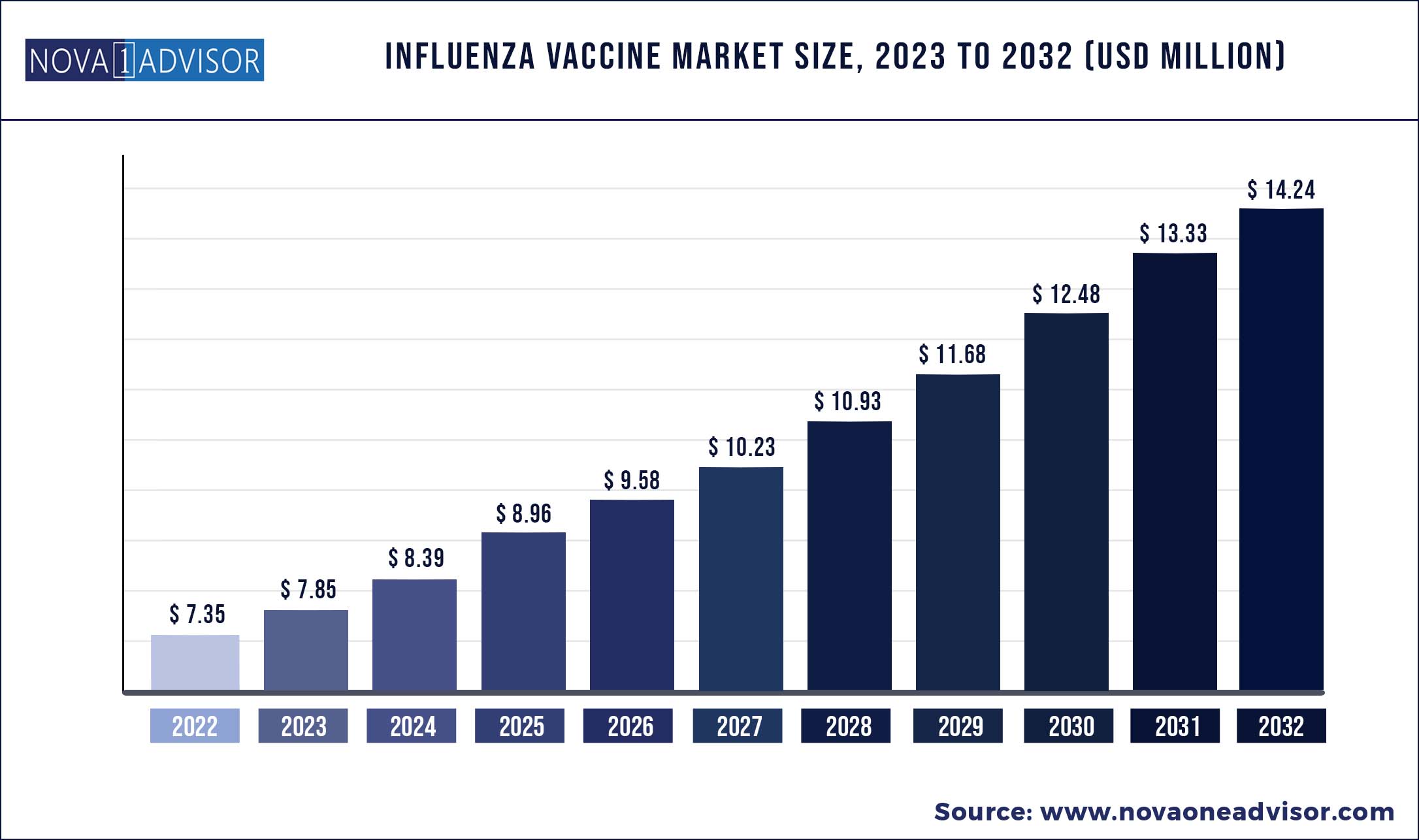
The global Influenza Vaccine market size was exhibited at USD 7.35 billion in 2022 and is projected to hit around USD 14.24 billion by 2032, growing at a CAGR of 6.84% during the forecast period 2023 to 2032.
