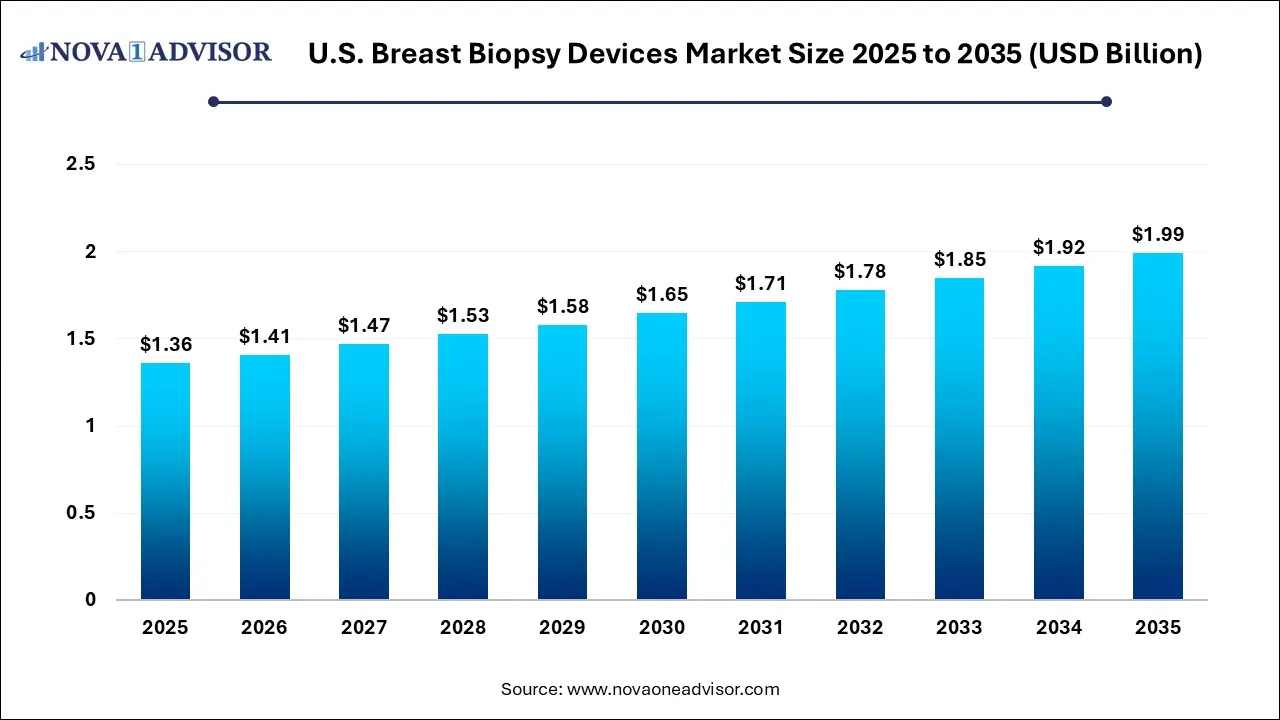
The U.S. breast biopsy devices market size was estimated at USD 1.36 billion in 2025 and is projected to increase from USD 1.41 billion in 2026 to approximately USD 1.99 billion by 2035, growing at a CAGR of 3.9% from 2026 to 2035. The U.S. breast biopsy devices market is driven by the increasing prevalence of breast cancer, integration of advanced imaging tools, such as 3D mammography and AI-enhanced guidance, which improves diagnostic accuracy, leading to faster, more accurate, and personalized procedures, and increased awareness of breast health, high screening rates and awareness, and increasing demand for high-efficiency tools.
U.S. breast biopsy devices comprise a sector providing dedicated instruments, such as vacuum-assisted needles, core needle systems, and image-guided accessories, used to remove tissue for pathologic evaluation of suspicious lesions. The modern U.S. biopsy devices include superior diagnostic accuracy, minimal invasiveness leading to faster recovery times, and reduced patient anxiety compared to traditional open surgery. These devices, particularly in outpatient settings, offer high-efficiency tissue retrieval with minimal scarring and provide crucial anatomical markers for later surgical guidance. The rising prevalence of breast cancer drives market growth, intensifies early-detection screening programs, and leads to the adoption of advanced technologies such as AI-integrated 3D tomosynthesis and MRI-guided imaging.
