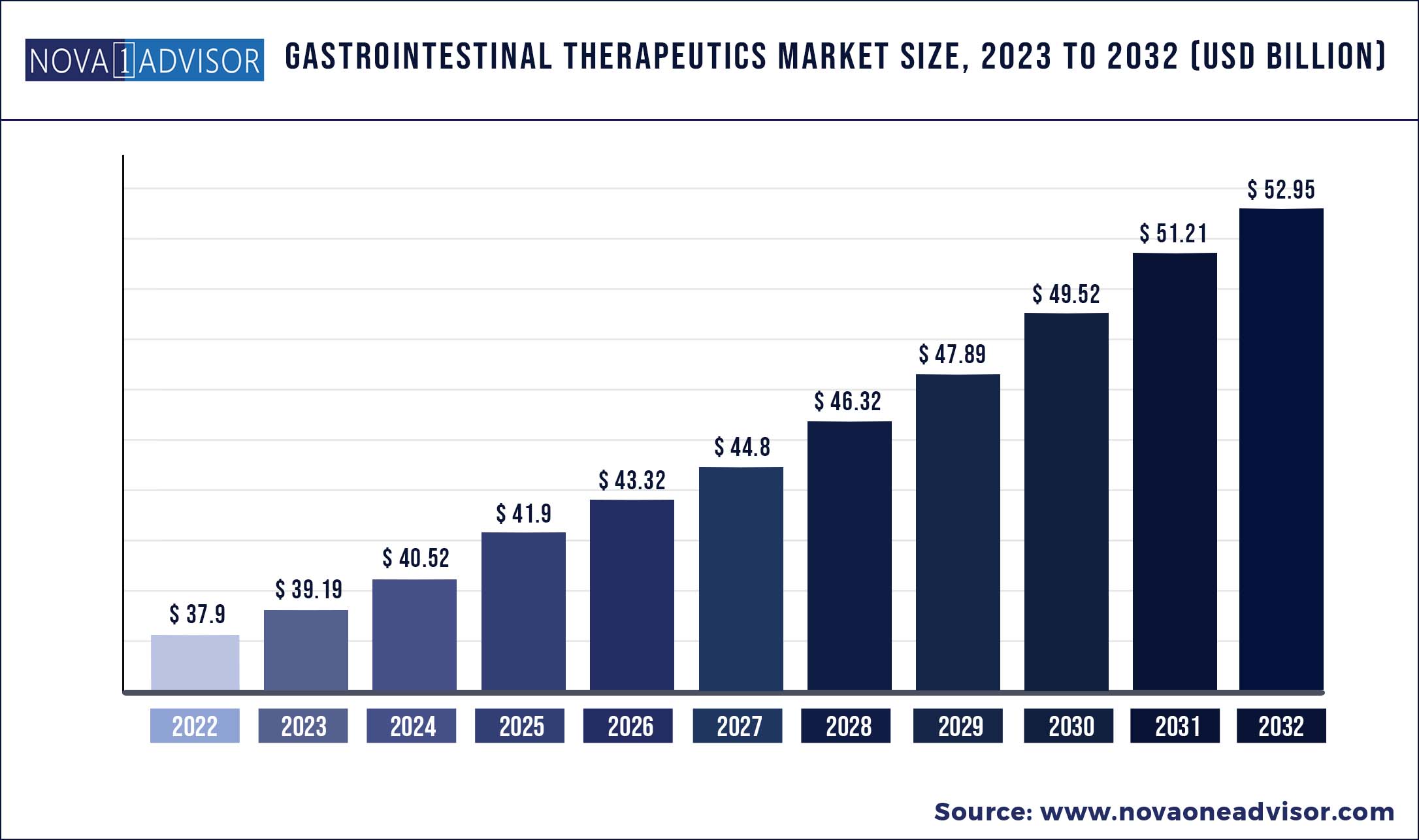
The global gastrointestinal therapeutics market size was exhibited at USD 37.9 billion in 2022 and is projected to hit around USD 52.95 billion by 2032, growing at a CAGR of 3.4% during the forecast period 2023 to 2032.
Key Pointers:
- North America led the overall market for gastrointestinal therapeutics with 37.69% of the revenue share in 2022
- Asia Pacific is projected to witness the fastest growth rate of 5.2% during the forecast period.
- The branded segment held the largest share of 67.90% in 2022
- The biologics/biosimilar segment dominated the market for gastrointestinal therapeutics with a 44.22% revenue share in 2022.
