Pharmacovigilance Market Size, Share and Trends 2026 to 2035
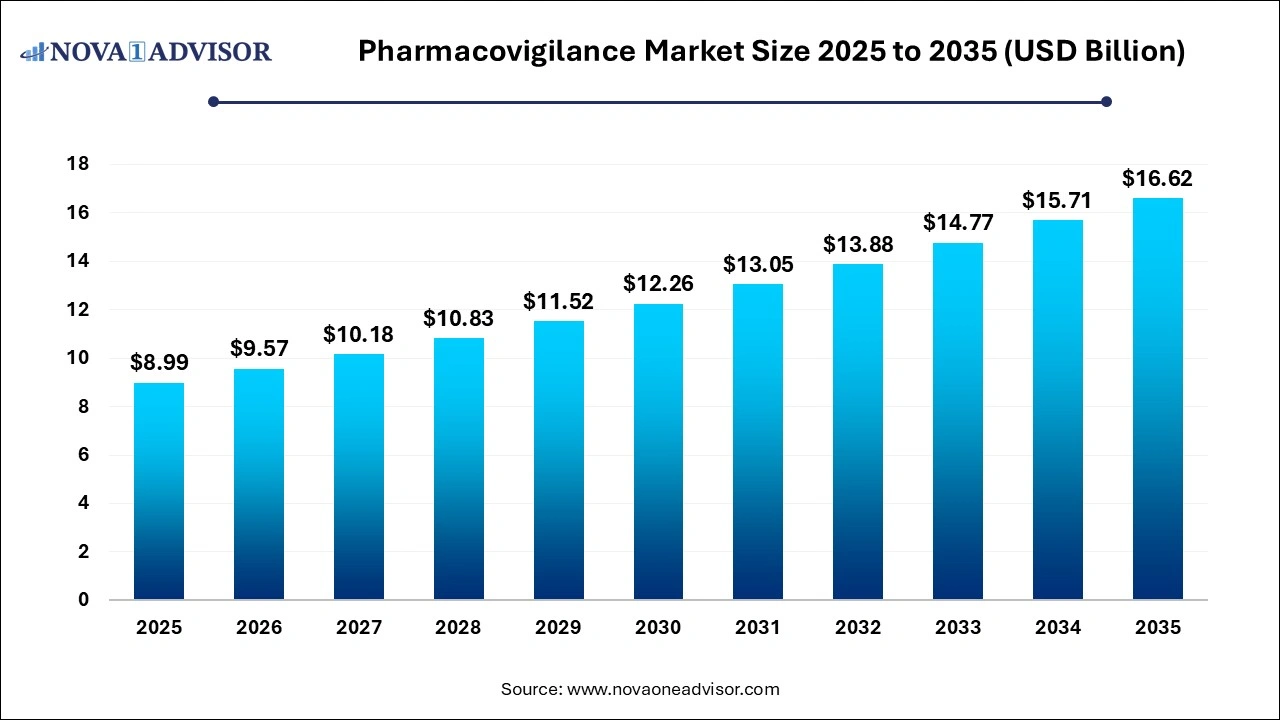
The pharmacovigilance market size was exhibited at USD 8.99 billion in 2025 and is projected to hit around USD 16.62 billion by 2035, growing at a CAGR of 6.34% during the forecast period 2026 to 2035. The pharmacovigilance market expansion is driven by adoption of digital technologies, rising incidences of adverse drug reactions and increased drug consumption. Globally expanding clinical trials is driving the need for robust pharmacovigilance systems.
Pharmacovigilance Market Key Takeaways:
- Phase IV (post-marketing) segment dominated the overall pharmacovigilance market in the product life cycle segment with over 75.9% revenue share in 2025.
- The contract outsourcing segment held the largest market share of 60.4% in 2025.
- The spontaneous reporting segment dominated the market in 2025 with a share of 30.2%.
- The oncology segment dominated the pharmacovigilance market, with a share of 26.9% in 2025.
- The signal detection segment held the largest revenue share in 2025.
- The case data management segment is anticipated to grow at a lucrative CAGR in the coming years.
- The pharmaceuticals segment dominated the market with a revenue share of over 46% in 2025.
- The biotechnology companies segment is expected to grow at the fastest CAGR during the forecast period.
