Cell Therapy Human Raw Materials Market Size and Growth 2026 to 2035
The global cell therapy human raw materials market size was estimated at USD 4.75 billion in 2025 and is expected to hit USD 25.76 billion in 2035, expanding at a CAGR of 18.42% over the forecast period of 2026-2035. The market growth is driven by rising demand for cell-based therapies, the industry is moving from autologous to allogeneic therapies, which requires higher volumes of raw materials, such as serum albumin drive growth, and significant investment for research and development.
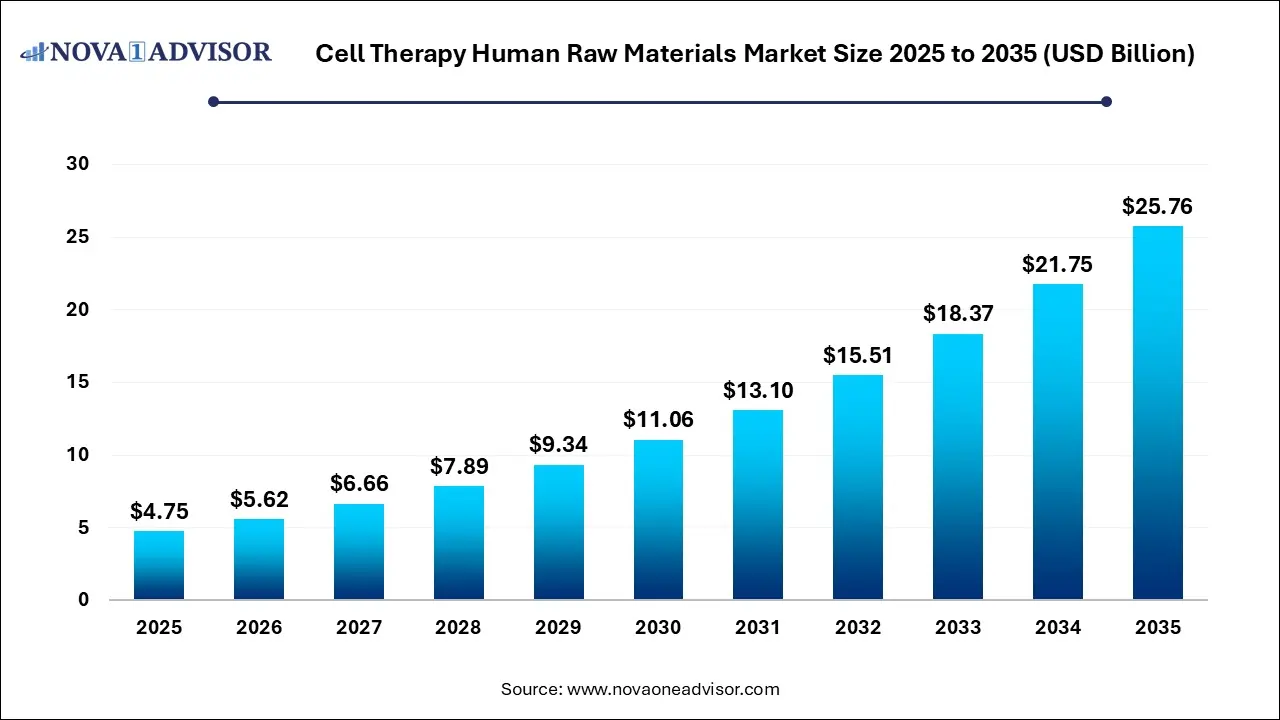
Cell Therapy Human Raw Materials Market Key Takeaways
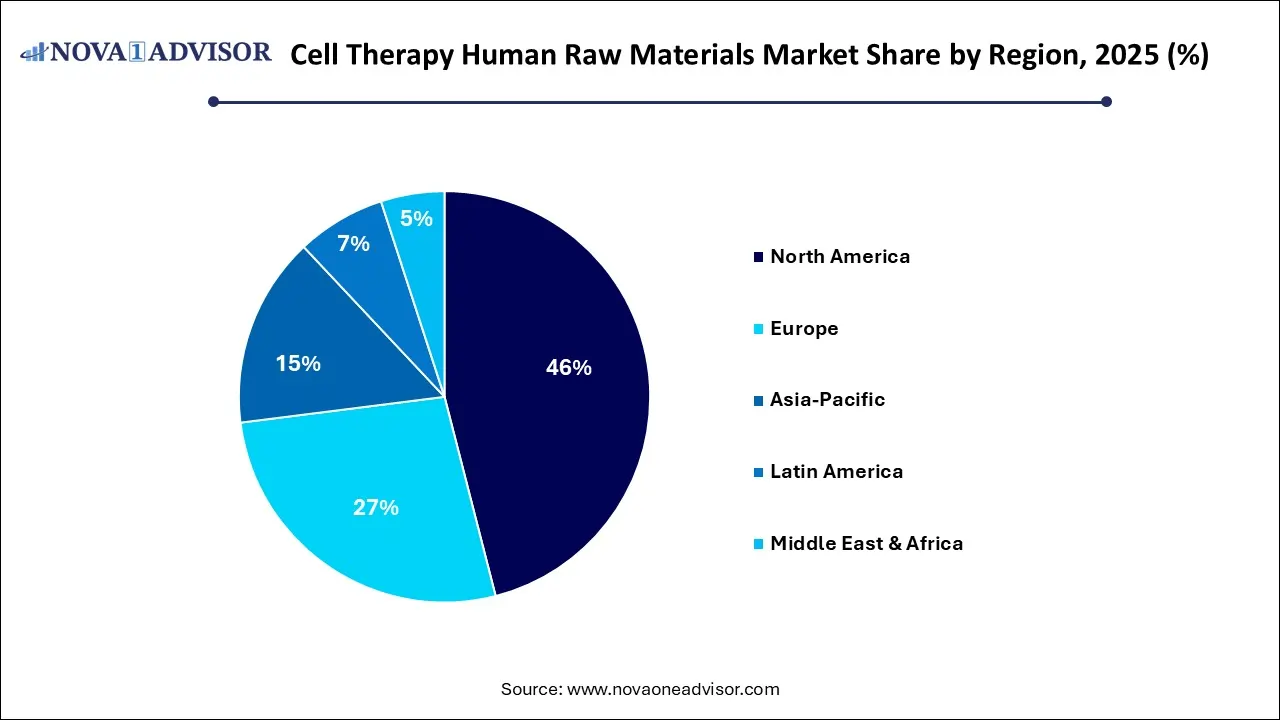
- By region, North America held the largest share of the cell therapy human raw materials market in 2025.
- By region, Asia Pacific is expected to experience the fastest growth between 2025 and 2035.
- By product, the cell culture supplements segment led the market in 2025.
- By product, the cell culture media segment is expected to expand at the highest CAGR over the projected timeframe.
- By end use, the biopharmaceutical & pharmaceutical companies segment led the market in 2025.
- By end use, the CROs and CMOs segment is the fastest growing in the cell therapy human raw materials market.
Artificial Intelligence: The Next Growth Catalyst in Cell Therapy Human Raw Materials
AI and ML are transforming the use of human raw materials in cell therapy by optimizing the identification, screening, and sourcing of critical donor materials, thereby significantly reducing reliance on manual processes. These technologies enable advanced predictive modeling to match donor profiles with specific therapeutic requirements, thereby increasing compatibility and potency. In production, ML algorithms facilitate real-time monitoring and quality control of cell culture media and raw materials, ensuring batch consistency and reducing contamination risks. AI drives the innovation of synthetic and semi-synthetic raw materials, helping to bridge the gap between in vitro and in vivo research.
