Viral Vectors & Plasmid DNA Manufacturing Market Size and Growth 2025 to 2034
The global viral vector & plasmid DNA manufacturing market was valued at USD 6.52 billion in 2024 and is projected to hit around USD 27.71 billion by 2034, growing at a CAGR of 15.57% during the forecast period 2025 to 2034. The growth of the market is attributed to the rising demand for cell and gene therapies and increasing R&D investments.
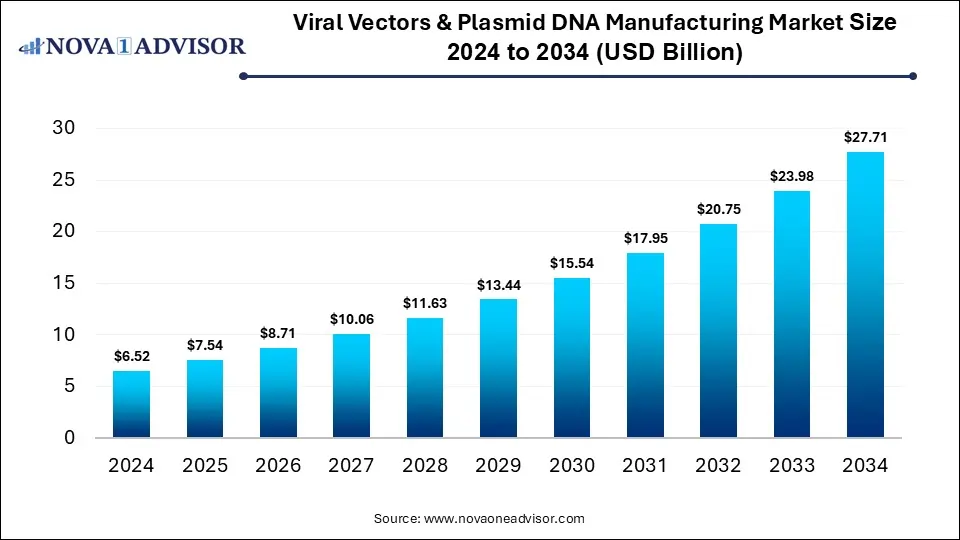
Viral Vectors & Plasmid DNA Manufacturing Market Key Takeaways
- North America dominated the viral vector & plasmid DNA manufacturing market with the largest share in 2024.
- Asia Pacific is likely to grow at the highest CAGR during the forecast period.
- By vector type, the AAV segment led the market in 2024.
- By vector type, the lentivirus segment is expected to grow at the fastest rate in the upcoming years.
- By workflow, the downstream processing segment sustained dominant position in the market in 2024.
- By workflow, the upstream processing segment is expected to experience rapid growth in the market.
- By application, the vaccinology segment accounted for the largest revenue share in 2024.
- By application, the cell therapy segment is likely to grow at a significant rate throughout the forecast period.
- By disease, the cancer segment dominated the market in 2024.
- By disease, the genetic disorders segment is likely to grow at a significant rate in the coming years.
- By end-user, the research institutes segment captured the largest share of the market in 2024.
- By end-user, the biopharmaceutical and pharmaceutical companies segment is expected to expand at the highest CAGR in the coming years.
How is AI Impacting the Viral Vector & Plasmid DNA Manufacturing Market?
AI is significantly transforming the market for viral vector & plasmid DNA manufacturing by enhancing process efficiency and accuracy. It enables predictive analytics for optimizing production parameters, reducing errors, and improving yield consistency. AI-driven automation accelerates scale-up and quality control, minimizing human intervention and operational costs. Additionally, machine learning models help in faster troubleshooting and maintenance, reducing downtime. Overall, AI is driving innovation, boosting manufacturing scalability, and supporting faster development of gene and cell therapies.
- In June 2025, Google DeepMind launched AlphaGenome, an AI tool that predicts how genetic variations affect gene regulation across up to 1 million DNA base pairs. Building on Enformer and complementing AlphaMissense, it focuses on interpreting the 98% of non-coding DNA linked to disease. AlphaGenome is available in preview via API for non-commercial research, with full release planned.
