Cell Culture Supplements Market?
Europe is expected to be a significantly growing region during the forecast period, with rapid development of advanced therapy medicinal products (ATMPs) and high-yield viral vector manufacturing. Strategic government initiatives, such as the UK life sciences vision and Germany’s national pharma strategy, are catalyzing the infrastructure required for the large-scale adoption of chemically defined and animal-component-free (ACF) media. A well-established pharmaceutical market and significant research output.
Cell Culture Supplements Market Companies
- Merck KGaA
- HiMedia Laboratories
- Thermo Fisher Scientific Inc.
- Danaher
- Corning Inc.
- Sartorius AG
- R&D Systems (Bio-Techne)
- Repligen Corporation
- STEMCELL Technologies
- Proteintech Group, Inc.
Recent Developments
- In December 2025, Ajinomoto CELLiST Korea launched CELLiST™ AAV Production Supplement 1, designed to increase viral vector productivity by 1.3 to 2 times for gene therapy manufacturing. Developed in collaboration with Forge Biologics, the supplement enhances yield for HEK293 cell lines to address growing demand in the gene therapy market.
- In May 2025, PL BioScience partnered with DewCell Biotherapeutics to develop the world's first artificial Human Platelet Lysate (HPL), a lab-grown, scalable cell culture supplement designed to replace animal-derived sera in cell therapy manufacturing. This xeno-free, proprietary solution, introduced ISCT conference, offers enhanced performance and consistency compared to natural HPL, addressing the supply limitations of donor-derived products.
- In November 2025, The ALS Therapy Development Institute (ALS TDI), LifeArc, and Axol Bioscience have launched PRISM ALS, a global initiative providing high-quality, patient-derived induced pluripotent stem cell (iPSC) models to better reflect the biological diversity of sporadic ALS. By offering diverse, well-characterized models from the ALS Research Collaborative (ARC) Study, this collaboration aims to accelerate drug discovery, improve clinical trial success rates, and advance precision medicine for the 85% of cases that are not inherited.
Cell Culture Supplements Market Segment
By Product
- Serum-Based Supplements
- Protein-Based & Recombinant Supplements
- Chemically Defined Supplements
- Others
By Application
- Biopharmaceutical Manufacturing
- Cell & Gene Therapy
- Drug Discovery
- Others
By End Use
- Cell Culture Media Manufacturers
- Pharmaceutical & Biotechnology Companies
- CDMOs/ CMOs & CROs
- Academic & Research Institutes
By Regions
- North America
- Europe
- Asia Pacific
- Middle East & Africa
- Latin America