Key Pointers:
- North America led the overall market at a revenue share of 53.39% in 2022
- The inactivated vaccine segment held the largest share of 91.89% in 2022
- The quadrivalent segment dominated the market and is projected to grow at the fastest rate of 7.12% during the projected period.
- The adult segment held a significant revenue share of more than 78.9% in 2022
- The injection was the leading segment in 2022 and is likely to hold its strong market position during the forecast period.
- The hospitals & pharmacies segment captured the largest revenue share in 2022.
Influenza Vaccine Market Report Scope
|
Report Coverage
|
Details
|
|
Market Size in 2023
|
USD 7.85 Billion
|
|
Market Size by 2032
|
USD 14.24 Billion
|
|
Growth Rate from 2023 to 2032
|
CAGR of 6.84%
|
|
Base year
|
2022
|
|
Forecast period
|
2023 to 2032
|
|
Segments covered
|
Vaccine type, Indication, Age group, Distribution channel, Route of administration
|
|
Regional scope
|
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa
|
|
Key companies profiled
|
GSK plc; AstraZeneca; SINOVAC, Pfizer Inc; Vaxess Technologies Inc; CSL Limited; EMERGENT; Merck & Co., Inc.; Viatris Inc.; OSIVAX; EMERGEX VACCINES
|
The rise in the incidences of seasonal influenza and increasing recommendations for vaccination against the disease is anticipated to increase the sales of vaccines. According to the CDC’s estimates of December 2022, there have been at least 15 million new cases of influenza with around 9,300 deaths and over 150,000 hospitalizations. In addition, the surge in surveillance and government support regarding influenza vaccination at country and global levels for monitoring the distribution, supply, and administration of the vaccine is one of the key drivers of growth in the market.
The increasing government initiatives to support influenza vaccination is one of the major factors propelling the global market growth. In association with other government bodies, the WHO steadily monitors the demand and conducts vaccination programs across the globe to manage the unmet need for vaccination. For instance, in July 2022, the government of Manitoba, Canada announced an expansion of eligibility for the high-dose influenza vaccine to include adults aged 65 years and above for enhanced protection from a life-threatening infection.
Moreover, there are many licensed seasonal influenza vaccines put forward by CDC, WHO, and other regulatory bodies to combat the infection in the current market scenario. In addition, governments around the world recommend people get immunized to attain maximum protection against diseases like the flu. Moreover, in September 2022, the Advisory Committee on Immunization Practices (ACIP) recommended the influenza vaccine to all people aged above 6 months, especially people with acute COVID-19 disease.
In addition, the increase in R&D investments by leading vaccine manufacturers and the presence of robust investigational pipeline products are anticipated to support market growth. For instance, the key pipeline products in various stages of development include mRNA-1010, OVX836, INNA-051, DAS181, OVX836, UniFlu, AV5080, ALVR106, GSK3206641A, and others.
The investigational candidates are showing promising effects with enhanced safety and protection against infection. For instance, in April 2022, Novavax, Inc. announced phase I/II results of its COVID-19 and influenza combination vaccine. The results show the vaccine is well tolerated and feasible.
Increasing investment to expand the production of influenza vaccines across the globe will boost the adoption of the vaccine. For instance, in June 2022, CSL Seqirus announced the completion of a USD 156 million expansion at its production facility in the U.S. This expansion is expected to support the formulation of its cell-based influenza vaccines in pre-filled syringes. Moreover, in March 2021, Sanofi invested USD 679 million in a new vaccine manufacturing facility in Toronto. This investment facilitated the filling capacity for Sanofi’s quadrivalent flu vaccine and boosted the supply chain in Canada, and Europe.
Influenza Vaccine Market Segmentation
| By Vaccine Type |
By Indication |
By Age Group |
By Route of Administration |
By Distribution Channel |
- Inactivated
- Live Attenuated
|
|
|
|
- Hospitals & Pharmacies
- Government & Institutional Supply
- Other
|