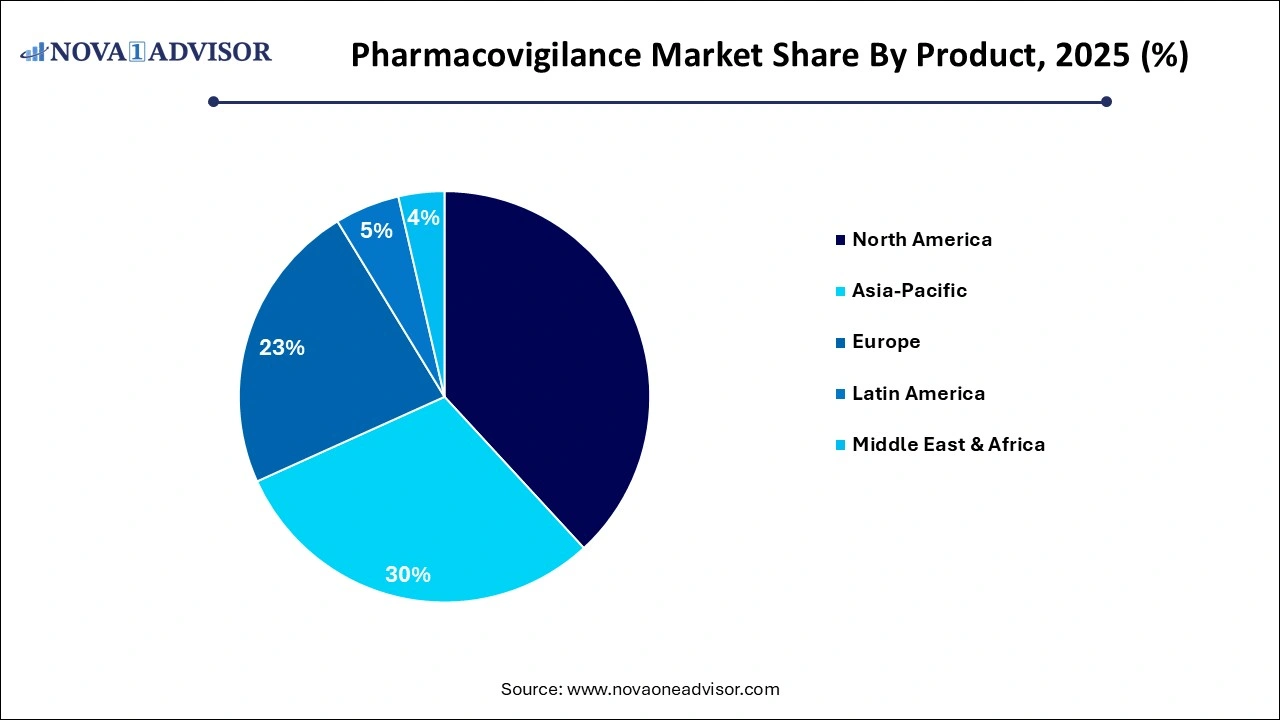
- North America dominated the pharmacovigilance market in 2025 and held the largest market share of over 35.16%.
Pharmacovigilance Market Overview
The global pharmacovigilance market has gained significant prominence in recent years as regulatory bodies, healthcare providers, and pharmaceutical companies emphasize patient safety and drug efficacy. Pharmacovigilance, defined as the science and activities related to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problems, has become an integral part of the drug development and post-marketing process.
The evolution of the market is driven by the increasing complexity of therapeutic modalities, a growing number of drug approvals, and the need for rigorous monitoring of adverse drug reactions (ADRs). As drug pipelines expand and treatments become more personalized, ensuring comprehensive safety surveillance across the product life cycle—from pre-clinical trials to post-marketing surveillance is critical. Furthermore, the emergence of digital health ecosystems, electronic health records (EHRs), and artificial intelligence has transformed pharmacovigilance into a more predictive and proactive discipline.
Pharmaceutical and biotechnology companies are under pressure to comply with stringent safety regulations imposed by entities like the U.S. FDA, EMA, and ICH. These agencies have set high expectations for signal detection, case management, and risk evaluation systems. As a result, there is a notable shift toward outsourcing pharmacovigilance services to specialized contract research organizations (CROs) and service providers, particularly in cost-sensitive or resource-limited settings.