- In 2022, the injectable segment held the dominant market share of 51.19%.
- The gastrointestinal therapeutics market, accounting for 32.96% of the revenue share.
- Ulcerative colitis (UC) is anticipated to be the fastest-growing application segment with 4.6% CAGR during the projection period.
- The retail pharmacies segment captured the largest revenue share of 48.14% in 2022.
Gastrointestinal Therapeutics Market Report Scope
|
Report Coverage
|
Details
|
|
Market Size in 2023
|
USD 39.19 Billion
|
|
Market Size by 2032
|
USD 52.95 Billion
|
|
Growth Rate from 2023 to 2032
|
CAGR of 3.4%
|
|
Base year
|
2022
|
|
Forecast period
|
2023 to 2032
|
|
Segments covered
|
Type, Drug class, Application, Distribution channel, Route of administration
|
|
Regional scope
|
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa
|
|
Key companies profiled
|
AbbVie Inc.; AstraZeneca; Janssen Pharmaceuticals NV; Sun Pharmaceutical Industries Ltd.; Takeda Pharmaceutical Company Limited; Salix Pharmaceuticals; Pfizer Inc.; Bayer AG; Abbott; Cipla Inc.; Gilead Sciences, Inc.; Biogen; Organon Group of Companies
|
Unhealthy eating habits, coupled with a sedentary lifestyle, have led to the development of gastrointestinal diseases among consumers, which is expected to drive market growth. Moreover, the rising adoption of biologics for the treatment of GI disorders is also fueling growth. For instance, REMICADE (Infliximab) by Janssen Biotech, Inc., and other biological drugs have been approved for the treatment of Crohn’s disease and ulcerative colitis.
The increasing incidences of industry-academia collaborations to promote gastrointestinal research and develop novel therapies for patients with GI disorders will likely facilitate market expansion. For instance, in November 2020, Takeda Pharmaceutical Company Limited, Columbia University, and New York University formed a collaborative research alliance to advance gastroenterology research programs, to develop new therapeutics for gastrointestinal diseases.
Moreover, market growth is also anticipated to increase research & development initiatives by government and private organizations for the development of new gastrointestinal drugs. As of November 2022, according to the U.S. National Library of Medicine, there were 7,586 ongoing clinical studies (including observational studies, drug development, and others) related to gastrointestinal diseases, at various stages of development. Moreover, in May 2022, AbbVie Inc. announced positive topline results from a phase-3 study that is evaluating upadacitinib in adult patients with severe to moderate Crohn's disease, who were intolerant to biologic therapy.
The presence of government and non-profit organizations that support gastrointestinal research, offer to fund studies, and promote awareness about GI disorders, is anticipated to augment GI therapeutics market growth. For instance, the International Foundation for Gastrointestinal Disorders, GI- research foundation, and American Gastroenterological Association are some of the key organizations supporting research activities and increasing awareness about gastrointestinal diseases among people. These organizations also raise funds for ongoing GI research activities.
Furthermore, various strategic initiatives such as product launches, collaboration, mergers, and acquisitions undertaken by leading players are expected to offer lucrative opportunities for market growth. For instance, in July 2022, Eli Lilly announced a partnership with the Chinese drug 3D printing firm to develop and research 3D-printed oral drugs for the GI tract application.
In addition, in April 2020acquired global rights, excluding Canada, Europe, and Israel, of Movantik (naloxegol) by AstraZeneca, for the management of opioid-induced constipation. This acquisition is expected to strengthen the company’s position in the global gastrointestinal therapeutics industry.
Some of the prominent players in the Gastrointestinal Therapeutics Market include:
- AbbVie Inc.
- AstraZeneca
- Salix Pharmaceuticals
- Takeda Pharmaceutical Company Limited
- Pfizer Inc.
- Bayer AG
- Abbott
- Janssen Pharmaceuticals NV
- Sun Pharmaceutical Industries Ltd.
- Cipla Inc.
- Gilead Sciences, Inc.
- Biogen
- Organon Group of Companies
Segments Covered in the Report
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2032. For this study, Nova one advisor, Inc. has segmented the global Gastrointestinal Therapeutics market.
By Type
By Drug Class
- Aminosalicylates
- Digestive enzymes
- Proton Pump Inhibitors
- Laxatives
- Anti-Emetics
- H2 Antagonists
- Anti-Diarrheal
- Biologics/Biosimilar
- Others
By Route of Administration
By Application
- Crohn's disease
- Ulcerative colitis
- GERD
- IBS
- Others
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa (MEA)


