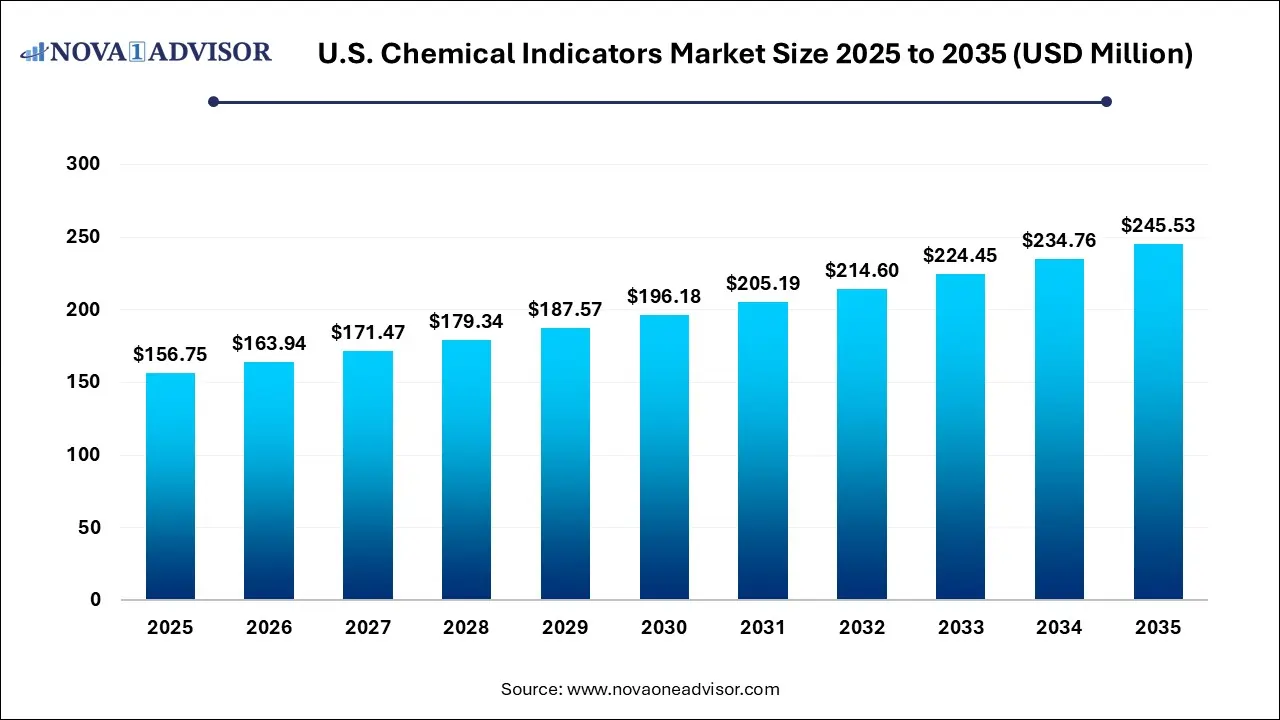
The U.S. chemical indicators market size was estimated at USD 156.75 million in 2025 and is projected to increase from USD 163.94 million in 2026 to approximately USD 245.53 million by 2035, growing at a CAGR of 4.59% from 2026 to 2035. The U.S. chemical indicators market is driven by the increasing interest in hospital-acquired infections, a surge in surgical procedures, the expansion of healthcare facilities, and increased pharmaceutical manufacturing, necessitating high volumes of indicator strips, tapes, and links, and increased use of disposable devices.
U.S. chemical indicators comprise specialized sterilization monitoring tools, such as tapes and strips, that provide immediate visual, color-changing verification that sterilization parameters have been met. These indicators offer significant benefits by distinguishing processed from unprocessed items, facilitating rapid, on-site assessment, reducing laboratory costs, and ensuring regulatory compliance with strict infection control standards in hospitals and among clinicians. The healthcare they are vital in pharmaceutical manufacturing and medical device packaging to guarantee sterility and product integrity.
There is a shift from simple, single-variable process indicators toward multi-parameter indicators that react to multiple sterilization variables, such as time, temperature, and steam. These advanced indicators provide higher accuracy and reliability, closer to that of biological indicators, helping to ensure compliance with stringent FDA and AAMI standards.
The healthcare facilities increasingly use low-temperature sterilization methods, such as hydrogen peroxide plasma and vaporized hydrogen peroxide for heat-sensitive instruments; demand for compatible indicators is growing. Its application in processing modern endoscopic and robotic surgical instruments.
Stringent FDA regulations and CDC guidelines for infection control in healthcare facilities mandate routine sterilization monitoring, accelerating the adoption of validated, high-quality chemical indicators. The increasing focus on patient safety and reducing hospital-acquired infections drives the mandatory use of internal chemical indicators for every sterilization pack.