Strategic Overview of the Global Cell Therapy Human Raw Materials Industry
The cell therapy human raw materials market comprises a specialized supply chain of biological and non-biological components, such as cytokines, GMP-grade media, serum, and enzymes, essential for the development and manufacturing of cell-based therapies. The market growth is driven by an increasing number of clinical trials and commercial approvals for CAR-T, TCR-T, and stem cell therapies aimed at treating cancer, autoimmune disorders, and rare diseases. A major advantage of using specialized human raw materials is that they ensure superior safety, high-consistency, and strict adherence to regulatory standards, which in turn boosts therapeutic efficacy.
Market Outlook
- Market Growth Overview: The cell therapy human raw materials market is expected to grow significantly between 2025 and 2035, driven by the increasing prevalanc eof cancer, autoimmune disorders, and diabetes is accelerating the need for, and development of, advanced cell-based therapies, growing in 3D cell culture and tissue engineering, particularly using scaffolds and matrices, and stong industrial demand for the strick regulated, good manufacturing practice compliant materials to ensure safety and quality in therapeutic production.
- Sustainability Trends: Sustainability trends are an intense focus on using good manufacturing practice components, traceable, and standardized materials to ensure safety and regulatory compliance, xeno-free & defined media minimize batch -to-batch variability and contamination risks, there is a strong shift away from animal-based ingredients towards chemically defined, serum-free, and xeno-free media. The industry shift towards allogenic therapies, which require a sustainable, high-volume supply of characterized human material like apheresis products.
- Major Investors: Major investors in the market include Thermo Fisher Scientific, which provides a comprehensive portfolio, while Merck KGaA and Danaher focus on high-purity, standardized components. STEMCELL Technologies, RoosterBio, and FUJIFILM Irvine Scientific focus on specific cell therapy needs. Pharmaceutical companies are major investors in the end-user space, driving demand for high-quality, clinical-grade materials for CAR-T and gene-modified therapies.
- Startup Economy: The startup economy in the market contains innovation in raw materials. Startups are creating synthetic or semi-synthetic materials and serum-free, chemically defined media, enhancing product safety, consistency, and reducing reliance on traditional, donor-derived, and potentially variable raw materials. Development of innovative technologies, such as improved cryopreservation and, in some cases, advanced cell expansion techniques, allows for more efficient, scalable, and cost-effective production, particularly for allogenic, off-the-shelf treatments.
Cell Therapy Human Raw Materials Market Report Scope
| Report Attribute |
Details |
| Market Size Value in 2026 |
USD 5.62 Billion |
| Revenue Forecast in 2035 |
USD 25.76 Billion |
| Growth Rate |
CAGR of 18.42% from 2026 to 2035 |
| Base Year For Estimation |
2025 |
| Forecast Period |
2026 - 2035 |
| Quantitative Units |
Revenue in USD Million/Billion, and CAGR from 2026 to 2035 |
| Segments Covered |
Products, End Use, Region |
| Regional Scope |
North America; Europe; Asia Pacific; Latin America; MEA |
| Key Companies Profiled |
Thermo Fisher Scientific, Inc; Merck KGaA; Actylis; ACROBiosystems; STEMCELL Technologies; Grifols, S.A.; Charles River Laboratories; RoosterBio, Inc.; PromoCell GMBH; Danaher; BioIVT; GeminiBio; Akron Biotech; AllCells; CGT Global |
Market Dynamics
Drivers
Surge in Cell Therapy Clinical Trials
The surge in cell therapy clinical trials, particularly in oncology and regenerative medicine, acts as a primary catalyst for the cell therapy human raw materials industry by creating an urgent need for high-grade biological inputs such as human serum, growth factors, and specialized cell culture media. As biopharmaceutical companies advance candidates through late-stage trials, the demand for large-volume, GMP-compliant, and xeno-free raw materials rises exponentially to ensure product safety, consistency, and efficacy.