Market Overview
The viral vector & plasmid DNA manufacturing market revolves around the production of essential components used in gene and cell therapies, vaccines, and advanced biologics. Viral vectors enable efficient delivery of genetic material into target cells, while plasmid DNA serves as a versatile tool for gene expression and therapeutic development. These technologies offer advantages such as high specificity, scalability, and the ability to target previously untreatable diseases. The growth of this market is driven by increasing demand for personalized medicine, rising prevalence of genetic disorders, and advances in bioprocessing technologies. Additionally, expanding investments in biotech R&D and supportive regulatory frameworks are accelerating innovation and commercialization in this field.
What are the Major Trends in the Viral Vector & Plasmid DNA Manufacturing Market?
- Increased Adoption of Gene and Cell Therapies: Growing approval and commercialization of gene and cell therapies, such as CAR-T, are driving demand for viral vectors and plasmid DNA, fueling market expansion.
- Advancements in Manufacturing Technologies: Innovations like continuous bioprocessing, automation, and AI integration are improving production efficiency, yield, and quality control in manufacturing processes.
- Expansion of Contract Manufacturing Organizations (CMOs): More biotech and pharma companies are outsourcing vector and plasmid production to CMOs, accelerating capacity expansion and reducing time-to-market.
Report Scope of Viral Vectors & Plasmid DNA Manufacturing Market
| Report Coverage |
Details |
| Market Size in 2025 |
USD 7.54 Billion |
| Market Size by 2034 |
USD 27.71 Billion |
| Growth Rate From 2025 to 2034 |
CAGR of 15.57% |
| Base Year |
2024 |
| Forecast Period |
2025-2034 |
| Segments Covered |
Vector Type, Application, Workflow, End-User, Disease By Region |
| Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
| Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
Market Dynamics
Drivers
Increasing Approvals of Gene Therapy Products
The increasing approvals of gene therapy products are driving the growth of the viral vector & plasmid DNA manufacturing market, as these therapies rely heavily on high-quality viral vectors and plasmid DNA for effective gene delivery. Regulatory green lights accelerate the commercialization of these treatments, boosting demand for scalable and reliable manufacturing solutions. As more gene therapies enter the market, manufacturers must expand production capacity to meet clinical and commercial needs. This growth also encourages investments in advanced manufacturing technologies to ensure consistent quality and supply.
Approved Gene Therapies as of Q3 2024
| Product Name |
Generic Name |
Year First Approved
|
Disease(s) |
Locations Approved |
Originator Company |
| Elevidys |
delandistrogene moxeparvovec
|
2023 |
Duchenne muscular dystrophy |
U.S. |
Sarepta Therapeutics |
| Vyjuvek |
beremagene geperpavec |
2023 |
Dystrophic epidermolysis bullosa |
U.S. |
Krystal Biotech |
| Fucaso |
equecabtagene autoleucel |
2023 |
Multiple myeloma |
China |
Nanjing IASO Biotechnology |
| Casgevy |
exagamglogene autotemcel |
2023 |
Sickle cell anemia; thalassemia |
US, UK, Bahrain, Saudi Arabia, EU, Canada
|
CRISPR Therapeutics |
|
inaticabtagene autoleucel
|
inaticabtagene autoleucel |
2023 |
Acute lymphocytic leukemia |
China |
Juventas Cell Therapy |
| Lyfgenia |
lovotibeglogene autotemcel |
2023 |
Sickle cell anemia |
U.S. |
bluebird bio |
|
zevorcabtagene autoleucel
|
zevorcabtagene autoleucel |
2023 |
Relapsed or refractory multiple myeloma |
China |
CARsgen Therapeutics |
| Beqvez |
fidanacogene elaparvovec |
2024 |
Hemophilia B |
Canada, US, EU |
Pfizer |
| Tecelra |
afamitresgene autoleucel |
2024 |
Synovial sarcoma |
U.S. |
Adaptimmune |
Supportive Government and Regulatory Initiatives
Supportive government and regulatory initiatives also drive the growth of the viral vector & plasmid DNA manufacturing market by creating a favorable environment for research, development, and commercialization of gene and cell therapies. Policies that streamline approval processes and offer incentives like grants, tax breaks, and orphan drug designations encourage investment and innovation in this space. Regulatory frameworks focused on ensuring safety and efficacy while expediting reviews help bring therapies to market faster. Additionally, government funding for biomanufacturing infrastructure and public-private partnerships boost manufacturing capabilities. These initiatives collectively accelerate market expansion and improve patient access to advanced therapies.