Additionally, the COVID-19 pandemic exposed the need for agile pharmacovigilance frameworks, as vaccines and therapeutics were developed and approved at record speed. This unprecedented pace emphasized the importance of real-time safety monitoring and post-marketing vigilance—laying the groundwork for a market transformation that is expected to continue throughout the next decade.
Major Trends in the Pharmacovigilance Market
-
Integration of AI and Machine Learning in Signal Detection
Companies are increasingly deploying AI algorithms for faster and more accurate identification of potential safety signals from vast data sources.
-
Outsourcing of Pharmacovigilance Services
Cost pressures and the need for specialized expertise are driving a global outsourcing trend, especially to markets in Asia Pacific and Eastern Europe.
-
Adoption of Electronic Health Records (EHR) and Real-world Data (RWD)
Real-world evidence from EHRs and patient registries is being used to complement clinical trial data in post-marketing safety assessments.
-
Expansion of Pharmacovigilance Beyond Pharmaceuticals
Medical device manufacturers and biologics developers are investing in dedicated pharmacovigilance teams and infrastructure.
-
Harmonization of Global Regulatory Standards
Initiatives by international regulatory bodies are promoting standardized pharmacovigilance reporting across multiple geographies.
-
Rise of Patient-centered and Mobile-enabled Reporting Tools
Mobile apps and patient portals are empowering consumers to report adverse drug reactions directly, increasing data quality and volume.
-
Proliferation of Risk Management Systems
Companies are adopting integrated risk evaluation and mitigation strategies (REMS) to monitor and address risks proactively.
Pharmacovigilance integrated with artificial intelligence is enhancing the accuracy and efficiency of drug safety processes. Machine learning algorithms can be trained on large volumes of adverse event reports data for identification of patterns and predicting potential risks. With the help of natural language processing, data can be extracted from different sources such as adverse event reports, patient narratives, medical literature and posts on social media for detection of safety signals more efficiently and quickly, further reducing manual effort. AI algorithms can be applied for building predictive models for identifying patient susceptible to adverse drug reactions, leading to anticipatory actions and personalized approaches.
Report Scope of Pharmacovigilance Market
| Report Coverage |
Details |
| Market Size in 2026 |
USD 9.57 Billion |
| Market Size by 2035 |
USD 16.62 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 6.2% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Service Provider, By Product Life Cycle, By Type, By Process Flow, By Therapeutic Area, By End use |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional scope |
North America, Europe, Asia Pacific, Latin America, MEA |
| Key Companies Profiled |
Accenture; IQVIA; Cognizant; Clinquest Group B.V. (Linical Americas); IBM; Laboratory Corporation of America Holdings; ArisGlobal; Capgemini; ITClinical; ICON plc.; TAKE Solutions Ltd.; PAREXEL International Corporation.; Wipro; United BioSource LLC; BioClinica Inc (Clario).; ClinChoice (formerly FMD K&L) |
Pharmacovigilance Market Dynamics
Driver: Increasing Drug Approvals and Market Penetration of Novel Therapies
A pivotal driver fueling the growth of the pharmacovigilance market is the surge in drug approvals and the corresponding expansion of therapeutic categories. In 2023 alone, the U.S. FDA approved over 50 novel therapeutics, ranging from small molecules to gene therapies. These innovations, while promising, bring uncertainties related to long-term safety profiles, making robust pharmacovigilance critical.
With accelerated approval pathways like the FDA’s Fast Track and EMA’s PRIME schemes, many drugs are entering the market with limited long-term data. This scenario heightens the importance of phase IV (post-marketing) surveillance. For instance, the approval of oncology biosimilars and immunotherapies has necessitated more sophisticated signal detection and adverse event reporting systems. As treatment complexity rises—particularly in immunology, neurology, and rare diseases—the demand for real-time pharmacovigilance solutions is expected to soar.
Restraint: High Operational Costs and Resource Burden
Despite its essential role, pharmacovigilance operations involve significant costs and infrastructure investments. Maintaining an in-house pharmacovigilance team requires skilled personnel, sophisticated software platforms, and constant regulatory updates. For mid-sized and emerging pharma companies, these costs can be overwhelming, leading to uneven implementation of pharmacovigilance practices.