Rising Prevalence of Chronic Diseases
The rising prevalence of chronic diseases, such as cancer, diabetics and cardiovascular disorders, is a primary driver fueling the demand for advanced cell-based therapies, which in turn accelerates the cell therapy human raw materials market. As these conditions become more widespread globally, the need for personalized medicine and regenerative treatments has led to an increase in clinical trials and commercial production, requiring large quantities of high-quality raw materials like cell culture media, serum, and growth factors.
Restraint
Stringent Regulatory Compliance
The market growth is hindered by imposing high costs and complexities on sourcing and validating donor-derived materials. Regulatory agencies like the FDA and EMA demand rigorous safety protocols, including extensive testing and heavy documentation for contamination control, which increases development timelines and adds to overall production expenses. The lack of global harmonization in these standards creates a fragmented landscape, compelling manufacturers to perform redundant testing and establish complex, segregated supply chains.
Opportunities
Standardization of Off-the-Shelf Allogeneic Therapies
The standardization of off-the-shelf allogeneic therapies is driving significant growth in the cell therapy human raw materials market by facilitating a shift from personalized, low-volume production to large-scale, industrial manufacturing. As companies aim to produce multiple doses from a single healthy donor, the demand for high-quality, standardized raw materials, such as GMP-grade culture media, growth factors, and cryopreservation agents, increases dramatically to ensure product consistency and safety.
Emergence of IPSC-based Therapies
The market is creating immersive opportunities by creating high demand for specialized, clinical-grade biological inputs. As iPSC-derived cell therapies transition from research to commercial-scale production for conditions such as Parkinson's and heart disease, developers require specialized, serum-free, and xeno-free culture media, growth factors, and high-quality human serum. This shift requires robust sourcing of donor-derived raw materials that meet strict GMP standards to ensure safety, consistency, and efficacy in personalized medicine.
How Macroeconomic Variables Influence the Cell Therapy Human Raw Materials Market?
Economic Growth and GDP
Economic growth and rising GDP generally lead to positive growth. By boosting investments in biotech R&D, accelerating clinical trials, and enhancing healthcare infrastructure for sophisticated treatments. Rising affluence leads to a higher demand for advanced, sophisticated therapies for chronic diseases, creating a surging need for high-quality, GMP-grade raw materials such as cell culture media and growth factors.
Inflation & Drug Pricing Pressures
It can negatively affect the growth of the cell therapy human raw materials market by increasing production costs and limiting profitability for manufacturers, particularly in a high-inflation environment where logistics and input costs are rising significantly. These economic headwinds create budgetary constraints that can delay clinical trials and reduce overall investment in the development of new therapies.
Exchange Rates
Exchange rate fluctuations can negatively affect, by introducing volatility and increasing procurement costs for specialized, imported components. Fluctuations in currency values can disrupt global supply chains and significantly impact the profitability and operational flexibility of manufacturers, particularly smaller biotech companies.
Segment Outlook
Product Insights
Why Did the Cell Culture Supplements Segment Dominate the Cell Therapy Human Raw Materials Market in 2025?
The cell culture supplements segment dominated the market with the largest share in 2025. This is due to the growing volume of clinical trials, which have established specialized supplements as indispensable for scalable and effective cell therapy manufacturing. This demand is driven by a critical industry shift toward high -quality, traceable raw materials that satisfy stringent safety and regulatory standards for clinical use. The advanced supplements have become the foundation for ensuring cell viability and consistent performance across both research and commercial-scale production workflows.
The cell culture media segment is expected to grow at the fastest CAGR during the projection period, driven by the convenience of increasing demand for high-quality standards and rising preclinical research has established specialized cell culture media as critical for safe and reproducible therapy production. This demand is further amplified by strict regulatory requirements for chemically defined formulations that ensure product purity and efficiency at a commercial scale. The optimized media has become the foundation for scaling manufacturing workflows while maintaining the consistent performance necessary for complex cell-based therapies.
End Use Insights
How Does the Biopharmaceutical & Pharmaceutical Companies Segment Lead the Cell Therapy Human Raw Materials Market in 2025?
