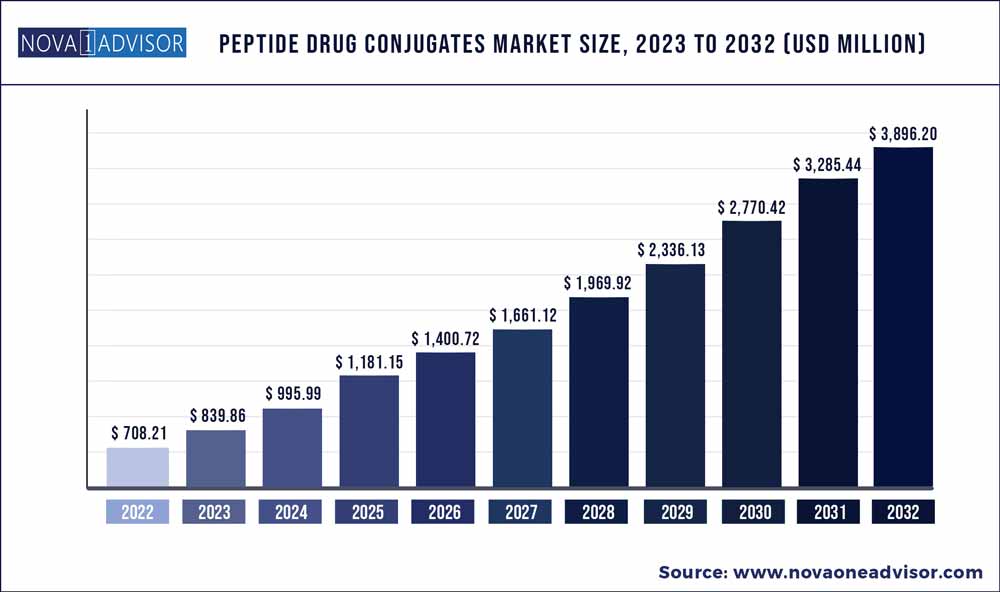
The global peptide drug conjugates market size was exhibited at USD 708.21 million in 2022 and is projected to hit around USD 3,282.41 million by 2032, growing at a CAGR of 18.59% during the forecast period 2023 to 2032.
Key Pointers:
- The Lutathera product segment dominated thepeptide drug conjugates market with a revenue share of 81.19% in 2022.
- Therapeutic segment dominated the peptide drug conjugatesmarket with the revenue share of 83.9% in 2022
- North America dominated the overall peptide drug conjugates (PDCs) market in terms of revenue share of 46.62% in 2022.
- Asia Pacific is expected to witness a growth rate of 27.6% during the forecast period.
Peptide Drug Conjugates Market Report Scope
|
Report Coverage
|
Details
|
|
Market Size in 2023
|
USD 839.86 million
|
|
Market Size by 2032
|
USD 3,896.2 million
|
|
Growth Rate from 2023 to 2032
|
CAGR of 18.59%
|
|
Base year
|
2022
|
|
Forecast period
|
2023 to 2032
|
|
Segments covered
|
Product, Type
|
|
Regional scope
|
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa
|
|
Key companies profiled
|
Novartis AG; Bicycle Therapeutics; AstraZeneca; Cybrexa Therapeutics; Oncopeptides AB; Angiochem Inc.; Innovasium Soricimed Biopharma; Theratechnologies
|
The robust clinical trial pipeline with peptide drug conjugates (PDCs) coupled with increase in R&D investments for developing PDCs and rising disease burden across the globe are the factors expected to contribute to the market growth. For instance, in July 2021, Takeda Pharmaceutical Company Limited in collaboration with Peptide Dream, announced to invest USD 3.5 billion to develop peptide drugs for neuromuscular disease.
Pharmaceutical companies are developing PDCs as targeted therapeutic drugs for the metabolic diseases, Covid-19 viral diseases, and cancer among others. Currently, the market has presence of two FDA approved drugs Pepaxto (Melflufen) and Lutathera (Lu 177 dotatate) for cancer treatment. Therefore, rising prevalence of such diseases is driving the peptide drug conjugates market. According to National Cancer Institute, in 2020, more than 1.8 million new cancer cases and more than 600,000 deaths occurred in the U.S. due to the cancer.
The strong product pipeline and increasing financial investments in research and development is offering lucrative opportunity for the peptide drug conjugates market growth. For instance, Angio Chem, Inc.’s product candidate- ANG1005 indicated for brain tumor is undergoing phase 3 clinical studies, Bicycle Therapeutics pipeline products- BT5528&BT1718 indicated for non-small cell carcinoma, are under phase 2 clinical trials. Hence, the expected new PDC approval for treatment of multiple types of cancer will boost the market growth in near future.
According to the national cancer institute, there are more than 250 FDA approved chemotherapeutic drugs used in the treatment of malignant tumors. However, the major drawback associated with such small molecules is uncontrolled toxicity resulting into severe side effects. Moreover, increasing drug resistance of tumor cells to such agents has led to demand for the better treatment approach like targeted PDCs and thereby, driving the peptide drug conjugates space. However, low bioactivity, poor stability, long research and development time, and slow clinical development process as therapeutic agents of PDC is expected to restrain the segment, leading to increase in overall time and cost incurred for the development.
Peptide Drug Conjugates Market Segmentation
| By Product |
By Type |
- Lutetium
- Melflufen
- ANG1005
- BT1718
- CBX-12
- Other Pipeline Products
|
|