AI and ML are significantly impacting the U.S. chemical indicators industry by enhancing sterilization validation, particularly through advanced data analytics that improve process accuracy and ensure compliance with strict infection prevention protocols. The integration of these technologies allows for the development of smart indicators capable of real-time monitoring and predictive diagnostics, reducing the reliance on manual inspection and enabling faster, more precise validation of sterilization parameters, such as temperature and time. Machine learning algorithms are being utilized to analyze high-dimensional sensors and data, spotting inconsistencies in chemical formulations and identifying potential defects in sterilization, which enhances the reliability of indicators used in healthcare and medical device manufacturing.
| Report Coverage | Details |
| Market Size in 2026 | USD 163.94 Million |
| Market Size by 2035 | USD 245.53 Million |
| Growth Rate From 2026 to 2035 | CAGR of 4.59% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Product, Method, End-use |
| Market Analysis (Terms Used) | Value (USD Million/Billion) or (Volume/Units) |
| Regional Scope | U.S |
| Key Companies Profiled | Propper Manufacturing Co.; STERIS; ASP; Solventum; Getinge; Tuttnauer; Andersen Sterilizers; Mesa Laboratories, Inc.; Hu-Friedy Mfg. Co.; True Indicating |
Driver
Rising Surgical and Outpatient Volumes
The expansion of ambulatory surgery centers and outpatient clinics means more medical devices require immediate, on-site sterilization monitoring, necessitating increased consumption of chemical indicators such as tapes and strips. The hospitals are adopting stricter infection control guidelines, mandating the routine, internal use of chemical indicators for every sterilization package to confirm compliance with regulatory standards.
Restraint
High Cost and Technical Complexity
The market growth is hindered by the healthcare and industrial sectors, where budgets for consumables are often tightly constrained. Production of advanced indicator inks requires high-precision chemical engineering, rigorous stability testing, and stringent regulatory sourcing. The need for advanced indicators to align with, or compete against, highly accurate digital tracking and biological monitoring systems often makes adoption slow in cost-sensitive settings.
Opportunity
Integration with Smart Technologies
The enabling of real-time, remote monitoring of the sterilization and packaging process. These connected indicators allow for immediate data collection and automated documentation, which improves accuracy and helps healthcare facilities and medical device manufacturers comply with increasingly stringent FDA and ISO regulatory standards. The adoption of IoT-enabled smart packaging and indicators provides essential traceability in critical applications, reducing human error and preventing contamination.
Why Did the Indicator Tapes Segment Hold a 53% Share for the U.S. Chemical Indicators Market?
The indicator tapes segment held a dominant position in the market with a share of 53% in 2025, owing to the widespread use in sterilization for its versatility and reliability, stringent regulatory requirements for sterilization, and the rising need to prevent hospital-acquired infections and surgical-site infections, which are encouraging widespread adoption. The increasing number of surgeries and diagnostic procedures in ambulatory surgical centers and hospitals, and the ease of use and versatility.
The cards segment held a 23% share of the market in 2025. Due to the use typically being limited to specific sterilization or temperature-monitoring applications, a moderate share, reflecting niche yet stable use. It is frequently used in autoclaves for internal pack monitoring of surgical instruments. They allow immediate visual confirmation of the sterilization process, helping to prevent the use of improperly processed equipment, which is crucial for reducing hospital-acquired infections.
The strips segment holds a 8% share of the market in 2025, with the competition from other more widely used methods. The presence of type 5 integrating indicators and type 6 emulating indicators, an increasing number of surgical and diagnostic procedures, and the rapid expansion of outpatient services and ambulatory surgical centers increase the volume of sterilizer loads, boosting the demand for disposable indicator strips.
The others segment held a 16% share of the market in 2025, due to the alternative products for sterilization monitoring, gaining share as new methods and innovations emerge. Increased use of chemical validation in smaller, specialized facilities and expansion of pharmaceutical and biotech applications.
Why Did the Others Segment Lead the U.S. Chemical Indicators Market?
The other segment registered its dominance over the market with a share of 83% in 2025, owing to the significant innovation, offering more versatile and newer solutions. The rise in minimally invasive surgeries and complex, heat-sensitive medical devices necessitated advanced, low-temperature sterilization methods. The growing awareness among healthcare facilities of the need for improved infection prevention, particularly to reduce hospital-acquired infections and high-volume surgical procedures.
The steam sterilization segment held the 7% share of the market in 2025, due to being the most common sterilization method in hospitals, generally preferred in standard clinical environments. The rising number of surgeries and reprocessed instruments necessitated more frequent sterilization cycles.
The ethylene oxide sterilization segment held a 7% share of the market in 2025 because of its effectiveness for a specific set of materials. Increased adoption of single-use and complex medical instruments requires specialized sterilization methods. This method is widely adopted by manufacturers and service providers for the sterilization of medical devices.
