Deep Brain Stimulation Devices Market Size, Growth and Trends 2026 to 2035
The global deep brain stimulation devices market size was estimated at USD 1.61 billion in 2025 and is expected to be worth around USD 4.26 billion by 2035 with a CAGR of 10.23% from 2026 to 2035.
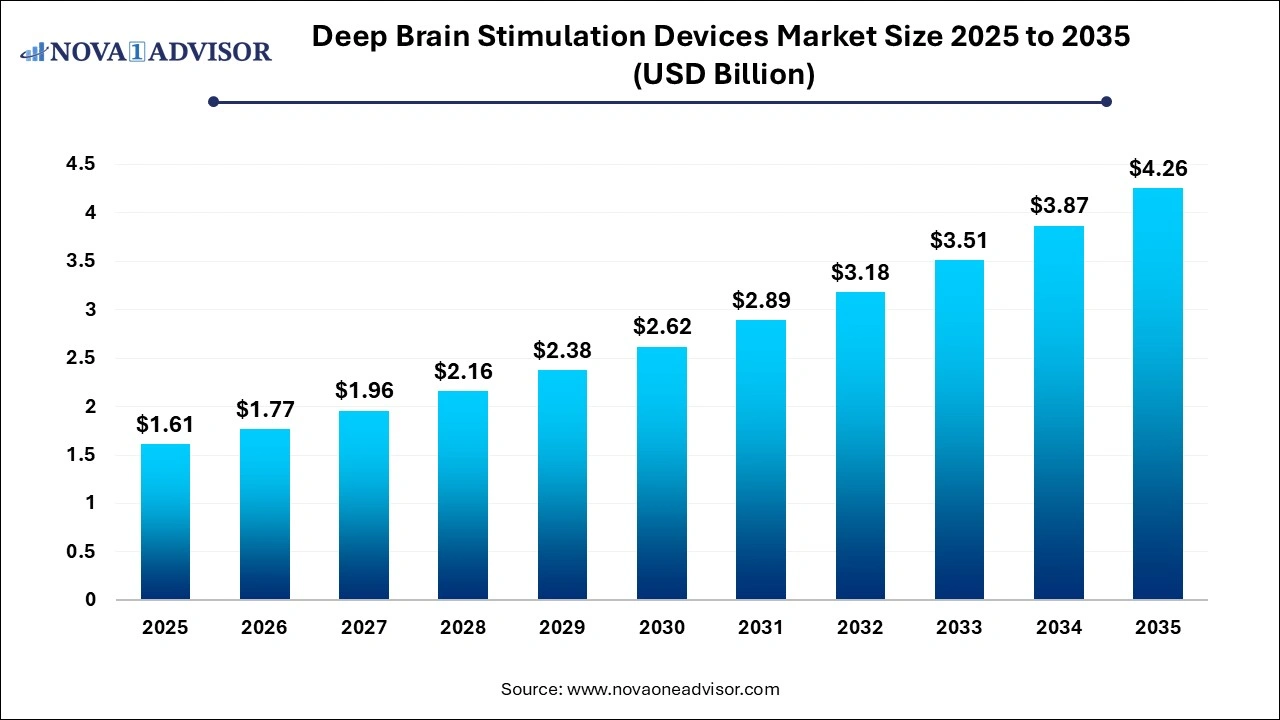
Deep Brain Stimulation Devices Market Outlook
- Market Growth Overview: The deep brain stimulation devices market is expected to grow significantly between 2026 and 2035, driven by the rising prevalence of neurological disorders, the growing geriatric population, and innovation in technological advancements.
- Sustainability Trends: Sustainability trends involve the rise of rechargeable implantable pulse generators, extended battery lifespan, and the development of self-sustainable systems.
- Major Investors: Major investors in the market include Medtronic, Boston Scientific, Abbot Laboratories, NeuroPace, Inc., and Abbott.
How AI is Impacting the Deep Brain Stimulation Devices Market
AI and ML are transforming the deep brain stimulation devices industry by shifting from static, constant stimulation to adaptive, personalized, closed-loop systems that automatically adjust to a patient’s real-time brain activity. These advanced algorithms analyze local field potentials and patient-specific imaging data to optimize stimulation, reducing unwanted side effects and improving symptom management for Parkinson's disease and other neurological conditions. By enabling remote programming, AI improves accessibility and reduces the need for in-clinic visits, boosting the market for next-generation, rechargeable devices.
Supply Chain Analysis of the Deep Brain Stimulation Devices Market
- R&D and Clinical Trials: This initial stage focuses on the development of new, more efficient, and smaller neurostimulators, including rechargeable batteries and advanced, steerable leads.
Key Players: Medtronic, Abbott Laboratories, Boston Scientific, Aleva Neurotherapeutics SA, Nexstim, NeuroPace Inc., Newronika.
- Raw Material Sourcing & Component Manufacturing: This stage involves sourcing high-grade, biocompatible materials, such as titanium for the pulse generator housing and platinum-iridium for the electrodes.
Key Players: Medtronic and Boston Scientific.
- Regulatory Approval & Quality Assurance: This crucial phase involves acquiring regulatory certifications (CE Mark, FDA approvals) to ensure compliance with global healthcare standards.
