U.S. Pressure Infusor Market Summary
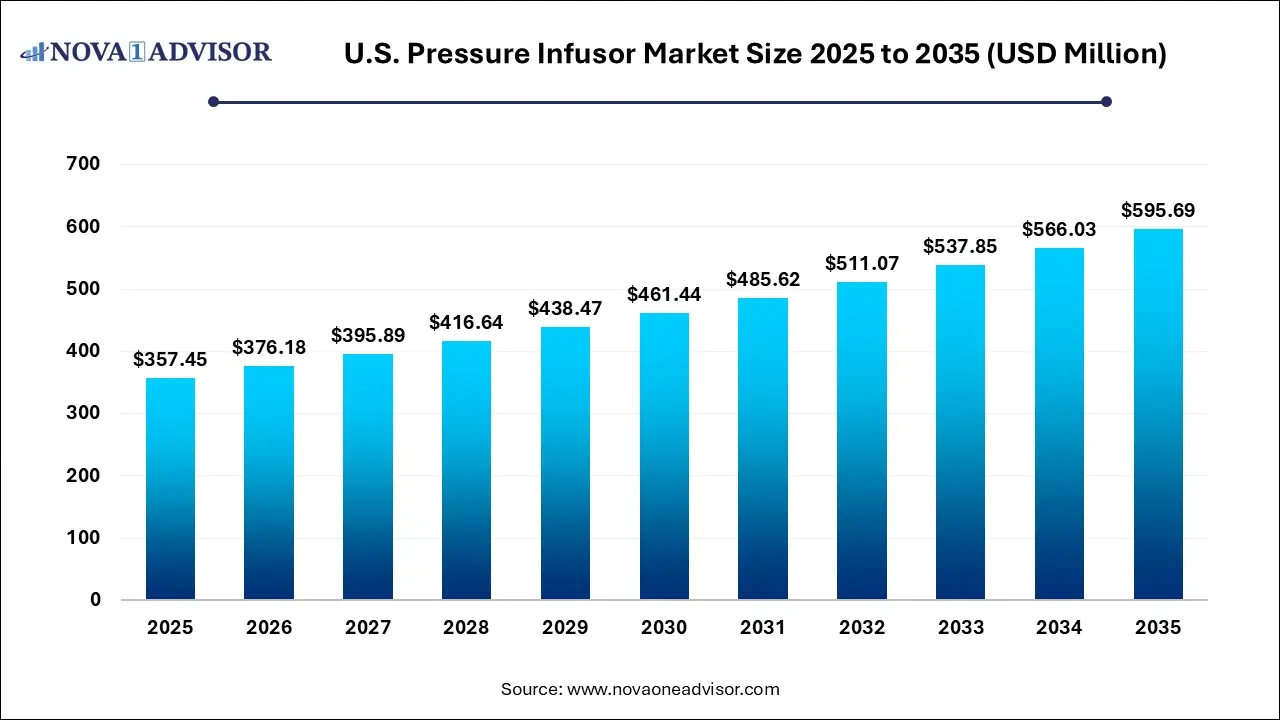
The U.S. pressure infusor market size was estimated at USD 357.45 million in 2025 and is projected to increase from USD 376.18 million in 2026 to approximately USD 595.69 million by 2035, growing at a CAGR of 5.24% from 2026 to 2035. The U.S. pressure infuser market is driven by the rising surge in surgeries, emergency room visits, and trauma cases, which drive the need for rapid, controlled infusion of blood products and IV fluids. Technology innovation is shifting the smart, automated, and electronic pressure infusors that improve accuracy, ease of use, and patient safety, and the rising demand for efficient healthcare.
Key Takeaways
- By product, the reusable pressure infusors segment held a dominant position in the market with a share of 65% in 2025.
- By product, the disposable pressure infusors segment is the fastest growing between 2026 and 2035.
- By capacity, the 1000 mL segment accounted for a considerable revenue share of 45% in the market in 2025.
- By capacity, the 500mL segment is the fastest growing during 2026 and 2035.
- By application, the emergency & critical care resuscitation segment is the dominant in the U.S. pressure infusor market with a share of 35% in 2025.
- By end use, the hospitals and clinics segment held the dominant position in the market with a share of 70% in 2025.