The biopharmaceutical & pharmaceutical companies segment led the cell therapy human raw materials market in 2025 due to the coverage of substantial R&D investments, and the transition to commercial-scale manufacturing has established validated raw materials as the backbone of the cell therapy industry. The stringent regulatory scrutiny from bodies, such as the FDA and EMA, which mandates the use of high-quality, GMP-compliant inputs to ensure patient safety. The integration of automated closed-system bioreactors has become essential for maintaining consistency and scalability across increasingly complex therapeutic pipelines.
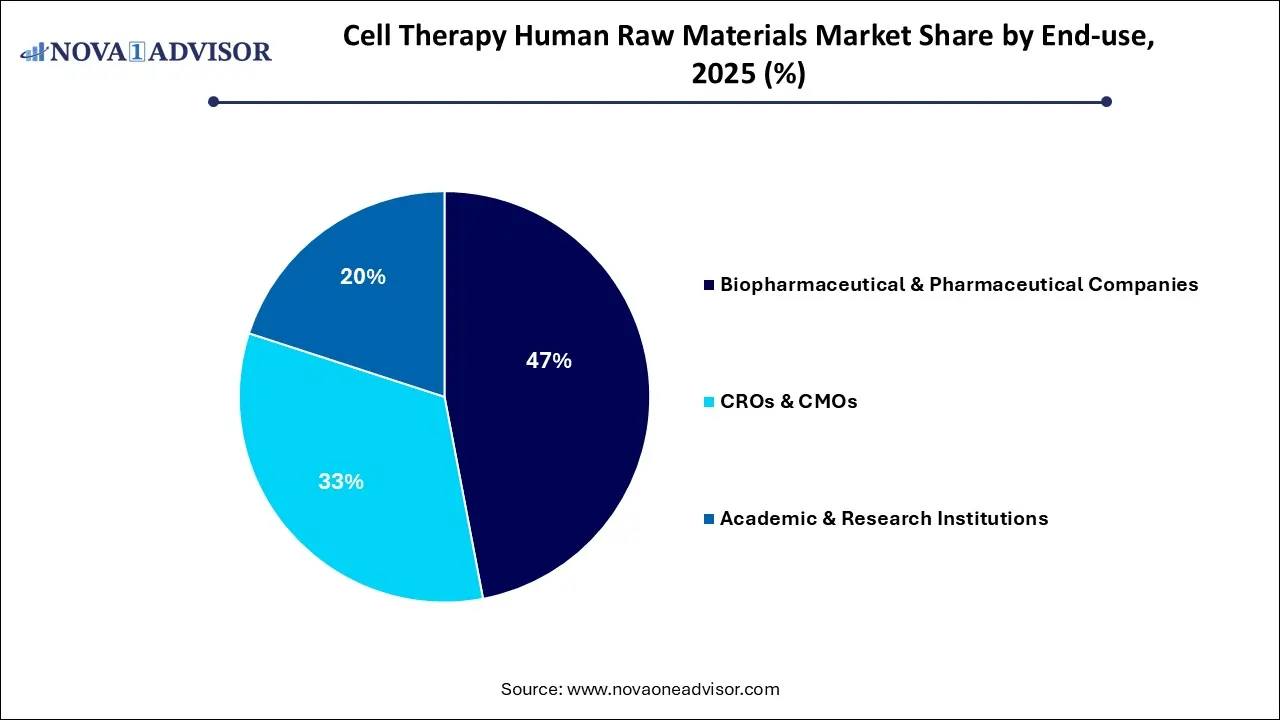
The CRO and CMOs segment is projected to grow at the highest CAGR in the coming years. This is mainly because of high-quality, GMP-compliant raw materials like cytokines and viral vectors. The industry's need for specialized expertise, cost efficiency, and accelerated clinical timelines that smaller biotech firms cannot achieve alone. The infrastructure expansion and technological innovations in automation within these contract organizations are setting new benchmarks for large-scale production reliability. These factors collectively create a robust, globalized supply chain that is essential for the commercial viability of next-generation cell therapies.
