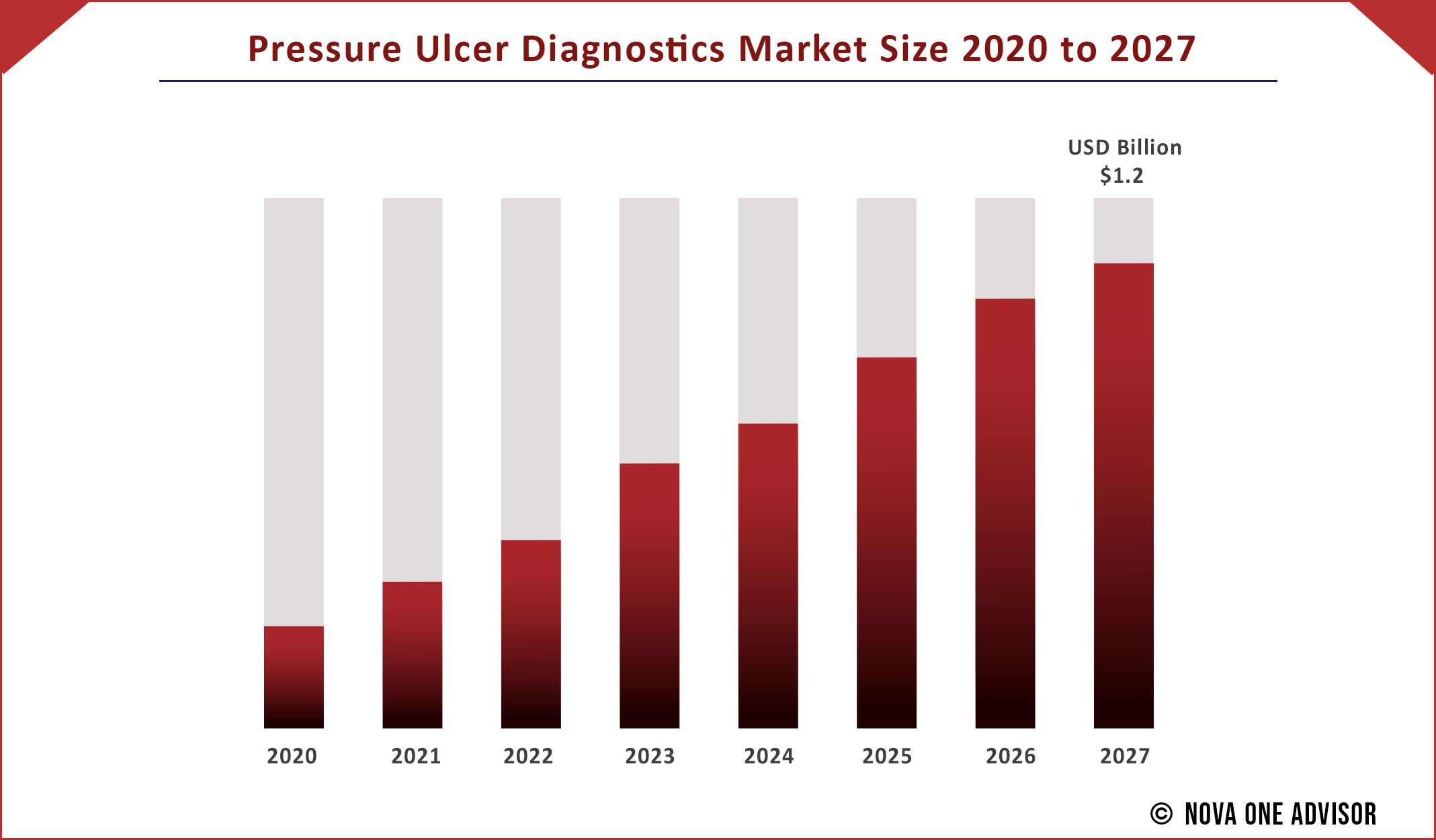
The global Pressure Ulcer Diagnostics market gathered revenue around USD 881.10 Million in 2020 and market is set to grow USD USD 1.2 Billion by the end of 2027 and is estimated to expand at a modest CAGR of 10% during the prediction period 2021 to 2027.
