Isothermal Nucleic Acid Amplification Technology/INAAT Market Size and Growth 2026 to 2035
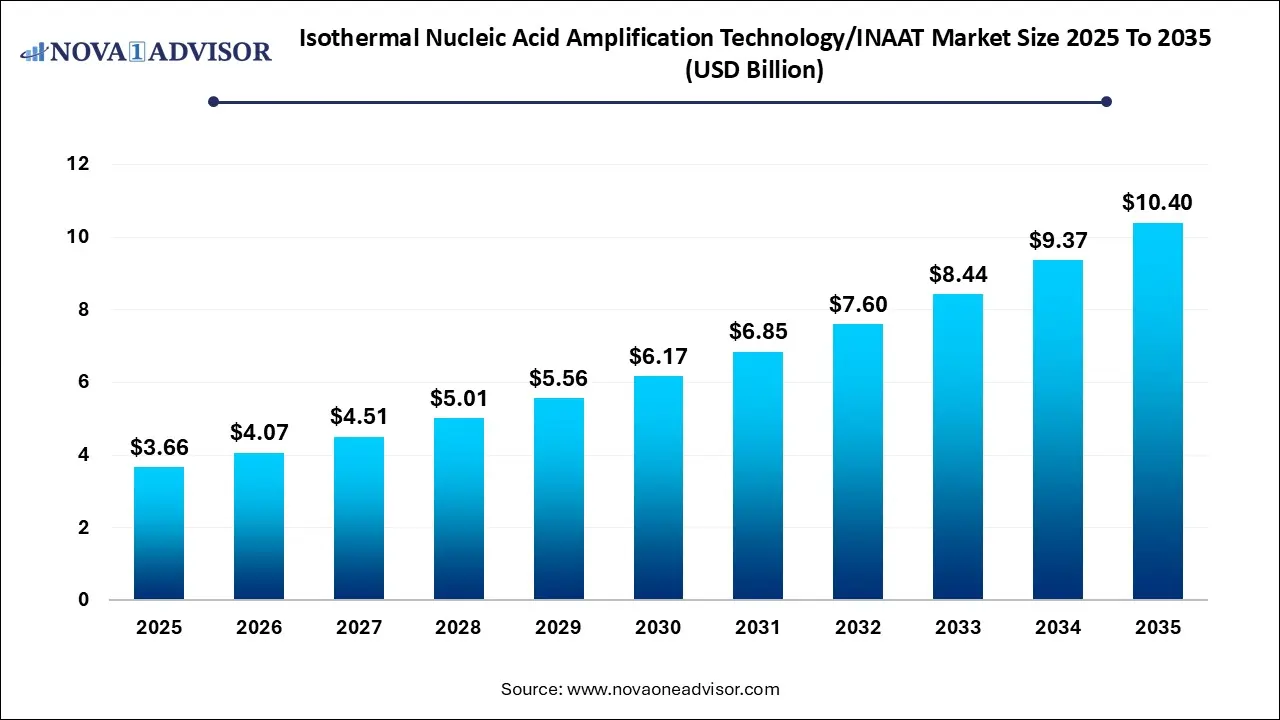
The global isothermal nucleic acid amplification technology (INAAT) market size was estimated at USD 3.66 billion in 2025 and is projected to hit around USD 10.40 billion by 2035, growing at a CAGR of 11.0% during the forecast period from 2026 to 2035.
Market Overview
The Isothermal Nucleic Acid Amplification Technology (INAAT) market has emerged as a transformative segment within molecular diagnostics, offering a simplified and rapid alternative to conventional amplification technologies such as polymerase chain reaction (PCR). Unlike PCR, which requires thermal cycling, INAAT operates at a constant temperature, enabling faster reaction times, lower energy requirements, and reduced dependence on sophisticated laboratory infrastructure.
INAAT technologies, including Loop-mediated Isothermal Amplification (LAMP), Transcription-Mediated Amplification (TMA), Strand Displacement Amplification (SDA), Helicase-Dependent Amplification (HDA), and Nucleic Acid Sequence-Based Amplification (NASBA), are widely used in diagnostic and research applications. These methods are particularly advantageous in settings where speed, portability, and ease of use are critical.