Key Players: Medtronic, Boston Scientific, Abbott Laboratories, Aleva Neurotherapeutics.
Report Scope Deep Brain Stimulation Devices Market
| Report Coverage |
Details |
| Market Size in 2026 |
USD 1.77 Billion |
| Market Size by 2035 |
USD 4.26 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 10.23% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product, By Application, By End-use |
| Market Analysis (Terms Used) |
Value (USDMillion/Billion) or (Volume/Units) |
| Key Companies Profiled |
Medtronic, Abbott, Aleva Neurotherapeutics SA |
Deep Brain Stimulation Devices Market Segmental Insights
By Product Insights
The dual channel deep brain stimulation device segment dominated the market in 2025 owing to higher adoption in surgical procedures. Dual deep brain stimulation devices are one of the safest and most effective devices used in the surgical procedures. Thus, increasing number of surgical procedures for Parkinson's disease, prevalence of disabling neurological diseases, and increasing number of hospitals using dual channel deep brain stimulation devices systems are driving the market growth.
The single channel deep brain stimulation device segment is expected to expand at the highest CAGR during the forecast period. Healthcare professionals believe that single channel offers more programming options to the neurologists, which led to increased preference for single channel deep brain stimulation device. According to the studies conducted by the Parkinson’s Foundation, geriatric population are more prone to neurological diseases such as PD. Increasing geriatric population, growing awareness about neurological diseases among patients, and preference by healthcare professionals are anticipated to trigger the segment growth.
By Application Insights
Based on application, the Parkinson's disease segment dominated the market in 2025. In December 2017, Boston Scientific Corporation received approval for the Vercise deep brain stimulation device system from the U.S. FDA for the treatment of PD. According to the Parkinson’s Foundation, around 60,000 people in the U.S. are diagnosed with PD every year. Thus, increasing number of U.S. FDA approvals for deep brain stimulation therapies and increasing prevalence of PD worldwide are driving the segment growth.
The Obsessive Compulsive Disorder (OCD) segment is expected to register the highest CAGR during the forecast period. Increasing incidence of OCD and less effective therapeutics such as serotonin reuptake inhibitors are anticipated to drive the demand for treatment options with long-term effects. Deep brain stimulation therapy for the treatment of treatment-resistant and severe OCD is approved in the European Union, Australia, and U.S. through Humanitarian Device Exemption (HDE) thus, fueling the market growth.
By End-use Insights
In the end-use segment, hospitals dominated the overall deep brain stimulation devices market growth in 2025,owing to use of technologically advanced deep brain stimulation devices in operation theaters and intensive care units, increasing number of deep brain stimulation surgeries, rising prevalence of PD. Moreover, favorable reimbursement criteria is expected to further boost the growth of hospital end-use segment.
The ambulatory surgical centers (ASC) segment is expected to grow at the highest rate owing to lower cost as compared to hospitals, convenient access for patient care, reduced waiting time, and low infection rate as compared to neurology clinics and hospitals. Furthermore, according to a study conducted by Advancing Surgical Care, 92% of patients were satisfied with the medical care and service provided in the ASCs, thus, boosting the segment growth.
Deep Brain Stimulation Devices Market Regional Insights
North America dominated the market owing to increase in FDA approvals for deep brain stimulation devices in clinical applications. For instance, in 2025, the U.S. FDA approved a deep brain stimulation device, Brio, for the treatment of Parkinson’s disease. Furthermore,increasing number of patients diagnosed with PD has led to the growth of overall market in the region.
Asia Pacific is expected to expand at the highest CAGR over the forecast period due to factors such as rising prevalence of neurodegenerative disorders coupled with unmet demand for effective and long term solutions. Rising awareness about neurological disease treatment options and improvements in clinical development framework of emerging economies are expected to drive the market growth in this region.
Deep Brain Stimulation Devices Market Share Insights
Major players in the industry include Medtronic, Abbott, Aleva Neurotherapeutics SA, and Boston Scientific Corporation. These key market players are focusing on launching innovative medical devices, building growth strategies, and technological advancements to strengthen their position in the global market. For instance, in October 2019, Medtronic launched its Activa neurostimulators for the treatment of patients suffering from essential tremor and associated symptoms. These advancements are anticipated to boost the market growth over the forecast period.
Key Players in the Deep Brain Stimulation Devices Market
- Medtronic: As a global leader in the field, Medtronic dominates the market with its Percept™ family of neurostimulators featuring BrainSense™ technology, which enables, records, and analyzes brain signals to personalize therapy for movement disorders and epilepsy.
- Abbott: Abbott contributes significantly to the market with its Infinity™ and Liberta RC™ DBS systems, notably launching the world's smallest rechargeable device that features remote programming capabilities via the NeuroSphere™ Digital Health platform.
- Aleva Neurotherapeutics SA: Aleva focuses on advancing the market by utilizing MEMS (Micro-ElectroMechanical System) technology to develop next-generation electrodes, such as the CE-marked directSTIM™ system, which offers superior precision in treating Parkinson's disease and essential tremors.
Segments Covered in the Report
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2026 to 2035. For the purpose of this study, has segmented the global deep brain stimulation devices market report on the basis of product, application, end use and region:
-
By Product
-
Single Channel
-
Dual Channel
-
By Application
-
By End-use
-
By Regional

