Spinal Implants and Surgery Devices Market Size, Growth and Trends 2026 to 2035
The global spinal implants and surgery devices market size was valued at USD 13.80 Bn in 2025 and is expected to be worth around USD 22.91 Bn by 2035 with a CAGR of 5.2% from 2026 to 2035.
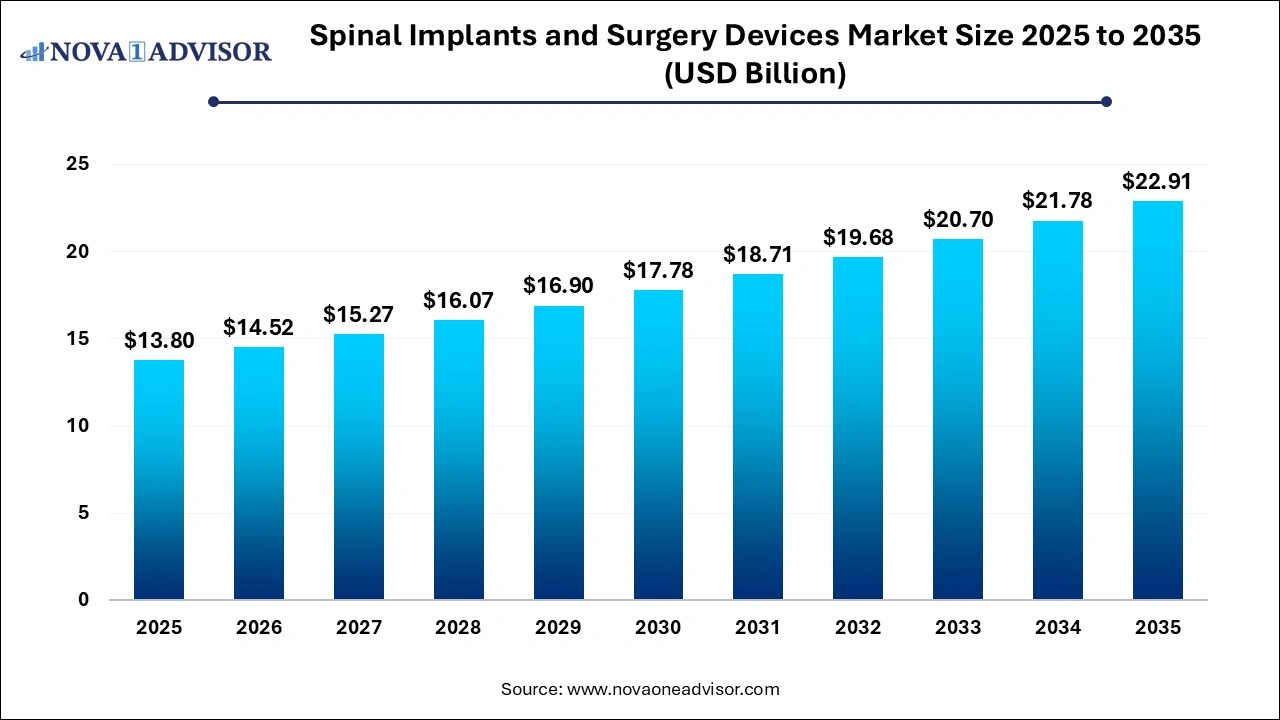
Spinal Implants and Surgery Devices Market Outlook
- Market Growth Overview: The spinal implants and surgery devices market is expected to grow significantly between 2026 and 2035, driven by the rising prevalence of spinal disorders, the growing geriatric population, and minimally invasive spine surgery.
- Sustainability Trends: Sustainability trends involve biological and regenerative materials, minimally invasive surgery, and robotic and navigation systems.
- Major Investors: Major investors in the market include Medtronic, Johnson & Johnson (DePuy Synthes), Stryker, NuVasive, Globus Medical, and Zimmer Biomet.
Spinal Implants and Surgery Devices Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 14.52 Billion |
| Market Size by 2035 |
USD 22.91 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 5.2% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product, By Technology, By Type of Surgery |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
Medtronic, DePuy Synthes, NuVasive, Inc, Stryker Corporation |
Spinal Implants and Surgery Devices Market Segment Insights
By Product Insights
How did the Thoracic & lumbar Fusion Devices Segment Dominate the Spinal Implants and Surgery Devices Market?
The thoracic and lumbar fusion devices segment is driven by the integration of NanoMetalene™ surface technologies, which significantly enhance graft incorporation and long-term structural integrity. By transitioning toward minimally invasive instrumentation, such as advanced pedicle screw systems, providers are achieving superior patient outcomes with drastically reduced recovery timelines. This synergy of proven reliability and iterative innovation ensures that fusion devices continue to lead the global spinal market as the primary solution for age-related degenerative disorders.
How did the Non-Fusion Devices Segment Expect to Hold the Fastest-Growing Spinal Implants and Surgery Devices Market in the Coming Years?
The non-fusion devices segment is driven by maintaining natural biomechanics through 3D-printed, patient-specific materials; these technologies significantly reduce the risk of adjacent-level degeneration for an increasingly active, younger demographic. The synergy of minimally invasive instrumentation and accelerated rehabilitation protocols is driving high-velocity adoption in outpatient clinical settings. These biocompatible innovations represent the high-growth frontier of spinal care, where preserving long-term mobility is as critical as achieving clinical stabilization.