The market has witnessed significant expansion in recent years, largely driven by the increasing need for rapid diagnostic solutions. The global response to infectious disease outbreaks, including COVID-19, highlighted the importance of scalable and decentralized diagnostic technologies. INAAT-based tests were widely deployed for rapid screening, especially in regions with limited access to centralized laboratories.
For instance, LAMP-based diagnostic kits were used extensively in rural healthcare settings due to their ability to deliver results in under an hour without requiring complex instrumentation. Similarly, TMA-based assays have been widely adopted for high-throughput screening of viral infections such as HIV and hepatitis in blood banks.
Beyond infectious diseases, INAAT technologies are gaining traction in oncology, veterinary diagnostics, food safety testing, and environmental monitoring. The integration of INAAT with emerging technologies such as microfluidics, biosensors, and digital health platforms is further enhancing its applicability.
The competitive landscape includes established diagnostics companies as well as innovative startups focusing on next-generation assay development and device miniaturization. As healthcare systems increasingly prioritize accessibility, efficiency, and early detection, the INAAT market is poised for sustained growth.
Market Outlook
- Market Growth Overview: The isothermal nucleic acid amplification technology/INAAT market is expected to grow significantly between 2025 and 2034, driven by the rising prevalence of infectious diseases, the rising need for rapid point-of-care testing, and increasing applications in molecular diagnostics.
- Sustainability Trends: Sustainability trends involve energy efficiency and eco-friendly design, reduced consumable waste, and cost-efficient accessibility.
- Major Investors: Major investors in the market include Abbott Laboratories, Hologic, Inc., bioMérieux SA, Qiagen N.V., Becton, Dickinson and Company, and QuidelOrtho Corporation.
Major Trends in the Market
- Rapid expansion of point-of-care and decentralized diagnostic testing
- Increasing adoption of LAMP-based assays due to ease of use and cost efficiency
- Integration of INAAT with portable and handheld diagnostic devices
- Growing use of multiplex assays for simultaneous detection of multiple pathogens
- Rising demand for home-based diagnostic kits
- Advancements in microfluidics and lab-on-a-chip technologies
- Increasing use of INAAT in low-resource and rural healthcare settings
- Expansion of applications beyond infectious diseases into oncology and genetic testing
- Development of AI-integrated diagnostic platforms for improved accuracy
- Rising investments in pandemic preparedness and surveillance infrastructure
Isothermal Nucleic Acid Amplification Technology/INAAT Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 4.07 Billion |
| Market Size by 2035 |
USD 10.40 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 11.0% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product,By Type, By Application, By End User, By Region |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
The major companies in the INAAT market include Grifols S.A. (Spain), Hologic, Inc. (US), Abbott Laboratories, Inc. (US), Becton, Dickinson & Company (US), and Meridian Bioscience (US). |
Market Driver
Growing Demand for Rapid and Decentralized Diagnostic Solutions
The increasing demand for rapid and decentralized diagnostic solutions is a major driver of the INAAT market. Traditional diagnostic techniques such as PCR, while highly accurate, require sophisticated laboratory infrastructure, trained personnel, and longer processing times. These limitations restrict their use in emergency settings, remote areas, and resource-constrained environments.
INAAT technologies address these challenges by enabling nucleic acid amplification at a constant temperature, significantly reducing the complexity and time required for testing. Results can often be obtained within 30–60 minutes, making INAAT highly suitable for point-of-care applications. For example, during infectious disease outbreaks, rapid identification of infected individuals is crucial for containment and treatment. INAAT-based tests have been successfully used in such scenarios to provide timely results and facilitate immediate intervention.
Furthermore, the shift toward decentralized healthcare models is driving the adoption of portable diagnostic devices. INAAT platforms can be integrated into compact systems that can be used in clinics, pharmacies, and even at home. This is particularly important in regions with limited access to centralized laboratories.
The increasing prevalence of infectious diseases, coupled with the need for efficient screening and surveillance, is further fueling demand. Governments and healthcare organizations are investing in diagnostic technologies that can be deployed quickly and at scale, thereby boosting the growth of the INAAT market.
