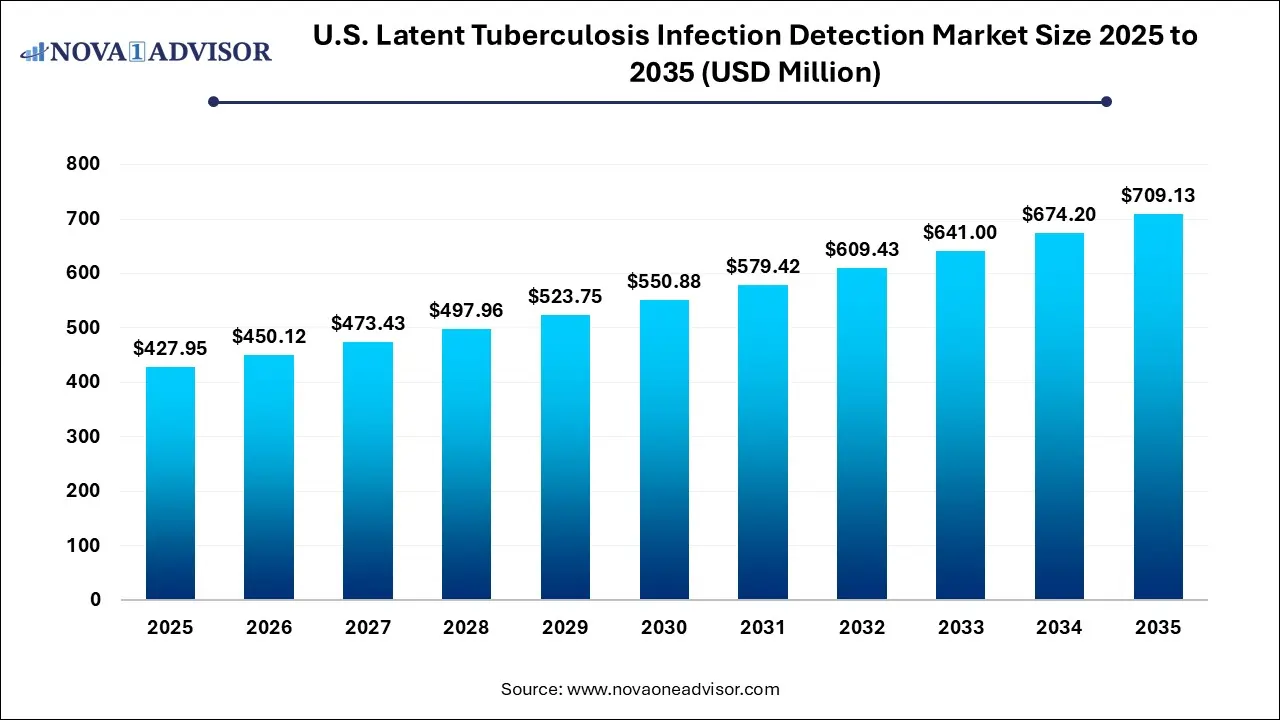
The U.S. latent tuberculosis infection detection market size was estimated at USD 427.95 million in 2025 and is projected to increase from USD 450.12 million in 2026 to approximately USD 709.13 million by 2035, growing at a CAGR of 5.18% from 2026 to 2035. The U.S. latent tuberculosis infection detection market is driven by the increasing adoption of infection gamma release assays and advanced molecular techniques, growing prevalence of immunocompromised individuals, and increasing awareness about TB, combined with a focus on controlling transmission through early intervention of latent cases.
U.S. latent tuberculosis infection detection comprises diagnostic tools, such as interferon-gamma release assays (IGRAs) and tuberculin skin tests, designed to identify dormant Mycobacterium tuberculosis in asymptomatic individuals, preventing progression to active TB. The market benefit contains improved diagnostic accuracy with high-sensitivity blood tests that are not affected by the BCG vaccine, offering reliable screenings for high-risk populations, including HIV patients and transplant recipients. The market, valued at hundreds of millions, is expanding rapidly, driven by increased government funding for screening programs and technological advancements, such as AI-integrated diagnostic platforms.
Interferon-gamma release assays (IGRAs), such as QuantiFERON-TB gold plus, are rapidly displacing the tuberculin skin test (TST) in the U.S. due to superior specificity. Unlike the TST, IGRAs are not affected by prior BCG vaccination and require only a single patient visit for a blood draw, improving compliance and providing objective, laboratory-based results.
The transition from broad population screening to targeted diagnostic protocols focused on high-risk demographics, including immunosuppressed individuals and foreign-born populations. CDC guidelines promoting targeted testing for these populations have led to higher adoption of LTBI diagnostics in primary care and occupational health settings, rather than mass population screening.