U.S. ressure infusor comprises a specialized medical device sector focused on providing controlled, external pressure to accelerate the delivery of intravenous fluids, blood products, or medications in acute care settings. Primarily utilized in emergency rooms, operating rooms, and intensive care units, these devices allow for rapid resuscitation and continuous fluid administration when gravity alone is insufficient. The primary advantages include high efficiency in critical care settings, improved patient outcomes through rapid blood transfusion, and enhanced infection control, especially with the growing adoption of single-use disposable pressure infusors.
U.S. Pressure Infusor Market Trends
- Dominance of Single-Use Infusos
The hospitals are increasingly adopting single-use bags to minimize the risk of cross-contamination, reduce sterilization costs, and adhere to strict hospital infection-control protocols.
- Technological Shift Toward Smart and Automated Devices
The shift from manual hand-pump systems to automated or smart electric pressure infusors integrated with IoT sensors. These advanced systems allow real-time monitoring of fluid delivery, accurate pressure control, and automated alerts to prevent accidental air embolism, thereby improving patient safety during critical procedures.
- Expansion in Ambulatory and Home Healthcare Settings
As more procedures move outside traditional hospital settings, there is a rising demand for portable, compact pressure infusors in ambulatory surgery centers and home-care programs. These portable devices are designed to be user-friendly, supporting the administration of specialty biologics or fluids in field emergencies.
Key AI Integration in the U.S. Pressure Infusor Market
AI and ML are transforming the U.S. pressure infusor industry by shifting devices from manual, reactive tools to intelligent, connected platforms that improve patient safety and clinical efficiency. These technologies enable real-time monitoring of fluid flow and pressure, utilizing predictive algorithms to detect anomalies such as occlusions, infiltration, or incorrect dosage administration before they cause harm. By integrating with hospital electronic health records and Internet of Medical Things systems, AI-powered infusors can analyze real-time patient data to provide automated, personalized treatment adjustments rather than relying on one-size-fits-all settings. AI enhances operational performance through predictive maintenance, identifying when a pump requires servicing to prevent unexpected failures and reduce downtime.
U.S. Pressure Infusor Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 376.18 Million |
| Market Size by 2035 |
USD 595.69 Million |
| Growth Rate From 2026 to 2035 |
CAGR of 5.24% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
Product, Capacity, Application, End use |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Key Companies Profiled |
ICU Medical, Inc.; Medline; Merit Medical Systems; ASP Global (acquired by Platinum Equity); Tapmedic LLC; Spacelabs Healthcare; AirLife; VBM Medical Inc. |
Market Dynamics
Driver
Increasing Surgical Procedures
The increasing number of surgical procedures in the U.S., particularly elective and minimally invasive surgeries, is driving demand for tools that enable rapid, controlled fluid management and blood transfusion during operations. Pressure infusors are essential in these settings for maintaining stable blood volume and managing trauma, ensuring patient safety during perioperative care. The rapid growth of ambulatory surgery centers (ASCs) for outpatient procedures is accelerating the adoption of disposable pressure infusion bags, which offer quick setup and infection control.
Restraint
Stringent Regulatory Standards
The market growth is hindered by imposing high compliance costs, which can consume up to 5% of device revenue. These rigorous approval processes for new materials and designs result in lengthy research and development cycles, creating bottlenecks that slow down innovation and entry into the market. The increased regulatory scrutiny on sterilization and infection control restricts the use of certain materials, escalating production costs for manufacturers and reducing their manufacturing flexibility.
Opportunity
Disposable and Eco-Friendly Products
The addressing critical need for infection control while aligning with sustainability goals. The rapid shift toward single-use, disposable infusion bags eliminates the costly and time-consuming sterilization processes associated with reusable devices, mitigating cross-contamination risks in high-acuity hospital and emergency settings. Manufacturers are capitalizing on this by introducing innovative materials, such as PVC- free polymers and biodegradable materials, which appeal to healthcare institutions aiming to improve their environmental footprint. The high volume-turnover in ambulatory surgical centers and home healthcare settings drives demand for sterile, ready-to-use disposable pressure infusor bags.
Segment Insights
By Product Insights
Why Did the Reusable Pressure Infusors Segment Hold a 65% Share for the U.S. Pressure Infusor Market?
The reusable pressure infusors segment held a dominant position in the market with a share of 65% in 2025, owing to the rising concerns over infection control and reuse limitations. The high concentration of advanced hospital infrastructure allows for proper cleaning and makes them ideal for high-volume, cost-conscious healthcare environments. The growing healthcare facilities still prioritize durable equipment to reduce total waste management.