Pressure ulcer diagnostics are devices used in diagnosis and detection of pressure ulcer in adults and elderly patient population. Pressure ulcers are also called bedsores or pressure injuries. These are caused by pressure on the skin and lead to damage to the underlying skin or tissue. There are four stages of pressure ulcer wounds: stage 1, stage 2, stage 3, and stage 4. Various invasive and non-invasive diagnostic tools are available, which are considered standard method of diagnosis of pressure ulcer. Blood tests are most commonly used for detection of bacteria load and location.
Based on diagnostic test, the global pressure ulcer diagnostics market has been classified into imaging technique, microbiological tests, and others. The imaging technique segment accounted for major market share in 2020 due to adoption of non-invasive tools such as fluorescent imaging, photoacoustic imaging, biomarkers, magnetic resonance, computed tomography, and other imaging technologies. Technological advancements have led to the innovation of new diagnosis techniques, which is expected to drive the segment during the forecast period.
This research report purposes at stressing the most lucrative growth prospects. The aim of the research report is to provide an inclusive valuation of the Pressure Ulcer Diagnostics market and it encompasses thoughtful visions, actualities, industry-validated market findings, historic data, and prognoses by means of appropriate set of assumptions and practice. Global Pressure Ulcer Diagnostics market report aids in comprehending market structure and dynamics by recognizing and scrutinizing the market sectors and predicted the global market outlook.
COVID-19 Impact Assessment on Market Landscape
The report comprises the scrutiny of COVID-19 lock-down impact on the income of market leaders, disrupters and followers. Since lock down was instigated differently in diverse regions and nations, influence of same is also dissimilar across various industry verticals. The research report offers present short-term and long-term influence on the market to assist market participants across value chain makers to formulate the framework for short term and long-lasting tactics for recovery and by region.
Pressure Ulcer Diagnostics market report empower readers with all-inclusive market intelligence and offers a granular outline of the market they are operational in. Further this research study delivers exceptional combination of tangible perceptions and qualitative scrutiny to aid companies accomplishes sustainable growth. This report employs industry-leading research practices and tools to assemble all-inclusive market studies, intermingled with pertinent data. Additionally, this report also emphases on the competitive examination of crucial players by analyzing their product portfolio, pricing, gross margins, financial position, growth approaches, and regional occurrence.
Pressure Ulcer Diagnostics Market: Prominent Regions
In terms of region, the global pressure ulcer diagnostics market has been segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa. North America dominated the global pressure ulcer diagnostics market in 2020, followed by Europe and Asia Pacific. Increase in patients suffering from pressure ulcers, rise in expenditure of the U.S. Government on management of pressure ulcers, surge in admissions of patients in ICU, and increase in baby boomers in the U.S. propel the market in North America during the forecast period.
The pressure ulcer diagnostics market in Asia Pacific is expected to expand at a higher CAGR from 2021 to 2027. Increase in geriatric patient population at risk of developing pressure ulcers and rise in prevalence of pressure ulcers in ICU admitted patients and long-term care older patients in hospitals and nursing homes are likely to boost the growth of the pressure ulcer diagnostic market in the region over the next few years.
| Report Highlights |
Details |
| Market Size |
US$ 1.2 Billion by 2027 |
| Growth Rate |
CAGR of 10% From 2021 to 2027 |
| Base Year |
2020 |
| Historic Data |
2017 to 2020 |
| Forecast Period |
2021 to 2027 |
| Segments Covered |
Product,Wound,Modality |
| Regional Scope |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa (MEA) |
| Companies Mentioned |
IR-MED, Bruin Biometrics, LLC, Canon Medical Systems Corporation, Esaote SPA, Fonar Corporation, Bruker Corporation, FUJIFILM Healthcare Corporation, GE Healthcare, Arjo, Cyberdyne, Inc., and MolecuLight, Inc |
Competitive Rivalry
Foremost players in the market are attentive on adopting corporation strategies to enhance their market share. Some of the prominent tactics undertaken by leading market participants in order to sustain the fierce market completion include collaborations, acquisitions, substantial spending in R&D and the improvement of new-fangled products or reforms among others.
Major manufacturers & their revenues, percentage splits, market shares, growth rates and breakdowns of the product markets are determined through secondary sources and verified through the primary sources.
- Company Overview
- Company Market Share/Positioning Analysis
- Product Offerings
- Financial Performance
- Recent Initiatives
- Key Strategies Adopted by Players
- Vendor Landscape
- List of Suppliers
- List of Buyers
Some of the prominent players in the Pressure Ulcer Diagnostics Market include: IR-MED, Bruin Biometrics, LLC, Canon Medical Systems Corporation, Esaote SPA, Fonar Corporation, Bruker Corporation, FUJIFILM Healthcare Corporation, GE Healthcare, Arjo, Cyberdyne, Inc., and MolecuLight, Inc.