Diagnostic laboratories are adopting automated IGRA platforms to increase throughput and reduce labor-intensive manual steps. This trend is crucial for managing large-scale screenings in hospitals and reference labs, allowing for faster turnaround times (often ~24 hours) and higher efficiency.
AI and ML are significantly impacting the U.S. latent tuberculosis infection (LTBI) detection industry by enhancing screening accuracy, efficiency, and scalability to support TB elimination goals. These technologies, particularly deep learning algorithms, are used to analyze complex patient data and medical imaging to identify individuals at higher risk of progressing from latent to active TB, allowing for better-targeted preventive treatments. AI-powered tools are streamlining workflows in diagnostic laboratories, reducing the time to result, and increasing the reliability of results, especially in high-risk populations like those living with HIV. ML models are being integrated into interferon-gamma release assays (IGRAs), the dominant, high-accuracy test type in the US, to refine diagnostic precision and reduce false-negative results.
| Report Coverage | Details |
| Market Size in 2026 | USD 450.12 Million |
| Market Size by 2035 | USD 709.13 Million |
| Growth Rate From 2026 to 2035 | CAGR of 5.18% |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Test, Application, End use |
| Market Analysis (Terms Used) | Value (USD Million/Billion) or (Volume/Units) |
| Key Companies Profiled | QIAGEN; Euroimmun US; bioMérieux; Oxford Immunotec; SD Biosensor, INC.; Quest Diagnostics; LabCorp; ARUP Laboratories; Mayo Clinic Laboratories; BioReference Laboratories; Par Pharmaceutical, Inc. |
Driver
High-Risk Population Screening
The prioritizing routine testing for vulnerable groups, including individuals with HIV, close contacts of active TB cases, and foreign-born populations from high-burden regions. Because these groups have a significantly higher risk of progression from latent to active TB, public health directives and CDC funding initiatives emphasize proactive screening to prevent disease transmission and reactivation. This targeted approach increases testing volumes in hospitals, clinics, and diagnostic laboratories, fueling the adoption of advanced, highly specific diagnostic tools, such as interferon gamma release assays (IGRAs).
Limited Awareness
The market growth is hindered by the asymptomatic nature of the condition, which leads many individuals to underestimate the necessity of screening. Because people with LTBI feel healthy, they often do not proactively seek testing, resulting in low voluntary screening rates and lower demand for diagnostic services. Misconceptions and a lack of knowledge about TB, particularly among non-U.S.–born populations who represent the majority of cases, can foster stigma and create delays in testing and treatment.
Automation in Lab and Expansion in Primary Care
The increasing testing accuracy and speed while lowering labor costs. Simultaneously, the expansion of LTBI screening into primary care settings, facilitated by electronic medical record (EMR) prompts, is significantly boosting testing volumes by identifying high-risk, asymptomatic patients who might otherwise go undiagnosed. These advancements directly address the shift toward routine, targeted screening for populations at risk of reactivation.
Why Did the Interferon Gamma Released Assay (IGRA) Segment Hold a 60% Share for the U.S. Latent Tuberculosis Infection Detection Market?
The interferon gamma released assay (IGRA) segment held a dominant position in the market with a share of 60% in 2025, owing to its transitioning toward high-precision IGRA platforms to support national elimination strategies and optimize clinical workflows for high-risk populations. This shift is driven by the need for superior diagnostic accuracy and operational efficiency, effectively displacing legacy testing methods in favor of scalable, laboratory-based solutions. These advanced diagnostics are now the cornerstone of reliable tuberculosis screening within the modern healthcare landscape.
The tuberculin skin test (TST) segment held a 40% share of the market in 2025. With the cost-effectiveness and deep integration into mandatory occupational and pediatric screening protocols. While newer technologies are rising, the established infrastructure and regulatory requirements for skin testing ensure its continued dominance and high-volume utilization.
Why Did the Household Contacts with Pulmonary TB Segment Lead the U.S. Latent Tuberculosis Infection Detection Market?
The household contacts with pulmonary TB segment registered its dominance over the market with a share of 50% in 2025, owing to the high risk of transmission within households, prompting more frequent testing. The adoption of modern testing, such as interferon-gamma release assays, has made screening more reliable, encouraging widespread use.
The PLHIV segment held the 30% share of the market in 2025, due to their heightened susceptibility to TB, but not as dominant as household contacts, which have broader testing needs. Mandatory protocols and a preference for IGRA technology ensure diagnostic accuracy and consistent testing within specialized treatment centers. The expanded access to preventive therapies reinforces the necessity of these screenings as a standard of care for immunocompromised populations.