Regional Analysis
What Made North America the Dominant Region in the Market?
North America maintained dominance in the cell therapy human raw materials market while holding the largest share in 2025. The region’s dominance is attributed to the convergence of high consumer demand, technological leadership, and robust financial markets, which have established North America as a premier global hub for innovation and industrial production. The vast natural resource wealth and the economic integration provided by the USMCA, which streamlines cross-border supply chains. The advanced logistics and world-class infrastructure ensure the region remains highly competitive for large-scale business investment and trade.
U.S. Cell Therapy Human Raw Materials Market Trends
The U.S. is a major contributor to the market in North America due to the industry-wide transition toward cGMP-compliant and xeno-free materials, which has established high-quality standards as the foundation for clinical and commercial safety. The shift toward allogeneic therapies and localized, domestic manufacturing secures the supply chain for complex products like viral vectors and CAR-T cells. The massive capital investments from market leaders are building the infrastructure necessary to support long-term scalability and global therapeutic reach.
What Makes Asia Pacific the Fastest-Growing Area in the Market?
Asia Pacific is expected to grow at the fastest rate in the coming years. This is due to accelerated by proactive government funding and a robust network of local CDMOs that are successfully driving down production costs through supply chain localization. The combination of an aging demographic and a massive patient pool is fueling a rapid transition toward commercial-scale allogeneic and autologous therapies. These factors collectively position the region as a pivotal global hub for innovative manufacturing and the widespread adoption of next-generation cellular medicines.
China Cell Therapy Human Raw Materials Market Trends
China is a key player in the Asia Pacific cell therapy human raw materials market due to a critical industry shift toward standardized, GMP-grade materials and automated, closed-system processing technologies to ensure safety and efficiency. The high demand across oncology and regenerative medicine is driving massive investment into the localized production of specialized media and reagents. These factors collectively create a high-velocity ecosystem that is fundamental to the commercial scalability of next-generation therapies in the region.
How is the Opportunistic Rise of Europe in the Cell Therapy Human Raw Materials Market?
Europe is experiencing a strategic growth in the market due to rigorous regulatory frameworks, which mandate a strategic transition toward standardized, xeno-free, and high-purity biological inputs. Consequently, massive capital injections into CDMO infrastructure across the UK, Germany, and Denmark are successfully localizing supply chains and enhancing manufacturing resilience. These factors collectively drive a sophisticated industrial ecosystem that is fundamental to the commercial scalability of advanced medicinal products within the region.
United Kingdom Cell Therapy Human Raw Materials Market Trends
The United Kingdom is leading the market in Europe due to a surge in demand for MHRA-compliant, GMP-grade inputs and the rapid adoption of automated, closed-system bioreactors for consistent commercial-scale production. The industry’s focus on allogeneic off-the-shelf therapies is driving massive investment into scalable raw material supply chains and long-term supplier partnerships. These factors collectively create a resilient and standardized manufacturing ecosystem essential for the global commercialization of next-generation medicinal products.
Cell Therapy Human Raw Materials Market Supply Chain Analysis
- Sourcing and Biological Extraction
This foundational stage involves the procurement of essential raw materials of human origin, such as human serum, plasma, leukopaks, and AB serum. It focuses on ethical, traceable sourcing and the initial extraction, requiring strict donor testing and screening to prevent contamination.
Key Players: Grifols, S.A., BioIVT, AllCells, CGT Global, Charles River Laboratories.
- GMP Manufacturing, Purification, and Formulation
This core stage involves converting raw inputs into high-quality, clinical-grade materials (cGMP) in ISO-certified cleanrooms, including culture media, growth factors, and cytokines. It includes advanced purification, chemical synthesis, and the development of chemically defined, serum-free formulations to ensure consistency and eliminate variability.
Key Players: Thermo Fisher Scientific, Merck KGaA, Danaher, STEMCELL Technologies, RoosterBio, Inc., PromoCell GmbH, Akron Biotech, and ACROBiosystems.