Unravelling the Critical Segments
This research report offers market revenue, sales volume, production assessment and prognoses by classifying it on the basis of various aspects including product type, application/end-user, and region. Further, this research study investigates market size, production, consumption and its development trends at global, regional, and country level for period 2017 to 2027 and covers subsequent region in its scope:
Global Pressure Ulcer Diagnostics Market: Segmentation
- Pressure Ulcer Diagnostics Market, by Product
- Imaging Technique
- Magnetic Resonance Imaging (MRI)
- Computed Tomography (CT) Scan
- Fluorescence Imaging
- Ultrasound Imaging
- Others
- Microbiological Tests
- Biopsy
- Blood Tests
- Others
- Others
- Pressure Ulcer Diagnostics Market, by Wound
- Stage 1
- Stage 2
- Stage 3
- Stage 4
- Pressure Ulcer Diagnostics Market, by Modality
- Hospitals
- Home Care Settings
- Nursing homes
- Others
By Geography
North America
Europe
- Germany
- France
- United Kingdom
- Rest of Europe
Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
Latin America
- Brazil
- Rest of Latin America
Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa
Highlights of the Report:
- Market Penetration: Comprehensive information on the product portfolios of the top players in the market.
- Product Development/Innovation: Detailed insights on the upcoming technologies, R&D activities, and product launches in the market
- Competitive Assessment: In-depth assessment of the market strategies, geographic and business segments of the leading players in the market
- Market Development: Comprehensive information about emerging markets. This report analyzes the market for various segments across geographies
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the market
Research Methodology
In the study, a unique research methodology is utilized to conduct extensive research on the growth of the Pressure Ulcer Diagnostics market, and reach conclusions on the future growth parameters of the market. This research methodology is a combination of primary and secondary research, which helps analysts ensure the accuracy and reliability of the conclusions.
Secondary resources referred to by analysts during the production of the Pressure Ulcer Diagnostics market study are as follows - statistics from government organizations, trade journals, white papers, and internal and external proprietary databases. Analysts have also interviewed senior managers, product portfolio managers, CEOs, VPs, marketing/product managers, and market intelligence managers, all of whom have contributed to the development of this report as a primary resource.
Comprehensive information acquired from primary and secondary resources acts as a validation from companies in the market, and makes the projections on the growth prospects of the Pressure Ulcer Diagnostics markets more accurate and reliable.
Secondary Research
It involves company databases such as Hoover's: This assists us recognize financial information, structure of the market participants and industry competitive landscape.
The secondary research sources referred in the process are as follows:
- Governmental bodies, and organizations creating economic policies
- National and international social welfare institutions
- Company websites, financial reports and SEC filings, broker and investor reports
- Related patent and regulatory databases
- Statistical databases and market reports
- Corporate Presentations, news, press release, and specification sheet of Manufacturers
Primary Research
Primary research includes face-to face interviews, online surveys, and telephonic interviews.
- Means of primary research: Email interactions, telephonic discussions and Questionnaire based research etc.
- In order to validate our research findings and analysis we conduct primary interviews of key industry participants. Insights from primary respondents help in validating the secondary research findings. It also develops Research Team’s expertise and market understanding.
Industry participants involved in this research study include:
- CEOs, VPs, market intelligence managers
- Procuring and national sales managers technical personnel, distributors and resellers
- Research analysts and key opinion leaders from various domains
Key Points Covered in Pressure Ulcer Diagnostics market Study:
- Growth of Pressure Ulcer Diagnostics in 2021
- Market Estimates and Forecasts (2017-2027)
- Brand Share and Market Share Analysis
- Key Drivers and Restraints Shaping Market Growth
- Segment-wise, Country-wise, and Region-wise Analysis
- Competition Mapping and Benchmarking
- Recommendation on Key Winning Strategies
- COVID-19 Impact on Demand for Pressure Ulcer Diagnostics and How to Navigate
- Key Product Innovations and Regulatory Climate
- Pressure Ulcer Diagnostics Consumption Analysis
- Pressure Ulcer Diagnostics Production Analysis
- Pressure Ulcer Diagnostics and Management