- For instance, in March 2023, Touchlight secured a UK government grant through the £277 million Life Sciences Innovative Manufacturing Fund to accelerate its £14M enzymatic DNA manufacturing scale-up. The funding supports acquisition of scale-up equipment and expansion of its Hampton facility. It also enhances Touchlight’s capacity to support client API programs in late-stage development and commercialization.
Restraints
Complexity of Manufacturing Processes and Limited Production Capacity
The complexity of manufacturing processes and limited production capacity significantly restrain the growth of the viral vector & plasmid DNA manufacturing market. Producing high-quality vectors and plasmids require specialized equipment, stringent quality controls, and highly skilled personnel, making scale-up challenging and time-consuming. Many facilities lack the infrastructure to meet the growing clinical and commercial demand, leading to production bottlenecks and longer timelines. This limited capacity can delay the development and availability of gene and cell therapies, impacting patient access and market expansion. As a result, overcoming these manufacturing challenges is critical to sustaining the market’s long-term growth.
High Manufacturing Costs and Regulatory Challenges
High manufacturing costs and stringent regulatory challenges are key restraints in the market. Producing these components involves complex processes, costly raw materials, and advanced technologies, making the overall production expensive and less accessible for smaller developers. Additionally, regulatory agencies impose rigorous quality standards and compliance requirements to ensure the safety and efficacy of gene therapies, which can prolong development timelines and increase operational costs. Navigating these regulatory frameworks requires significant expertise and resources, creating barriers for new entrants.
Opportunities
Emergence of Non-Viral Alternatives and Hybrid Vectors
The emergence of non-viral alternatives and hybrid vectors is creating immense opportunities in the viral vector & plasmid DNA manufacturing market by expanding the scope of gene delivery technologies. Non-viral methods, such as lipid nanoparticles and electroporation, offer safer and potentially more cost-effective options, reducing reliance on traditional viral systems. Meanwhile, hybrid vectors combine the high efficiency of viral vectors with the safety and scalability of non-viral approaches, opening new avenues for therapeutic applications. These innovations address some of the limitations of conventional vectors, such as immunogenicity and production complexity. As demand for flexible and targeted gene delivery solutions grows, these alternatives are poised to complement and strengthen the overall market landscape.
Increased Focus on Personalized Medicine
The increased focus on personalized medicine is creating significant opportunities in the market. Viral vectors are essential for developing tailored gene and cell therapies. Personalized treatments often require custom vectors and plasmids to target specific genetic profiles, driving demand for flexible, small-batch, and high-quality manufacturing solutions. This trend is pushing manufacturers to adopt more adaptive and scalable platforms that can support individualized therapies. As precision medicine continues to gain traction in oncology, rare diseases, and genetic disorders, the need for specialized vector and plasmid production will grow rapidly. This shift not only fuels innovation but also expands the market’s reach across diverse therapeutic areas.
Segment Outlook
By Vector Type
What Made AAV the Dominant Vector Type in 2024?
The adeno-associated virus (AAV) segment dominated the viral vector & plasmid DNA manufacturing market in 2024 due to its strong safety profile, low immunogenicity, and ability to deliver genetic material to both dividing and non-dividing cells. AAV has become the preferred vector for many gene therapy applications, particularly in treating genetic disorders like spinal muscular atrophy and inherited retinal diseases. Its long-term gene expression and tissue-targeting capabilities have made it a top choice for clinical and commercial gene therapy programs. Additionally, a growing number of FDA and EMA approvals for AAV-based therapies have further accelerated investment and production capacity for AAV vectors. This rising demand across clinical trials and commercial applications positioned AAV as the dominant vector type in the market.