The disposable pressure infusors segment held a 35% share of the market in 2025. Due to the safety benefits and regulatory preference for single-use devices. The growing need for a costly and time-consuming sterilization process required for reusable alternatives streamlines workflows in emergency and critical care settings.
By Capacity Insights
Why Did the 1000 mL Segment Lead the U.S. Pressure Infusor Market?
The 1000 mL segment registered its dominance over the market with a share of 45% in 2025, owing to the growing services as a middle-ground solution in emergency care, intensive care units, and operating rooms, offering higher volume capacity, and remaining portable. These bags are ideal for surgeries requiring consistent moderate fluid delivery and are frequently used to treat chronic diseases that require consistent hospital interventions, with a growing demand.
The 500 ml segment held the 30% share of the market in 2025, due to limited use cases compared to standard capacities. This capacity is considered the standard in many hospital environments, making it the most utilized volume for emergencies and blood pressure monitoring. It is essential in ICU, OR, and trauma settings for rapid, consistent flow rate control of intravenous fluids and blood products. The growing demand for improved patient care, increasing surgical volumes, and advancements in bag safety drive the demand.
The 3000 ml segment held a 25% share of the market in 2025 because of these are extensively used in trauma, OR, and intensive care units for handling serious blood loss injuries and rapid fluid resuscitation. This is ideal for consistent, large-volume infusion over a longer period in surgical environments.
By Application Insights
Why Did the Emergency & Critical Care Resuscitation Segment Hold a 35% Share for the U.S. Pressure Infusor Market?
The emergency & critical care resuscitation segment led the market with a share of 35% in 2025, due to the increasing emergency cases and need for rapid response. The growing prevalence of severe cardiovascular emergencies and trauma cases necessitates fast fluid resuscitation, advancement in, and the adoption of high-quality, safe, and disposable pressure infusion solutions to improve patient outcomes.
The blood & blood product transfusion segment held a 30% share of the market in 2025, with the high incidence of emergency and critical care and increased surgical procedures, such as cardiovascular and orthopedic surgeries, demand efficient blood transfusion tools. The rising need for rapid, controlled administration and adoption of disposable technology.
The surgical fluid management segment held a 25% share of the market in 2025, due to the gradual adoption of advanced fluid management systems. Pressure infusors are critical for rapid blood transfusion and intravenous fluid management, minimizing risks associated with fluid overload and high demand in acute care. Growing adoption of minimally invasive procedures and rising surgical procedures.
The others segment held a 10% share of the market in 2025, due to the rapid adoption of automated, smart, and user-friendly pressure infusers that offer better precision and safety, particularly in advanced, specialized care settings. Growth in specialized therapeutic areas and high-value features.
By End-Use Insights
Why Did the Hospitals and Clinics Segment Hold a 70% Share for the U.S. Pressure Infusor Market?
The hospitals and clinics segment led the market with a share of 70% in 2025, due to the hospitals' high-volume utilization and adoption of advanced and safe technologies. The rising prevalence of chronic conditions requires hospital-based interventions and intravenous therapies. The widespread availability of specialized equipment and the shift to an alternative setting.
The ambulatory surgical centers (ASCs) segment held a 15% share of the market in 2025. With the shift from inpatient to outpatient care, it is cost-effective and efficient, often using disposable pressure infusor technology. The rise in minimally invasive and surgical procedures, infusion kits that improve patient throughput, and simplify infection control compared to reusable systems used in traditional hospitals.