The significant transition from conventional core needle biopsy to the vacuum-assisted system, which allows for the removal of larger, continuous tissue samples with a single needle insertion. These devices improve diagnostic confidence, especially for microcalcifications and small lesions, and are gaining rapid market share with the superior tissue yield and reduced patient trauma.
Healthcare providers are increasingly shifting breast biopsy procedures from inpatient hospital settings to outpatient and ambulatory surgical centers. Due to the cost-containment efforts and patient preference, this trend increases demand for rapid, same-day diagnosis.
The adoption of disposable needles, probes, and site markers is accelerating to minimize cross-contamination risks and lower sterilization costs in clinical settings. This trend is sustained by the high volume of procedures, making single-use components a dominant, recurring revenue source for manufacturers.
AI and ML are revolutionizing the U.S. breast biopsy devices industry by enhancing image-guided procedures with superior precision and speed, often integrating AI-powered computer-aided detection and diagnosis tools directly into imaging platforms. These technologies improve diagnostic accuracy by roughly 15% and significantly reduce procedure times by detecting subtle abnormalities that may elude human interpretation, particularly in dense breast tissue. The integration of AI has directly contributed to reducing unnecessary benign biopsies by as much as 50% in certain clinical applications.
| Report Coverage | Details |
| Market Size in 2026 | USD 1.41 Billion |
| Market Size by 2035 | USD 1.99 Billion |
| Growth Rate From 2026 to 2035 | CAGR of 6.71% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Procedure type, Guidance Technology, End use |
| Market Analysis (Terms Used) | Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope | North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled | Hologic, Inc.; Mammotome (Danaher); BD; Argon Medical; Merit Medical Systems; GE HealthCare; Cook Medical; MOLLI Surgical (Stryker); Planmed Oy; Siemens Medical Solutions USA, Inc.; Fujifilm Healthcare; INRAD; Resitu Medical; TransMed7, LLC. |
Driver
Increasing Incidence of Breast Cancer
The rising case numbers, partially driven by a growing, aging population, necessitate higher volumes of diagnostic tissue sampling. With breast cancer being one of the most commonly diagnosed cancers among American women, more women are actively engaging in routine screening, such as mammography and MRI, which directly leads to higher detection rate of suspicious lesions. As these screening programs identify more abnormalities, the demand for timely, accurate, and minimally invasive diagnostic confirmation using devices like vacuum-assisted and image-guided systems has accelerated significantly.
High Costs of Equipment
The market growth is hindered by limited adoption among smaller healthcare facilities and rural clinics. These sophisticated systems require substantial capital investment for purchase and maintenance, creating significant financial pressure that forces many providers to continue using older, less efficient technologies. The high equipment costs often translate to elevated procedural fees, limiting patient access and placing pressure on healthcare systems already navigating limited reimbursement policies.
Disposable and Single-Use Devices
The ability to enhance patient safety by eliminating risks of cross-contamination. These devices, including core needles and vacuum-assisted probes, are essential for outpatient settings and ambulatory surgical centers (ASCs), where the shift toward minimally invasive, same-day procedures is accelerating. By providing a sterile, ready-to-use option for every procedure, these single-use components streamline workflows and reduce the need for costly sterilization processes.
Why Did the Biopsy Consumables Segment Hold a 35% Share for the U.S. Breast Biopsy Devices Market?
The biopsy consumables segment held a dominant position in the market with a share of 35% in 2025, owing to the high repeat usage per procedure and rising biopsy volumes. The healthcare providers frequently rely on complete, ready-to-use procedure kits, which include multiple consumables to improve procedural efficiency and reduce patient time in diagnostic settings. The widespread use of vacuum-assisted systems and innovation in the technology of needles.