Market Restraint
Variability in Sensitivity and Lack of Standardization
Despite its advantages, INAAT technology faces challenges related to variability in sensitivity and lack of standardization. Compared to PCR, which is considered the gold standard in molecular diagnostics, INAAT methods may exhibit inconsistencies in performance depending on assay design, sample quality, and environmental conditions.
One of the primary concerns is the potential for false-positive or false-negative results. In cases where the target nucleic acid concentration is low, INAAT assays may not achieve the same level of sensitivity as PCR. This can impact diagnostic accuracy and clinical decision-making. For instance, early-stage infections with low viral loads may be difficult to detect using certain INAAT methods.
Additionally, the absence of universally accepted protocols and standards for INAAT technologies creates variability across different platforms and manufacturers. This lack of standardization can hinder widespread adoption, particularly in regulated markets where consistency and reliability are critical.
Regulatory approval processes also pose challenges, as authorities require extensive validation data to ensure the safety and efficacy of diagnostic tests. This can increase development timelines and costs, potentially slowing innovation in the market.
Market Opportunity
Expansion in Emerging Markets and Resource-Limited Settings
The expansion of INAAT technologies in emerging markets and resource-limited settings presents a significant growth opportunity. Many regions, particularly in Asia Pacific, Africa, and Latin America, face challenges related to limited healthcare infrastructure and access to advanced diagnostic facilities.
INAAT technologies are well-suited for these environments due to their minimal equipment requirements, rapid turnaround times, and ease of use. Portable diagnostic devices can be deployed in rural clinics, mobile health units, and field settings, enabling on-site testing and immediate treatment decisions.
For example, INAAT-based assays have been used in sub-Saharan Africa for the detection of diseases such as malaria and tuberculosis, significantly improving access to diagnostic services. Similarly, in Southeast Asia, these technologies are being used for early detection of dengue and other infectious diseases.
The increasing focus on global health initiatives and disease surveillance is driving investments in scalable diagnostic solutions. Governments and international organizations are prioritizing the development and deployment of cost-effective technologies that can address healthcare challenges in underserved regions.
The integration of INAAT with digital health platforms further enhances its potential, enabling real-time data collection, remote monitoring, and improved disease tracking. This capability is particularly valuable in managing outbreaks and implementing targeted interventions.
Segmental Insights
By Product
How did the assays, kits, and reagents segment dominate the Isothermal Nucleic Acid Amplification Technology/INAAT market?
The assays, kits, and reagents segment is driven by a recurring revenue model where every diagnostic test requires a fresh set of consumables. The global push for point-of-care (POC) testing, which relies on simple, portable kits for the rapid detection of HIV, Hepatitis, and respiratory infections. Innovation in enzyme stability and multiplexing by industry leaders ensures these products remain essential for high-accuracy molecular diagnostics. The shift toward decentralized testing across hospitals and field settings cements consumables as the primary value driver in the isothermal amplification market.
How did the systems segment expect to hold the fastest-growing Isothermal Nucleic Acid Amplification Technology/INAAT market in the coming years?
The systems segment is driven by the development of compact, battery-powered platforms that enable point-of-care (POC) testing in remote and resource-constrained environments. These advanced systems integrate microfluidics and automation, significantly increasing the speed and accuracy of molecular biomarker detection for infectious diseases like HIV and Hepatitis. The technological breakthroughs are transforming diagnostic infrastructure, making high-precision molecular testing more accessible and efficient worldwide.
By Type
How did the transcription-mediated amplification (TMA) segment account for the largest share in the Isothermal Nucleic Acid Amplification Technology/INAAT market?
The transcription-mediated amplification (TMA) segment is driven by the exceptional sensitivity, capable of generating a 10 billion-fold increase in target sequences within just 15 to 30 minutes. Its unique ability to amplify RNA targets directly without a separate reverse transcription step makes it the gold standard for detecting viruses like HIV and HCV with high efficiency. This technology is heavily utilized by industry leaders like Hologic for large-scale blood screening, ensuring a consistent and high-volume market demand.
How did the loop-mediated isothermal amplification (LAMP) segment expect to hold the fastest-growing Isothermal Nucleic Acid Amplification Technology/INAAT market in the coming years?