The others segment held the 20% share of the market in 2025, with its broader, less targeted scope, compared to the higher-risk categories, such as household contacts and PLHIV. The CDC and regional health departments prioritize LTBI screening to prevent active TB, driving high volumes of tests through public health laboratories and clinics, and the adoption of advanced technology in labs.
Why Did the Hospitals/Clinics Segment Lead the U.S. Latent Tuberculosis Infection Detection Market?
The hospitals/clinics segment registered its dominance over the market with a share of 40% in 2025, owing to the centralized platform for high-risk patient management and comprehensive diagnostic services. Their ability to integrate advanced testing into routine preventive care protocols ensures a high-volume, reliable workflow that aligns with national health mandates. The hospital segment remains the dominant setting for capturing and treating latent infections through efficient, scalable screening solutions.
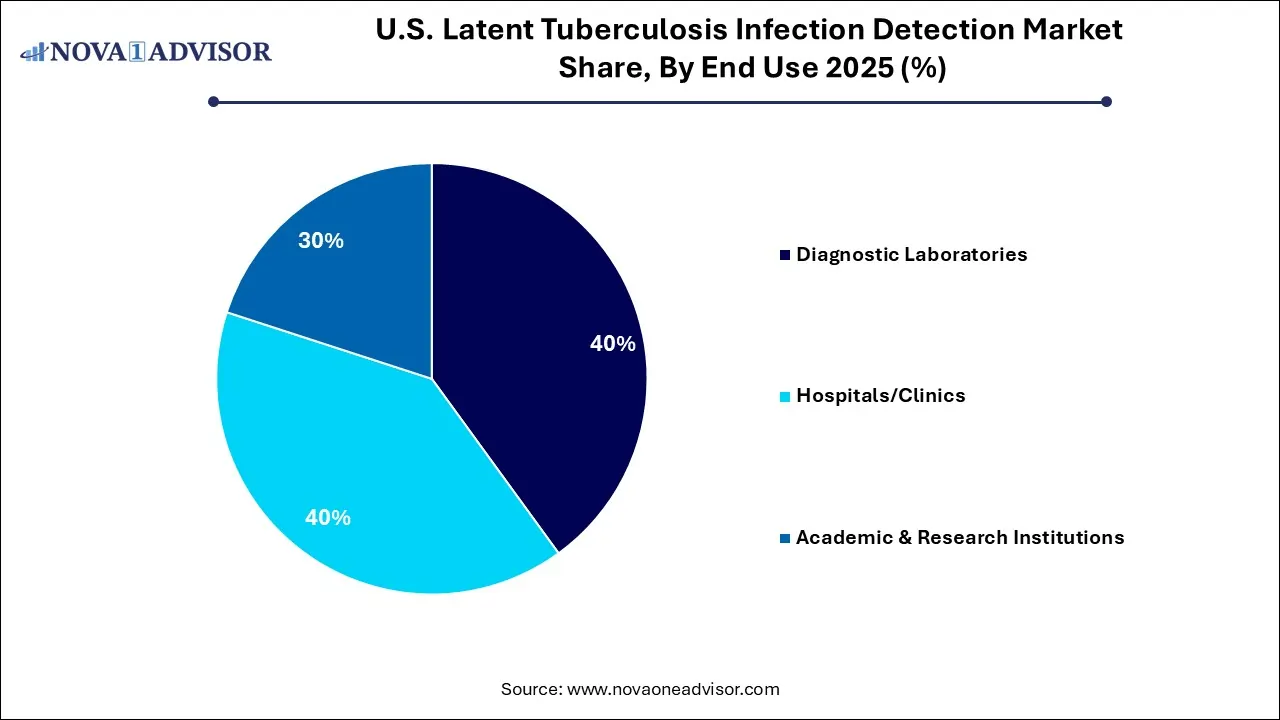
The diagnostic laboratories segment held the 40% share of the market in 2025, with its specialization in conducting tests, serving as the primary testing location for both IGRA and TST. The demand for rapid, accurate diagnostics to initiate timely treatment has increased reliance on commercial diagnostic labs. Growing expansion of retail/ independent settings and growing adoption of advanced technology.
The academic & research institutions segment held 20% share of the market in 2025, due to its focus on research rather than direct patient care, limiting its dominance compared to diagnostic labs and hospitals/clinics. Academic centers act as hubs for public health surveillance and clinical research, supporting the nationwide goal of identifying and treating latent TB in high-risk populations.
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the U.S. latent tuberculosis infection detection market.
By Test
By Application
By End Use