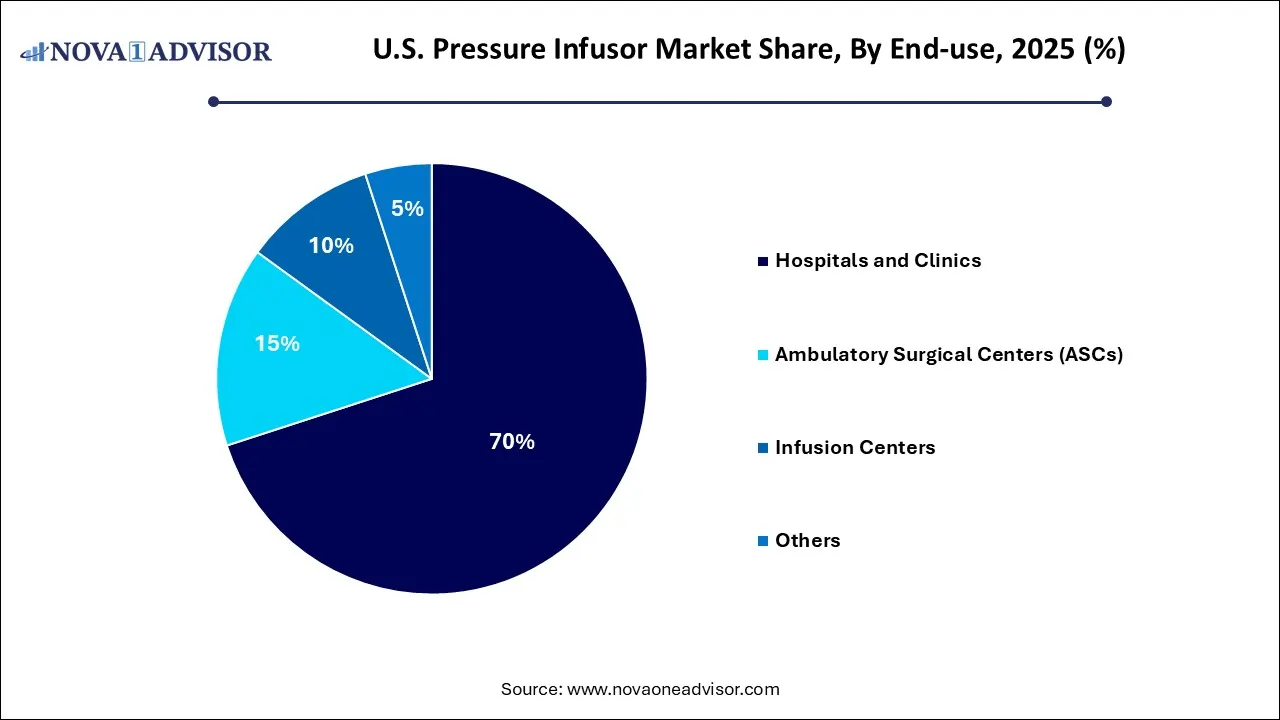
The infusion centers segment held a 10% share of the market in 2025, as the rising demand for specialized care services. The increasing cases of chronic illnesses necessitate frequent hospitalizations and reliable, continuous intravenous therapies. Introduction of user-friendly, automated, and reliable pressure infusors encourages higher adoption rates in hospitals and outpatient centers.
The others segment held a 5% share of the market in 2025, due to the lower adoption and limited scale compared to institutional care. Strong, rapid adoption of technological advancement, growth in specialized therapeutic areas. Its high value features and develops strategic customization.
Recent Developments
- In February 2025, iV-GO Mechanical IV Pump: Introduced by OSAA Innovation, this is the industry's first pole-free, fully mechanical IV pump. It eliminates power and pole constraints to significantly enhance patient mobility in mobile and on-the-go care environments.
- In December 2024, Endo, Inc. introduced the ADRENALIN® (epinephrine in 0.9% sodium chloride injection) ready-to-use premixed bag. This is described as the first and only FDA-approved, manufacturer-prepared epinephrine premixed intravenous (IV) bag. The product is indicated for use in adult patients to increase mean arterial blood pressure during hypotension associated with septic shock.
- In February 2026, the FDA finalized the transition to the quality management system regulation (QMSR). This aligns U.S. domestic requirements with international ISO 13485:2016 standards, streamlining global compliance for manufacturers.
U.S. Pressure Infusor Market Companies
- ICU Medical, Inc.
- Merit Medical Systems.
- Medline
- ASP Global (acquired by Platinum Equity)
- Spacelabs Healthcare
- Tapmedic LLC
- VBM Medical Inc.
- AirLife
U.S. Pressure Infusor Market Market Segment
By Product
- Reusable pressure infusors
- Disposable pressure infusors
- By Capacity
- 500 mL
- 1000 mL
- 3000 mL
By Application
- Emergency & Critical Care Resuscitation
- Blood and Blood Product Transfusion
- Surgical Fluid Management
- Others
By End Use
- Hospitals and Clinics
- Ambulatory Surgical Centers
- Infusion Centers
- Others