Moreover, pharmacovigilance processes particularly case data management and signal detection are labor-intensive and time-consuming. Processing adverse event reports, complying with global safety reporting timelines, and responding to regulatory audits require stringent operational discipline. In countries with limited digital infrastructure or workforce shortages, meeting compliance standards can hinder new market entry or delay clinical development.
Opportunity: Leveraging Big Data and EHR Mining for Predictive Safety
The increasing digitization of health records and the availability of vast datasets present an immense opportunity for predictive pharmacovigilance. By leveraging EHR mining, real-world data, and patient registries, pharmaceutical companies can detect safety signals earlier and with greater accuracy. This approach not only enhances drug safety but also supports more informed regulatory and clinical decisions.
For instance, EHR mining tools can help identify off-label drug use patterns, correlations between comorbidities and ADRs, and long-term adverse effects that may not surface during clinical trials. Companies such as Oracle Health and ArisGlobal are investing in pharmacovigilance platforms that integrate AI-based analytics and machine learning for automated case classification and signal prioritization. This convergence of technology and pharmacovigilance is expected to become a core differentiator in the years ahead.
Pharmacovigilance Market Segment Insights
By Product Life Cycle Insights
Phase IV (post-marketing) segment dominated the overall pharmacovigilance market in the product life cycle segment with over 75.9% revenue share in 2024. Drugs on the market are exposed to broader populations, diverse demographics, and longer usage periods—making them susceptible to unforeseen side effects. Pharmaceutical companies are required to establish active safety monitoring systems, submit periodic safety update reports (PSURs), and respond to safety alerts issued by health authorities.
Phase III is the fastest-growing segment, driven by the expansion of late-stage clinical trials across global sites. As regulatory agencies push for comprehensive safety data before approval, phase III trials now include more robust pharmacovigilance components. These include real-time AE monitoring platforms, centralized safety data review boards, and patient engagement technologies to ensure accurate ADR reporting. With the globalization of clinical trials, especially in oncology and rare diseases, the scope of pharmacovigilance in phase III continues to grow.
By Service Provider Insights
The contract outsourcing segment held the largest market share of 60.4% in 2025. largely due to the scalability, expertise, and cost advantages offered by CROs and pharmacovigilance service providers. Major pharmaceutical firms are increasingly forming long-term strategic alliances with vendors like IQVIA, ICON, and PAREXEL to manage case processing, regulatory submissions, and signal detection on a global scale. Outsourcing also enables smaller companies to comply with regulatory mandates without building expensive internal infrastructure.
In-house services are growing steadily, particularly among top-tier pharma companies that prioritize direct control and integration with clinical and regulatory teams. Companies with complex drug pipelines or unique safety requirements often prefer to maintain in-house pharmacovigilance systems to streamline operations, ensure faster feedback loops, and safeguard proprietary data. Internal pharmacovigilance teams are also integral to developing company-specific risk management strategies.
By Type Insights
The spontaneous reporting segment dominated the market in 2025 with a share of 30.2%, Reflecting its widespread use as the standard method for collecting adverse event data from healthcare professionals, patients, and regulatory bodies. It plays a central role in signal detection and post-marketing surveillance, particularly for rare or severe ADRs. National databases like EudraVigilance (EU), FAERS (U.S.), and VigiBase (WHO) primarily rely on this reporting mechanism.
EHR mining is the fastest-growing type, fueled by advances in data integration and analytics. With vast amounts of patient data stored in digital systems, companies are tapping into these repositories to conduct real-world safety assessments. EHR mining enables pattern recognition and signal validation that would be difficult to detect via spontaneous reports alone. As interoperability and AI improve, this type is expected to revolutionize pharmacovigilance practices globally.
By Therapeutic Area Insights
The oncology segment dominated the pharmacovigilance market, with a share of 26.9% in 2025. As cancer drugs particularly immunotherapies and targeted therapies often present complex, unpredictable side effects. These drugs may have narrow therapeutic windows and affect immune function, necessitating close pharmacovigilance monitoring. With many oncology drugs approved via accelerated pathways, phase IV surveillance is especially critical to manage long-term risks.
