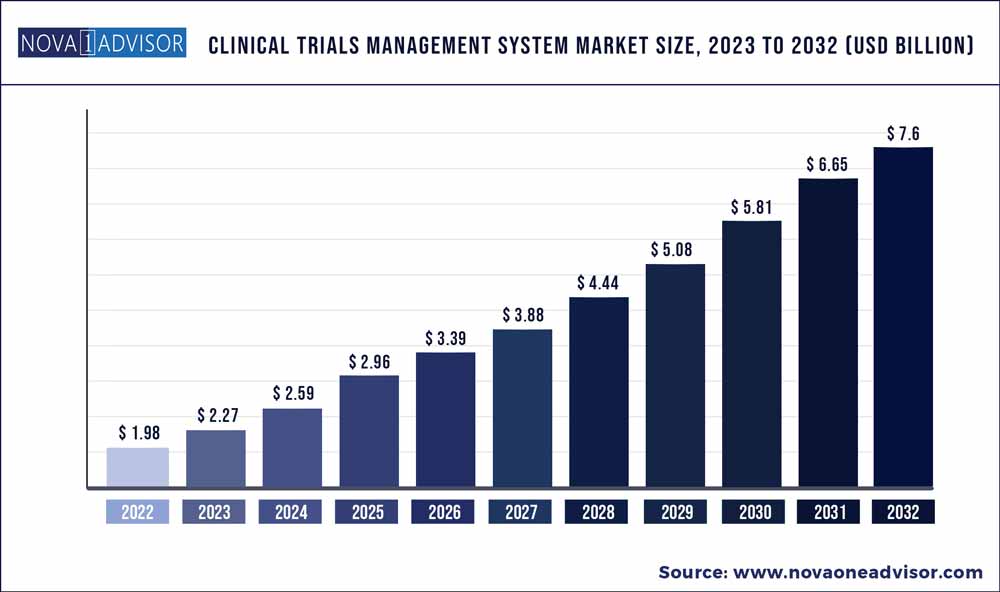
The global clinical trials management system market size was exhibited at USD 1.98 billion in 2022 and is projected to hit around USD 7.6 billion by 2032, growing at a CAGR of 14.4% during the forecast period 2023 to 2032.
Key Pointers:
- North America held about 51.1% of the revenue share of the Clinical Trials Management System (CTMS) market in 2022.
- Asia Pacific is estimated to attain the fastest CAGR of about 18.3% in the next few years.
- Web & cloud-based systems held the largest share in 2022 owing to benefits such as remote access to data and minimal technical issues.
- The segment is also anticipated to exhibit the fastest growth over the forecast period.
- The highest share of the global Clinical Trials Management System (CTMS) market, in terms of components, was held by the software segment in 2022. The services segment on the other hand is expected to grow at the fastest rate of over 14% in the coming years.
- Enterprise accounted for the largest share of the Clinical Trials Management System (CTMS) market by solution type in 2022 and is also expected to expand at the fastest CAGR of about 14.2% over the forecast period.
- By end-user, the CROs & others segment held the largest share of over 35.19% of the Clinical Trials Management System (CTMS) market in 2022.
Clinical Trials Management System Market Report Scope
| Report Coverage |
Details |
| Market Size in 2023 |
USD 2.27 Billion |
| Market Size by 2032 |
USD 7.6 Billion |
| Growth Rate From 2023 to 2032 |
CAGR of 14.4% |
| Base Year |
2022 |
| Forecast Period |
2023 to 2032 |
| Segments Covered |
Solution type, Delivery mode, Component, End-user |
| Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
IQVIA Inc.; Medidata (Dassault Systèmes); Oracle; DATATRAK International, Inc.; Clario; SimpleTrials; Calyx; RealTime Software Solutions, LLC; Laboratory Corporation of America Holdings; Veeva Systems; Wipro Limited; PHARMASEAL International Ltd.
|
The rapid growth of healthcare IT, preference for decentralized clinical trials, initiatives by key companies, and an increasing number of clinical studies are anticipated to drive the growth of the market. According to the WHO, about 65,409 clinical trials were conducted in 2020 across the globe. This number was notably greater than the 60,543 trials conducted in 2019. This is estimated to boost demand for CTMS solutions.
The COVID-19 pandemic significantly impacted the Clinical Trials Management System (CTMS) market. This included disruption in clinical trials, hurdles in patient recruitment, and canceled or delayed studies. However, the negative impact was gradually reduced by several strategic measures taken by governments, regulatory bodies, and market stakeholders to ensure the continuity of R&D. The need to develop vaccines for the coronavirus also boosted the adoption of decentralized clinical trials. The International Federation of Pharmaceutical Manufacturers & Associations (IFPMA) released guidelines to urgently advance COVID-19 vaccine equity. The IFPMA reported that 5 of its members had received approval for COVID-19 vaccines while 14 more were under clinical development. Also, as per COVID-NMA (an initiative by WHO), over 750 studies were registered for COVID-19 vaccine development as of February 2022. This is expected to increase the adoption of CTM systems.
As per a study published in the U.S. National Library of Medicine in 2020, the pandemic resulted in delayed subject enrollment and operational gaps in most ongoing clinical trials. This in turn negatively impacted trial programs and data integrity. Globally, most sites conducting clinical trials other than COVID-19 were found to experience delays in timelines. In some cases, clinical trials were found to reach a complete halt of operations, thus impacting clinical research outcomes. In March 2020, the U.S. FDA published a guidance document on the Conduct of Clinical Trials of Medical Products during the pandemic. Similar guidelines supported the recovery of the clinical trial operations, and the number is anticipated to increase over time.
Rising government funding and investments by biotechnology and pharmaceutical firms are promoting medical research activities. This factor combined with technological advancements is anticipated to boost the market growth over the forecast period. For instance, cloud-based CTMS eliminates the expenses of hardware acquisition, installation, provisioning, maintenance, support, and software licensing. These systems automatically update software and patch management systems, which reduces the burden of in-house IT staff and saves costs. Furthermore, the cloud-based software enables access to the server through mobile with maximum data security.
The increasing number of decentralized clinical trials is anticipated to boost market growth in the coming years. These are also referred to as virtual, digital, mobile, siteless, and remote trials that often utilize telemedicine solutions. Labcorp's decentralized clinical trials (DCTs) solution for instance offers a suite of solutions to design and implement decentralized clinical trials, including the necessary infrastructure, technology, and services. Increasing product availability from key companies is also estimated to fuel market growth. Rave CTMS by Medidata, for instance, offers an intelligent automation and workflow management solution to efficiently manage and oversee clinical trials.
Clinical Trials Management System Market Segmentation
| By Solution Type |
By Delivery Mode |
By Component |
By End-user |
|
|
- Web & Cloud-based
- On-premise
|
|
- Pharmaceutical and Biotechnology Firms
- Medical Device Firms
- CROs & Others
|