The Lentivirus segment is expected to grow at the fastest CAGR during the forecast period due to its widespread use in gene therapy, particularly for treating chronic and genetic disorders. Lentiviral vectors offer the unique advantage of stable gene integration into both dividing and non-dividing cells, making them highly effective for long-term therapeutic applications. The rising number of clinical trials and regulatory approvals involving lentiviral-based therapies, especially in oncology and rare diseases, further fuels this growth. Additionally, advancements in vector engineering and manufacturing scalability are enhancing their safety and efficiency, increasing their adoption.
By Workflow
Why Did the Downstream Processing Segment Lead the Market in 2024?
The downstream processing segment led the viral vector & plasmid DNA manufacturing market in 2024 due to its critical role in ensuring product purity, potency, and safety, key factors in meeting stringent regulatory standards for gene therapies. As demand for high-quality vectors increased, manufacturers prioritized robust downstream processes such as purification, concentration, and formulation to remove impurities and achieve clinical-grade quality. Technological advancements in chromatography and filtration systems also improved efficiency, scalability, and yield, further boosting reliance on downstream operations. Given the complexity and sensitivity of viral vectors and plasmid DNA, downstream processing emerged as a bottleneck and a major cost driver, prompting heavy investments in this workflow.
The upward processing segment is expected to grow at the fastest rate in the upcoming period due to its efficiency in scaling production while maintaining product quality. Advances in bioprocessing technologies have improved yield and reduced costs, making this segment more attractive for large-scale manufacturing. Additionally, the rising demand for gene therapies and vaccines drives the need for robust, scalable upstream processes. The growth of this segment is further fueled by ongoing innovation and automation that enhance process control and consistency.
By Application
How Does the Vaccinology Segment Dominate the Viral Vector & Plasmid DNA Manufacturing Market?
The vaccinology segment dominated the market in 2024 due to the continued global demand for innovative vaccine platforms, especially following the success of viral vector-based COVID-19 vaccines. Viral vectors and plasmid DNA play a crucial role in the development of next-generation vaccines targeting infectious diseases like HIV, Zika, and influenza, as well as emerging pathogens. Governments and health organizations significantly increased funding and strategic partnerships to accelerate vaccine development, boosting production needs for viral vectors and plasmids. Additionally, DNA vaccines are gaining traction for their stability, rapid design, and ease of manufacturing, contributing to the segment’s growth.
The cell therapy segment is expected to expand at the highest CAGR over the projection period, owing to the increasing adoption of advanced therapies like CAR-T and stem cell treatments. These therapies require high volumes of viral vectors and plasmid DNA for gene modification and cell engineering, driving significant demand in the market. Moreover, a growing pipeline of cell therapy candidates, along with rising investments from both public and private sectors, is accelerating research and clinical development. Regulatory support for innovative cell-based treatments and their potential to address previously untreatable diseases further fuel this growth.
By Disease
Why Did the Cancer Segment Dominate the Viral Vector & Plasmid DNA Manufacturing Market?
The cancer segment dominated the market in 2024 due to the rapid expansion of gene and cell therapies targeting various cancers, including leukemia, lymphoma, and solid tumors. Viral vectors and plasmid DNA are critical components in the development of CAR-T cell therapies, oncolytic viruses, and cancer vaccines, which have shown promising clinical outcomes. As oncology remains a primary focus for biopharmaceutical companies, significant R&D investments and a growing number of clinical trials have driven up the demand for scalable, GMP-grade vector and plasmid production. The urgency to address unmet needs in cancer treatment, along with regulatory approvals of novel gene therapies, further accelerated market dominance in this segment.
The genetic disorders segment is expected to experience the fastest growth in the coming years due to the rising prevalence of inherited diseases and the increasing potential of gene therapy to provide long-term or curative treatments. Viral vectors and plasmid DNA are essential components in delivering corrective genes to patients with genetic conditions, making them critical to therapeutic development. Advances in gene editing technologies, such as CRISPR and AAV-based therapies, are accelerating the development pipeline for rare and monogenic disorders. Additionally, growing regulatory support and orphan drug incentives are encouraging investment and innovation in this area. As a result, demand for high-quality vector and plasmid production tailored to genetic disorder treatments is rapidly expanding.
By End-User
How Does Research Institutes Contribute the Largest Market Share in 2024?
The research institutes segment dominated the viral vector & plasmid DNA manufacturing market in 2024 due to their pivotal role in early-stage development of gene therapies, vaccines, and molecular biology research. These institutes are often at the forefront of innovation, conducting preclinical studies that rely heavily on custom viral vectors and plasmid DNA. With increased funding from governments, academic collaborations, and global health organizations, research institutes expanded their use of advanced vector platforms for exploratory and translational research.
Additionally, their partnerships with biotech companies for technology transfer and pilot-scale production further boosted demand for high-quality, research-grade vectors. This active involvement in discovery and proof-of-concept studies positioned research institutes as leading contributors to market growth in 2024.
The pharmaceutical and biopharmaceutical companies segment is expected to register the highest CAGR in the market during the forecast period due to their increasing focus on developing gene and cell therapies. These companies are heavily investing in R&D and expanding their pipelines to include advanced therapies, which require large-scale, high-quality viral vectors and plasmid DNA. Strategic collaborations, acquisitions, and in-house manufacturing expansions are also accelerating production capabilities to meet growing clinical and commercial demands. As more gene-based therapies receive regulatory approvals, pharma and biotech firms are scaling up manufacturing to support global distribution.
Regional Insights
What Made North America the Dominant Region in the Market in 2024?
North America dominated the viral vector & plasmid DNA manufacturing market by capturing the largest share in 2024 due to its strong presence of leading pharmaceutical and biotechnology companies actively developing gene and cell therapies. The region benefits from advanced healthcare infrastructure, substantial R&D investments, and supportive regulatory frameworks that accelerate the approval and commercialization of innovative treatments. Additionally, the high number of ongoing clinical trials and early adoption of cutting-edge biotechnologies have driven demand for viral vectors and plasmid DNA. Government funding and strategic collaborations between academia and industry have further strengthened manufacturing capabilities.
The U.S. is the major contributor to the North America viral vector & plasmid DNA manufacturing market. The country is home to a large number of biotechnology and pharmaceutical companies that are heavily invested in gene and cell therapy development. The country also leads in clinical trial activity, research funding, and regulatory support, all of which drive demand for viral vector and plasmid DNA production. Additionally, the presence of advanced manufacturing facilities and skilled workforce and government support further strengthens the U.S.'s dominant position in the regional market.
- In May 2024, the White House introduced new rules for synthetic DNA manufacturers to prevent misuse of mail-order genetic material, addressing biosecurity concerns. The move follows a 2023 executive order by President Biden focused on AI safety, including its application in biotechnology. Synthetic DNA enables breakthroughs in diagnostics, therapeutics, and research without sourcing natural organisms.
What Makes Asia Pacific the Fastest-Growing Market for Viral Vector & DNA Manufacturing?
Asia Pacific is expected to experience the fastest growth in the coming years due to increasing investments in biotechnology and expanding healthcare infrastructure across the region. Growing awareness and adoption of gene and cell therapies, coupled with a rising number of clinical trials, are driving demand for viral vectors and plasmid DNA. Additionally, supportive government initiatives and favorable regulatory reforms are encouraging local manufacturing and innovation. The presence of a large patient population and cost advantages also attract pharmaceutical companies to expand their operations in this region, fueling rapid market growth.
China is leading the charge in the Asia Pacific viral vector & plasmid DNA manufacturing market due to its significant investments in biotech research and infrastructure. The country has a rapidly growing gene therapy pipeline and a large patient population, which drives strong demand for viral vectors and plasmid DNA. Additionally, favorable government policies and increasing collaborations between local and international companies are accelerating innovation and manufacturing capacity. China’s focus on becoming a global hub for advanced biopharmaceuticals positions it at the forefront of the regional market.