- Cold Chain and Distribution
Most human raw materials are highly temperature-sensitive, requiring sophisticated cold-chain logistics to maintain biological activity. This stage involves real-time monitoring and specialized handling to ensure material integrity from the factory to the point of use.
Key Players: Thermo Fisher Scientific, Danaher.
Cell Therapy Human Raw Materials Market Companies
- Thermo Fisher Scientific, Inc: Provides a comprehensive portfolio of high-quality raw materials, including Gibco cell culture media, serum, reagents, and CTS (Cell Therapy Systems) products designed for GMP-compliant cell therapy manufacturing.
- Merck KGaA (MilliporeSigma): Supplies high-purity, chemically defined cell culture media, supplements, and raw materials essential for the safe and efficient cultivation of stem cells and CAR-T therapies. They recently invested heavily in expanding their U.S. cell culture media production capacity to meet the increasing demand for regulatory-compliant inputs in the cell and gene therapy sector.
- Actylis: Specializes in sourcing and manufacturing complex raw materials, providing tailored solutions such as customized media, reagents, and buffers that meet strict GMP requirements. They focus on streamlining supply chains for pharmaceutical and biopharmaceutical partners, ensuring the secure procurement of key inputs crucial for cell therapy development.
- ACROBiosystems: Offers high-quality GMP-grade recombinant proteins, cytokines, and activation beads under their Resilient Supply brand to facilitate the expansion and activation of immune cells.
- STEMCELL Technologies: Specializes in providing specialized, serum-free, and animal-component-free cell culture media, such as the TeSR family, to ensure high-quality and consistent cell growth for therapeutic research.
- Grifols, S.A.: Contributes to the cell therapy market by supplying plasma-derived biological materials, such as Human Serum Albumin, which are vital as raw materials in cell and gene therapy processes.
Recent Developments
- In February 2026, Gemini Bioproducts (GeminiBio) and Tolemy Bio launched aiMOS™ (Artificial Intelligence Media Optimization Service). This service uses artificial intelligence and machine learning to design custom media supplements. These supplements improve the performance, consistency, and scalability of cell therapy manufacturing.
- In April 2025, the U.S. Pharmacopeia (USP) launched a comprehensive solutions package specifically designed to standardize the manufacturing and quality control of Adeno-Associated Virus (AAV) based gene therapies. This initiative addresses the technical and regulatory hurdles that currently slow the development of treatments for genetic and rare diseases.
- In August 2025, ProBio Inc., a global leader in contract development and manufacturing (CDMO), officially launched its cGMP Adeno-Associated Virus (AAV) manufacturing services at its 128,000 sq. ft. facility in Hopewell, New Jersey. This expansion is designed to provide a single-site solution for gene therapy developers, significantly reducing the complexity and timelines associated with viral vector production.
Exclusive Analysis on the Cell Therapy Human Raw Materials Market
The global cell therapy human raw materials market is leveraging the rising number of CAR-T and stem cell clinical trials, forcing a shift towards scalable, GMP-compliant, and well-characterized biological inputs like leukopaks, human serum, and serum-free media. Key trends include the transition from xenogeneic materials to xeno-free, chemically defined alternatives to minimize contamination risks and ensure regulatory compliance. Biopharmaceutical companies dominate demand, while CDMOs are experiencing the fastest growth in procurement due to increased outsourcing. Although North America currently holds the largest market share, Asia-Pacific is set to witness the fastest growth, reshaping the global sourcing landscape through heightened localization
Cell Therapy Human Raw Materials Market Report Segmentation
By Product
- Cell Culture Media
- Cell Culture Sera
- Cell Culture Supplements
-
- Proteins
- Growth Factors
- Nucleotides
- Other Supplements
- Reagents & Buffers
- Other Raw Materials
By End Use
- Biopharmaceutical & Pharmaceutical Companies
- CROs & CMOs
- Academic & Research Institutions
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa



