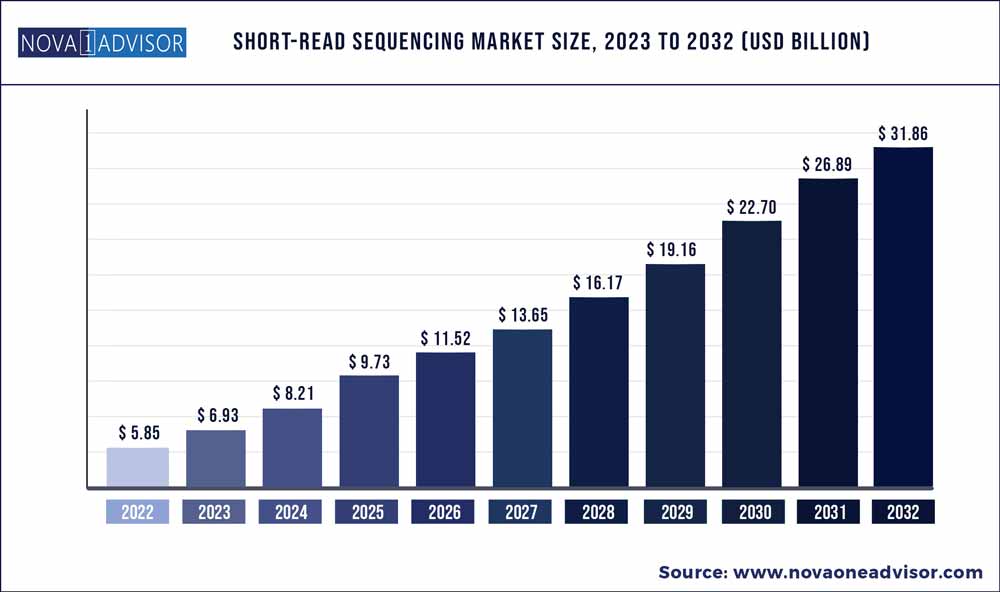
The global short-read sequencing market size was exhibited at USD 5.85 billion in 2022 and is projected to hit around USD 31.86 billion by 2032, growing at a CAGR of 18.47% during the forecast period 2023 to 2032.
Key Pointers:
- The Consumables segment held the major share of 62.65% of the short-read sequencing industry in 2022.
- The services segment is expected to become the fastest-growing segment in the short-read sequencing industry by 2032. It is expected to grow at a CAGR of 21.98% between 2023 and 2032.
- The sequencing segment accounted for the largest market share of 56.72% in the workflow segment in 2022.
- Data Analysis is projected to be the fastest-growing segment in the short-read sequencing industry and is expected to grow at a CAGR of 20.53% between 2023 and 2032.
- The academic & research institutes segment accounted for the largest market share of 48.69% in 2022
- The targeted sequencing & resequencing segment had the largest market share of 68.25% in 2022.
- North America accounted for the largest share of the short-read sequencing industry in 2022 and accounted for around 49.78% of the market size.
- Asia Pacific is estimated to grow at the fastest rate during the forecast period.
Short-read Sequencing Market Report Scope
| Report Coverage |
Details |
| Market Size in 2023 |
USD 6.93 Billion |
| Market Size by 2032 |
USD 31.86 Billion |
| Growth Rate from 2023 to 2032 |
CAGR of 18.47% |
| Base year |
2022 |
| Forecast period |
2023 to 2032 |
| Segments covered |
Product, workflow, Application, End-use |
| Regional scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key companies profiled |
Illumina, Inc.; Thermo Fisher Scientific, Inc.; Pacific Biosciences of California, Inc.; BGI; QIAGEN; Agilent Technologies; PerkinElmer, Inc.; ProPhase Labs, Inc. (Nebula Genomics); Psomagen; Azenta US, Inc. (GENEWIZ) |
Increasing demand for personalized medicine and companion diagnostics is anticipated to boost the industry growth. Short reads are generated massively in a single run and the technology is easy to use and inexpensive. These advantages are projected to further fuel growth. Moreover, the COVID-19 pandemic involved increased exposure to the new strains of the SARS-CoV-2 virus which displayed a high level of contagiousness and resistance to drugs and treatment. Early detection of the virus variant lineage was essential to treat the viral infection and improve public health prospects. Although the market was negatively affected in 2020 due to the lockdown strictures, genome sequencing methods witnessed robust demand for virus characterization and this had a positive impact on the global short-read sequencing industry in 2021. In addition, the pandemic led to increased awareness about short-read techniques, which coupled with the development of cost-effective genome sequencing approaches is expected to boost the market revenue.
The increase in demand and usage of cancer therapeutics has significantly enhanced the adoption of short-read technologies for companion diagnostics. Short-read sequencing is a rapid method for the detection of genetic abnormalities. It is useful in the diagnosis of genetic deformities. Moreover, the high throughput nature of the short-reads enables the sequencing of millions of genome fragments simultaneously in each run, which is projected to drive the adoption of short-read sequencing-based approaches. These methods are also considered to be an ideal choice for targeted therapies and precision medicine due to the competence of short-read platforms to measure different biomarkers and map genomic sequences in a single test.
Strategic initiatives undertaken by key market players are expected to boost the market in the near future. For instance, in June 2022, Element Biosciences launched its AVITI 150-Cycle Sequencing Kit, designed for short-read sequencing applications including single-cell RNA-seq, and bulk RNA-seq, among others.
The increase in the demand for drug discovery and development for chronic illnesses such as cancer boosts the demand for short-read technologies. Similarly, a rise in the R&D for genomic research drives the market. Moreover, increasing adoption of short-read techniques due to their cost-effectiveness and short run times boosts the global market. For instance, in October 2022, Illumina, Inc. launched the NovaSeq X sequencing system for genomic and clinical discoveries generating 20000+ genomes per year. The cost of the machine was reduced to USD 200.
Some of the prominent players in the Short-read Sequencing Market include:
- Illumina, Inc.
- Thermo Fisher Scientific, Inc.
- Pacific Biosciences of California, Inc.
- BGI
- QIAGEN
- Agilent Technologies
- Psomagen
- Azenta US, Inc. (GENEWIZ)
- PerkinElmer, Inc.
- ProPhase Labs, Inc. (Nebula Genomics)
Segments Covered in the Report
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2032. For this study, Nova one advisor, Inc. has segmented the global Short-read Sequencing market.
By Product
- Instruments
- Consumables
- Services
By Workflow
- Pre-Sequencing
- Sequencing
- Data Analysis
By Application
- Whole Genome Sequencing
- Whole Exome Sequencing
- Targeted Sequencing & Resequencing
- Others
By End-Use
- Academic & Research Institutes
- Hospitals & Clinics
- Pharmaceutical & Biotechnology Companies
- Others
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa (MEA)