The hydrogen peroxide sterilization segment held a 2% share of the market in 2025, with the lower widespread application and adoption. Hydrogen peroxide breaks down into oxygen and water, offering a safer, non-toxic environmental alternative to traditional chemical sterilants.
Why Did the Research and Academic Institutes Segment Lead the U.S. Chemical Indicators Market?
The research and academic institutes segment registered its dominance over the market with a share of 47.0% in 2025, owing to the heavy reliance on controlled sterilization procedures for research and experimentation. The institution frequently uses autoclaves to sterilize laboratory equipment, petri dishes, and biological samples, necessitating a constant, high-volume supply of chemical indicators to verify each cycle.
The other end-use segment held the 23.9% share of the market in 2025, including industries and institutions outside of healthcare, maintaining a stable share as new applications emerge. The rising number of surgeries and diagnostic procedures, and strict guidelines regarding the prevention of healthcare-associated infections in hospitals, demand reliable sterilization verification.
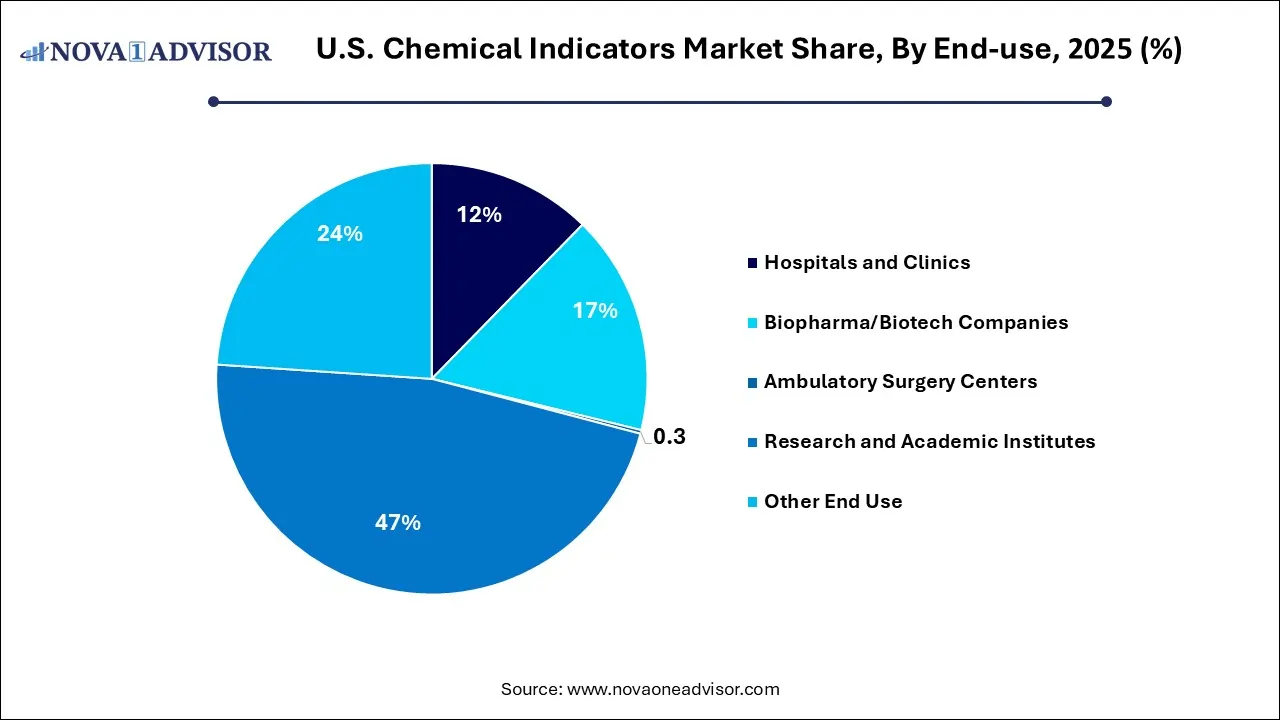
The biopharma/biotech companies segment held 16.5% share of the market in 2025, due to the industry's expansion, requiring stringent sterilization control for research and production, and gaining dominance as the industry grows. The surge in biotech startups and preference for outsourcing manufacturing to CDMOs and contract research organizations, which utilize chemical indicators for quality assurance.
The hospitals and clinics segment held a 12.3% share of the market in 2025. With its use of chemical indicators, essential for sterilization monitoring, reliance on established sterilization methods. The opening of new state-of-the-art sterilization facilities in hospitals, ambulatory surgical centers, and outpatient care centers.
The ambulatory surgery centers segment held 0.3% share of the market in 2025, because they are less dependent on chemical indicators compared to larger hospitals and clinics. Increasing infection control and compliance requirements, cost-effectiveness, and efficiency.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the U.S. chemical indicators market.
By Product
By Method
By End-use