Neurology is the fastest-growing area, reflecting a surge in drug development for conditions like Alzheimer’s, Parkinson’s, and epilepsy. Many neurological drugs act on the central nervous system and may have subtle or delayed ADRs. Additionally, real-world data is increasingly used to study long-term neuropsychiatric outcomes. This has created a demand for specialized pharmacovigilance teams capable of assessing cognitive and behavioral AEs.
By Process Flow Insights
The signal detection segment held the largest revenue share in 2025. As regulatory bodies demand earlier identification and mitigation of risks. Advanced software solutions and data mining techniques are being used to identify emerging patterns from multiple data sources. Integration of adverse event logging and automated scoring systems for signal prioritization is enabling faster response times and proactive interventions.
The case data management segment is anticipated to grow at a lucrative CAGR in the coming years. Accounting for the highest volume of pharmacovigilance activities. This includes case intake, validation, medical coding, narrative writing, and submission to regulatory agencies. Given the volume of adverse event reports, especially for blockbuster drugs and vaccines, automation and global case processing hubs have become standard practice.
By End Use Insights
The pharmaceuticals segment dominated the market with a revenue share of over 46.0% in 2025. As they are the primary stakeholders responsible for ensuring drug safety from development through market maturity. Regulatory compliance, liability risk, and brand reputation drive significant investments in pharmacovigilance infrastructure. Multinational pharma firms typically manage global safety databases, operate signal review committees, and invest in automation tools to manage high case volumes.
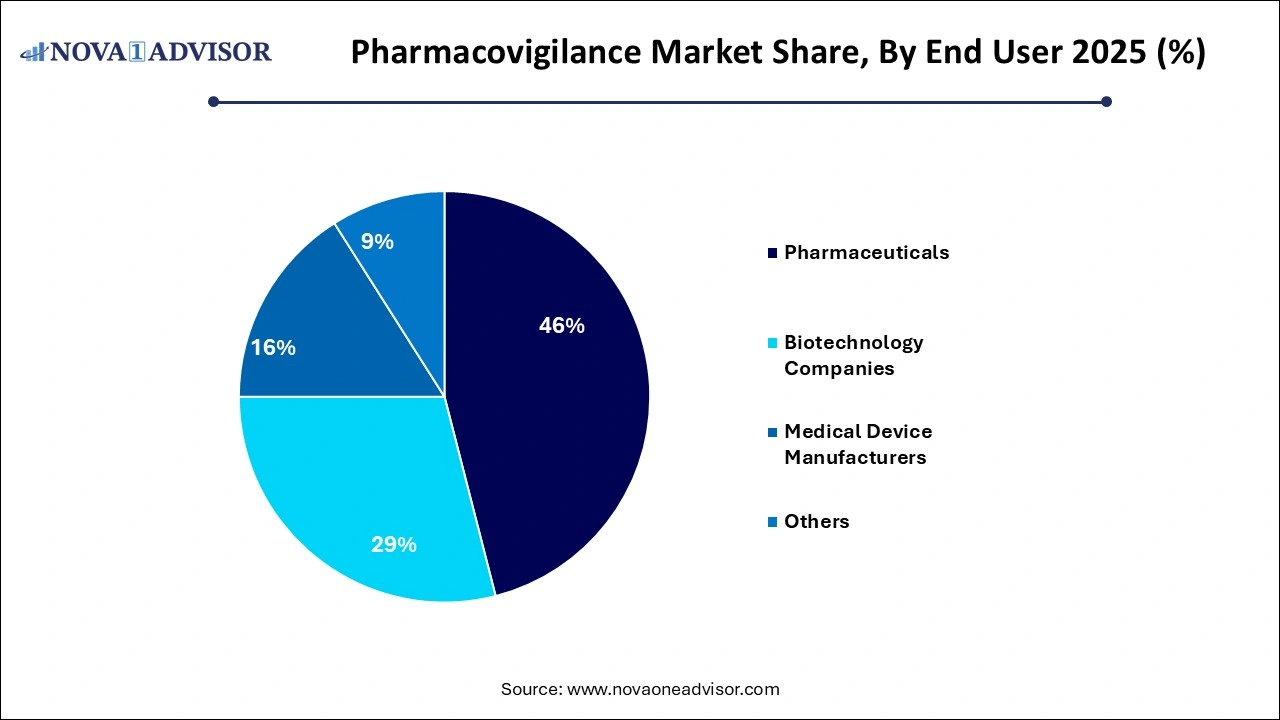
Medical device manufacturers are the fastest-growing end users, as regulatory authorities now mandate rigorous post-market surveillance for devices. With new classifications under MDR (Medical Device Regulation) in the EU and similar reforms in other regions, companies must report device-related adverse events, conduct periodic safety updates, and implement vigilance plans. The rising complexity of combination products and
digital therapeutic is also expanding the pharmacovigilance remit in this sector.
U.S. Pharmacovigilance Market Size, Share and Trends 2026 to 2035
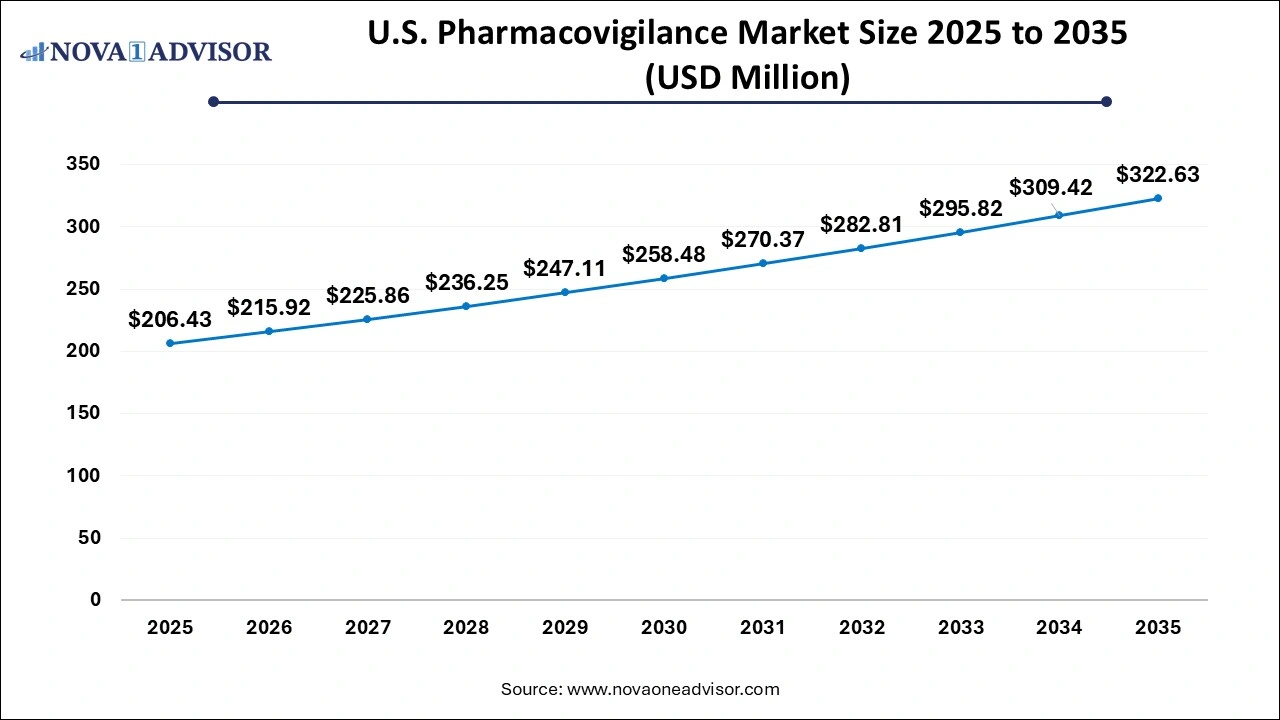
The U.S. pharmacovigilance market size was exhibited at USD 206.43 million in 2025 and is projected to hit around USD 322.63 million by 2035, growing at a CAGR of 4.57% during the forecast period 2026 to 2035.
North America, led by the United States, holds the largest share of the pharmacovigilance market, owing to a mature regulatory environment, high drug development activity, and robust digital infrastructure. The U.S. FDA has established comprehensive pharmacovigilance frameworks, including FAERS and REMS, driving extensive compliance among pharmaceutical companies. Major players like Pfizer, Merck, and Johnson & Johnson operate sophisticated safety departments that collaborate with third-party vendors for global PV operations. The region also hosts numerous pharmacovigilance conferences and academic programs, contributing to talent development and best practices.
Asia Pacific is the Fastest-Growing Region
Asia Pacific is rapidly emerging as the fastest-growing region, driven by increasing drug trials, favorable outsourcing policies, and rising investments in life sciences. Countries like India, China, and the Philippines are becoming global pharmacovigilance hubs due to a skilled workforce, English proficiency, and cost advantages. Regulatory bodies such as India's CDSCO and China’s NMPA are strengthening safety monitoring infrastructure, improving pharmacovigilance maturity. Additionally, domestic pharmaceutical companies are adopting global standards, expanding demand for end-to-end pharmacovigilance services.
China Pharmacovigilance Market Trends
China is emerging as a major contributor to the pharmacovigilance market in Asia Pacific. Increased consumption of drugs due to high prevalence of lifestyle diseases such as cardiovascular diseases, cancer, diabetes and obesity is creating the need for robust pharmacovigilance systems for drug monitoring. Rapidly growing pharmaceutical and biotechnology industries in China with development and commercialization of innovative drugs is driving the demand for pharmacovigilance services for effective post-market surveillance. Rising trend of outsourcing pharmacovigilance activities, increasing disposable incomes, increased public awareness regarding drug safety and digitalization of pharmacovigilance systems is fuelling the market expansion. The Good Pharmacovigilance Practices (GVP) guideline issued by the National Medical Products Association (NMPA) in China, focuses on enhancing safety of drugs and harmonizing them with the International Council for Harmonization (ICH) guidelines.
Pharmacovigilance Market Recent Developments
- In May 2025, Government of India introduced the AyushSuraksha Portal at Ayush Bhawan, New Delhi. The portal was launched to address issues regarding adverse drug reaction and misleading advertisements.
- In October 2024, EVERSANA, a globally leading provider of commercial services in the life sciences industry, expanded its safety case management capabilities with the selection of Oracle Argus Cloud Service as its end-to-end pharmacovigilance services platform.
- In June 2024, a Federal Register Notice issued by the U.S. Food and Drug Administration (FDA) announced the Emerging Drug Safety Technology Meeting (EDSTM) program for launching the Emerging Drug Safety Technology Program (EDSTP) which is a part of the Center for Drug Evaluation and Research’s (CDER) multifaceted approach. The new program focuses on utilizing artificial intelligence (AI) and other emerging technologies in pharmacovigilance.
- In March 2024, Klick Health introduced its first-of-a-kind social media comment moderator, Klick Comment Moderator+AI (KCM+AI) which is powered by Microsoft Azure AI and a proprietary Klick model trained on over 400,000 historical comments for enhancing the pharmacovigilance detection and reporting.
Some of the prominent players in the pharmacovigilance market include:
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2026 to 2035. For this study, Nova one advisor, Inc. has segmented the pharmacovigilance market
Product Life Cycle
- Pre-clinical
- Phase I
- Phase II
- Phase III
- Phase IV
Service Provider
- In-house
- Contract Outsourcing
Type
- Spontaneous Reporting
- Intensified ADR Reporting
- Targeted Spontaneous Reporting
- Cohort Event Monitoring
- EHR Mining
End Use
- Pharmaceuticals
- Biotechnology Companies
- Medical Device Manufacturers
- Others
Therapeutic Area
- Oncology
- Neurology
- Cardiology
- Respiratory Systems
- Others
Process Flow
-
- Case Logging
- Case Data Analysis
- Medical Reviewing & Reporting
-
- Adverse Event Logging
- Adverse Event Analysis
- Adverse Event Review & Reporting
-
- Risk Evaluation System
- Risk Mitigation System
Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)


 Medical device manufacturers are the fastest-growing end users, as regulatory authorities now mandate rigorous post-market surveillance for devices. With new classifications under MDR (Medical Device Regulation) in the EU and similar reforms in other regions, companies must report device-related adverse events, conduct periodic safety updates, and implement vigilance plans. The rising complexity of combination products and
Medical device manufacturers are the fastest-growing end users, as regulatory authorities now mandate rigorous post-market surveillance for devices. With new classifications under MDR (Medical Device Regulation) in the EU and similar reforms in other regions, companies must report device-related adverse events, conduct periodic safety updates, and implement vigilance plans. The rising complexity of combination products and