The biopsy guidance systems segment held a 22% share of the market in 2025. Due to the increasing adoption of image-guided precision diagnostics. Increasing screening programs in the U.S. drive the need for faster, more accurate, and minimally invasive diagnostic devices. Strategic-guided systems are crucial for detecting early-stage microcalcifications.
The core needle biopsy (CNB) guns segment holds a 20% share of the market in 2025, with the high-quality tissue samples allowing for definitive diagnoses without full surgical excision, rising ability to be used in outpatient settings for better patient care, and rising number of breast cancer screenings and the need to analyze suspicious lumps in the US ensures high demand for reliable biopsy devices.
The biopsy tables segment held a 10% share of the market in 2025, due to the replacement by multifunctional imaging platforms. The rising demand for high-precision, minimally invasive, image-guided biopsy procedures necessitates specialized, dedicated, and high-performance biopsy tablets. Rising incidence of breast cancers and high adoption of advanced healthcare infrastructure.
The other consumables segment held 13% share of the market in 2025, because of the ancillary disposable products used alongside biopsy procedures. The declining as product standardization reduces variability and demand. The shift toward image-guided and minimally invasive procedures and the advancement in accessories.
Why Did the Core Needle Biopsy (CNB) Segment Lead the U.S. Breast Biopsy Devices Market?
The core needle biopsy (CNB) segment registered its dominance over the market with a share of 32% in 2025, owing to the clinical gold standard for accurate and cost-effective diagnosis. This procedure is frequently preferred in ambulatory and outpatient settings, reducing the overall cost of care and hospital stay duration.
The vacuum-assisted biopsy (VAB) segment held the 28% share of the market in 2025, due to the higher accuracy and reduced need for repeat procedures. The increasing incidence of breast cancer in the U.S. creates high demand for effective diagnostic tools, supported by widespread screening programs and strong reimbursement policies. The innovation in VAB, including disposable, faster-acting needles, has enhanced workflow efficiency in busy clinical settings.
The liquid biopsy segment held a 10% share of the market in 2025 because of the innovation and shift toward non-invasive diagnostics. The continuous monitoring of cancer recurrence and treatment efficacy in real time, significant advancement in molecular biology, next-generation sequencing and the presence of major firms drive innovation and adoption.
The surgical (Open) biopsy segment held a 18% share of the market in 2025, with the minimally invasive alternatives become preferred. Open biopsy enables the removal of the entire lesion or a significant portion, allowing pathologists to provide a definitive diagnosis, reducing the risk of false negative compared to needle biopsies.
The fine needle aspiration (FNAB) segment held a 12% share of market in 2025, with the lower diagnostic accuracy compared to CNB and VAB. The widely used for initial assessment and, combined with cytologic analysis, offers high diagnostic accuracy, particularly in distinguishing benign from malignant lesions.
Why Did the Ultrasound-Guided Segment Lead the U.S. Breast Biopsy Devices Market?
The ultrasound-guided segment registered its dominance over the market with a share of 45% in 2025, owing to the affordability, accessibility, and real-time imaging capability. A growing preference for outpatient and ambulatory care centers, which often adopt portable and cost-effective ultrasound units. The rising cancer cases require efficient and accessible screening techniques, boosting the demand for efficient diagnostic tools.
The mammography/stereotactic-guided segment held the 40% share of the market in 2025, due to a slight decline as ultrasound becomes more widely preferred. The widespread adoption of digital mammography and 3D tomosynthesis for screening has directly increased the demand for follow-up stereotactic biopsy procedures.
The MRI-Guided segment held the 15% share of the market in 2025, due to the increasing use in complex and high-risk cases. The integration of improved, MRI-safe devices, such as vacuum-assisted biopsy systems and, in some cases, advanced AI-powered tools for improved lesion detection, has increased efficiency and safety. The minimally invasive with better outcomes and superior diagnostic accuracy.
Why Did the Hospitals Segment Hold a 50% Share of the U.S. Breast Biopsy Devices Market?
The hospitals segment led the market with a share of 50% in 2025, due to the shift toward outpatient and specialized facilities. The concentration of specialized radiologists and expert care teams allows hospitals to handle high patient volumes and complex cases. The advancement in imaging technology, reimbursement, and access.