The loop-mediated isothermal amplification (lamp)loop-mediated isothermal amplification (LAMP) segment is driven by its exceptional speed and sensitivity, utilizing multiple primers to achieve target amplification in under 45 minutes. Its ability to operate at a constant temperature eliminates the need for expensive thermal cyclers, making it the premier choice for field-based and resource-limited diagnostic settings. Continuous innovation, including integration with microfluidics and CRISPR-based systems, is further expanding its versatility for detecting complex infectious diseases.
By Application
How did the infectious disease diagnosis segment account for the largest share in the Isothermal Nucleic Acid Amplification Technology/INAAT market?
The infectious disease diagnosis segment is driven by the urgent global need for rapid, point-of-care testing that operates at constant temperatures, bypassing the complexities of traditional PCR. Its versatility in detecting diverse DNA and RNA targets. Increasing healthcare investments and the demand for cost-effective tools in emerging markets for malaria and tuberculosis ensure this application remains the primary engine of market growth.
How did the blood screening segment expect to hold the fastest-growing Isothermal Nucleic Acid Amplification Technology/INAAT market in the coming years?
The blood screening segment is driven by technological advancements in multiplexed devices, which allow for the simultaneous detection of multiple pathogens with extreme efficiency. Adoption is further accelerated by the shift toward portable point-of-care diagnostics and increasingly stringent global safety regulations for blood supply management. These factors position molecular-based isothermal screening as a critical, high-growth frontier for ensuring the safety and reliability of global donor banks.
By End User
How did the hospitals segment account for the largest share in the Isothermal Nucleic Acid Amplification Technology/INAAT market?
The hospitals segment is driven by the rapid diagnosis of critical respiratory and infectious diseases. This leadership is further strengthened by a strategic shift toward in-house molecular diagnostics, allowing hospitals to manage personalized medicine and cancer screening internally. The integration of these high-speed isothermal technologies into routine clinical workflows ensures hospitals remain the primary revenue-generating hub for the market.
How did the reference laboratories segment expect to hold the fastest-growing Isothermal Nucleic Acid Amplification Technology/INAAT market in the coming years?
The reference laboratories segment is driven by its role as a centralized hub for high-volume, specialized diagnostics, with an expected revenue. These labs are aggressively adopting automated technologies, such as transcription-mediated amplification (TMA) and LAMP, to achieve high-sensitivity detection without the need for complex thermal cyclers. The ability of reference labs to provide efficient, multiplexed colorimetric detection ensures they remain the most critical infrastructure for modern infectious disease surveillance.
Key Players in the Isothermal Nucleic Acid Amplification Technology/INAAT Market
Grifols contributes to the INAAT market by providing high-throughput blood screening solutions, primarily through their Procleix NAT assays and automated Panther systems, which utilize Transcription-Mediated Amplification (TMA) to detect infectious diseases like HIV and HCV in donated blood.
Hologic is a leading player in the INAAT market with a strong market share, offering extensive automated molecular diagnostics and assays based on TMA technology, primarily utilized through its Panther Fusion platform.
- Abbott Laboratories, Inc.
Abbott Laboratories contributes significantly through its rapid point-of-care INAAT solutions, most notably with the ID NOW platform, which provides highly efficient molecular diagnostic results for COVID-19, influenza, and respiratory viruses in approximately 15 minutes.
Recent Developments
- March 2026 – A leading diagnostics firm launched a next-generation LAMP-based portable testing device for respiratory infections.
- January 2026 – A biotechnology company introduced a high-throughput TMA assay for HIV detection in blood screening applications.
- November 2025 – A global diagnostics player expanded its INAAT reagent manufacturing facility to meet increasing demand.
- September 2025 – A collaboration between a research institute and a diagnostics company led to the development of a multiplex INAAT platform.
- June 2025 – A major company received regulatory approval for a new INAAT-based influenza diagnostic test.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the isothermal nucleic acid amplification technology (INAAT) market.
By Product
- Assay, Kits and Reagents
- Systems
By Type
- TMA
- LAMP
- SDA
- HDA
- NASBA
- Other
By Application
- Infectious Disease Diagnosis
- Hepatitis
- CT/NG
- HIV
- Influenza
- Other
- Blood Screening
- Other
By End User
- Hospital
- Reference Laboratories
- Academic and Research Institutes
- Other end user
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)