By Technology Insights
How did the Spinal Fusion and Fixation Segment Account for the Largest Share in the Implants and Surgery Devices Market?
The spinal fusion and fixation segment is driven by high-volume demand from an aging demographic requiring definitive resolutions for complex lumbar and thoracic deformities. The aggressive M&A activity and portfolio diversification by industry leaders ensure that fixation remains the foundational gold standard for global spinal care delivery.
How did the Vertebral Compression Fracture (VCF) Segment Expect to Hold the Fastest-Growing Implants and Surgery Devices Market in the Coming Years?
The vertebral compression fracture (VCF) segment is driven by a global aging demographic requiring immediate pain relief and minimized hospital stays through minimally invasive techniques. By integrating next-generation self-locking implants and bioactive cements, providers are achieving superior structural restoration and long-term vertebral stability. The synergy of rapid technological innovation and favorable conditions ensures that VCF treatments remain a critical, high-growth pillar of the global spinal care ecosystem.
By Type of Surgery Insights
How did the Open Surgeries Segment Account for the Largest share in the Spinal Implants and Surgery Devices Market?
The open surgeries segment is driven by a deep-seated clinical familiarity and a long-standing track record of proven longitudinal success rates across a broad spectrum of spinal disorders. Continuous iterative enhancements in biologics and instrumentation specifically engineered for open access have ensured that these procedures remain highly effective even as the industry pivots toward smaller incisions.
How did the Minimally Invasive Surgeries Segment Expect to Hold the Fastest-Growing Spinal Implants and Surgery Devices Market in the Coming Years?
The minimally invasive surgeries segment is driven by facilitating complex spinal pathologies through micro-incisions; these technologies deliver superior patient outcomes, including drastically reduced muscle trauma and accelerated hospital discharge. This shift is critically supported by an escalating geriatric disease burden and a surge in FDA-approved instrumentation, which have combined to drive market penetration to unprecedented levels. The transition to MIS-centric frameworks provides a high-velocity pathway for healthcare systems to optimize operational efficiency while maintaining the highest quality of surgical care.
Key Players in the Spinal Implants and Surgery Devices Market
- Medtronic: As a global leader, Medtronic provides a comprehensive portfolio of spinal implants and biologics, pioneering research in robotic-assisted navigation (Mazor) and minimally invasive technologies to enhance surgical safety.
- DePuy Synthes: A Johnson & Johnson company, DePuy Synthes offers a vast, comprehensive range of products for cervical and lumbar fusion, emphasizing patient-centric solutions.
- NuVasive, Inc.: NuVasive (now part of Globus Medical) specialized in revolutionary, minimally invasive, and lateral spine procedures, such as the XLIF system, designed to reduce tissue damage. They focused on enhancing surgical efficiency through proprietary technology that integrates navigation and, more recently, robotic guidance.
- Stryker Corporation: Stryker contributes with advanced spinal hardware and specialized, high-performance implants, including their 3D-printed Tritanium technology for improved bone fusion.
Segments Covered in the Report
By Product
- Thoracic Fusion and Lumbar Fusion Devices
- Posterior Thoracic Fusion and Lumbar Fusion Devices
- Interbody Thoracic Fusion and Lumbar Fusion Devices
- By Approach
- Anterior Lumbar Interbody Fusion Devices
- Posterior Lumbar Interbody Fusion Devices
- Transforaminal Lumbar Interbody Fusion Devices
- Axial Lumbar Interbody Fusion Devices
- By Material
- Non-bone Interbody Fusion Devices
- Bone Interbody Fusion Devices
- Anterior Thoracic Fusion and Lumbar Fusion Devices
- Cervical Fusion Devices
- Anterior Cervical Fusion Devices
- Anterior Cervical Plates
- Cervical Interbody Fusion Devices
- Anterior Cervical Screw Systems
- Posterior Cervical Fusion Devices
- Posterior Cervical Plates
- Posterior Cervical Screws
- Posterior Cervical Rods
- Spine Biologics
- Demineralized Bone Matrix
- Bone Morphogenetic Proteins
- Bone Substitutes
- Machined Bones
- Cell-based Matrices
- Allograft Bone
- VCF Treatment Devices
- Balloon Kyphoplasty Devices
- Vertebroplasty Devices
- Spinal Decompression Devices
- Discectomy
- Laminoplasty, Laminectomy, and Laminotomy
- Foraminotomy and Foraminectomy
- Facetectomy
- Corpectomy
- Non-fusion Devices
- Dynamic Stabilization Devices
- Interspinous Process Spacers
- Pedicle Screw-based Dynamic Rod Devices
- Facet Replacement Products
- Artificial Discs
- Artificial Cervical Discs
- Nuclear Disc Prostheses
- Annulus Repair Devices
- Nuclear Disc Prostheses
- Spine Bone Stimulators
- Noninvasive Spine Bone Stimulators
- Pulsed Electromagnetic Field Devices
- CC and CMF Devices
- Invasive Spine Bone Stimulators
By Technology
- Spinal Fusion and Fixation
- Motion Preservation/Non-fusion
- Vertebral Compression Fracture Treatment
- Spinal Decompression
By Type of Surgery
- Open Surgeries
- Minimally Invasive Surgeries
By Region
- North America
- Europe
- Germany
- France
- UK
- Italy
- Spain
- RoE
- Asia Pacific
- Japan
- China
- India
- Australia
- South Korea
- RoAPAC
- Latin America
- Middle East & Africa