The ambulatory surgical centers (ASCs) segment held 15% share of the market in 2025, due to the cost efficiency and shorter patient turnaround times. The growing preference for image-guided and vacuum-assisted biopsies over traditional surgery is well-suited for ambulatory environments. The expansion of screening programs.
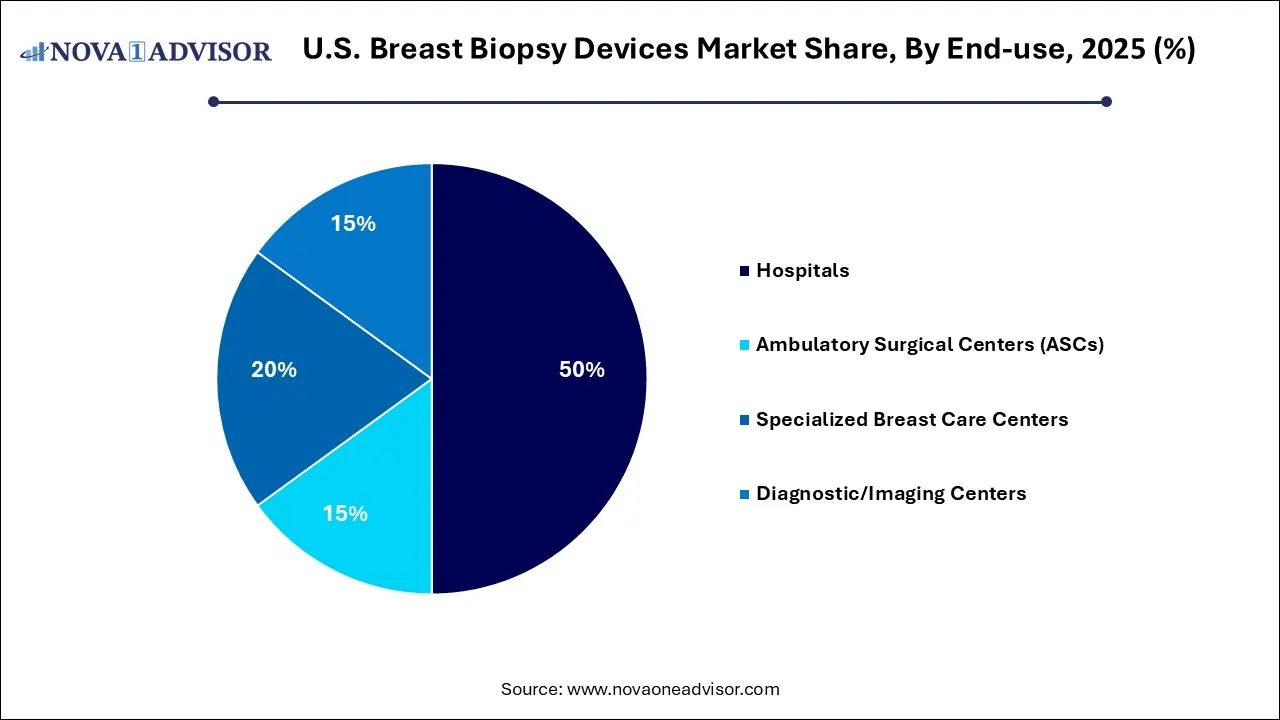
The specialized breast care centers segment held a 20% share of the market in 2025. With the increasing preference for specialized and integrated care. Increased awareness and screening, particularly among the baby boomer population, drive up patient volume in a specialized diagnostic environment. The shift toward minimally invasive, same-day procedures.
The diagnostic/imaging centers held 15% share of the market in 2025, due to the rising demand for early detection and screening services. Breast care centers provide faster scheduling, targeted care, and specialized expertise, making them a preferred choice for patients seeking rapid diagnosis. The rapid adoption of minimally invasive vacuum-assisted biopsy.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the U.S. breast biopsy devices market.
By Product Type
By Procedure Type
By Guidance Technology
By End Use