Region-Wise Growth Overview of the Market:
| Region |
Market Size (2024) |
Projected CAGR (2025-2034) |
Key Growth Drives |
Key Challenges |
Market Outlook |
| North America |
USD 2.7 Bn |
~6.57%
|
Strong biotech presence, high R&D investment, supportive regulations, advanced healthcare infrastructure |
High manufacturing costs, complex regulations |
Dominant market with steady, strong growth |
| Asia Pacific |
USD 1.9 Bn |
~7.73%
|
Rising biotech investments, expanding healthcare infrastructure, large patient population, favorable policies |
Regulatory challenges, quality control issues |
Fastest-growing region |
| Europe |
USD 1.5 Bn |
~11.11%
|
Increasing gene therapy approvals, growing clinical trials, government funding |
Lengthy regulatory processes, reimbursement challenges |
Significant growth |
| Latin America |
USD 0.5 Bn |
~6.05%
|
Growing healthcare access, emerging biotech startups, increasing awareness |
Limited infrastructure, regulatory uncertainties |
Moderate growth with emerging opportunities |
| MEA |
USD 0.3 Bn |
~5.24%
|
Developing healthcare infrastructure, government initiatives, rising biotech interest |
Political instability, shortage of skilled workforce |
Gradual growth with potential for future expansion |
Viral Vector & Plasmid DNA Manufacturing Market Value Chain Analysis
1. Research & Development (R&D)
This initial stage involves identifying therapeutic targets, developing vector/plasmid designs, and conducting preclinical studies. It includes design optimization, vector selection (viral or non-viral), and in vitro/in vivo testing for efficacy and safety.
Key Players:
Sangamo Therapeutics, CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics
2. Raw Material Supply
This stage involves the sourcing of high-quality raw materials such as enzymes, cell culture media, plasmid backbones, and reagents critical for plasmid propagation and vector production. The quality and consistency of these materials directly impact the success and scalability of downstream manufacturing.
Key Players:
Thermo Fisher Scientific, Merck KGaA, Cytiva, Danaher Corporation
3. Process Development & Optimization
At this stage, scalable and GMP-compliant manufacturing processes are developed for producing viral vectors or plasmids. This includes upstream (cell culture, transfection) and downstream (harvesting, purification) process optimization to ensure yield, purity, and batch-to-batch consistency.
Key Players:
Lonza, Catalent, Oxford Biomedica, WuXi Advanced Therapies
4. Manufacturing (GMP Production)
This is the core of the value chain, where large-scale production is carried out under Good Manufacturing Practices (GMP). It involves bioreactor-based vector production, purification, and formulation for clinical or commercial supply.
Key Players:
FUJIFILM Diosynth Biotechnologies, Aldevron, Thermo Fisher Scientific, Charles River Laboratories
5. Quality Control & Regulatory Compliance
This stage ensures that vectors and plasmids meet stringent regulatory standards for purity, potency, safety, and identity. QC testing includes sterility, endotoxin levels, vector genome integrity, and compliance with regulatory guidelines like FDA and EMA.
Key Players:
Eurofins Scientific, Charles River Laboratories, SGS, BioReliance (MilliporeSigma)
6. Distribution & End-Use Applications
Finally, the manufactured products are distributed to biopharma companies, hospitals, and research institutions for use in gene therapy, vaccine development, and clinical trials. Timely delivery, cold chain logistics, and global reach are critical at this stage.
Key Players:
Pfizer, Novartis, Gilead Sciences (Kite Pharma), bluebird bio, Moderna
Key Players Operating in the Viral Vector & Plasmid DNA Manufacturing Market
Thermo Fisher is a leading global supplier of plasmid DNA, viral vectors, and GMP-compliant manufacturing services. It offers end-to-end solutions, from raw materials to commercial-scale production, supporting gene therapy developers worldwide.
A major contract development and manufacturing organization (CDMO), Lonza provides comprehensive services for viral vector production including process development, GMP manufacturing, and quality testing. The company supports both early-phase and commercial therapies.
Catalent offers specialized expertise in viral vector manufacturing through its gene therapy division. Its facilities provide GMP-grade adeno-associated virus (AAV), lentivirus, and plasmid DNA manufacturing, with scalable production capacity.
- Aldevron (a Danaher company)
Aldevron is a global leader in the production of research-grade, GMP-grade, and clinical-grade plasmid DNA. It is known for supporting early-stage biotech and major pharma companies with scalable DNA manufacturing.
A pioneer in lentiviral vector manufacturing, Oxford Biomedica partners with major gene therapy companies to deliver clinical and commercial-scale vector supply. It has extensive GMP-compliant facilities and long-standing expertise.
- FUJIFILM Diosynth Biotechnologies
It offers end-to-end viral vector and plasmid DNA manufacturing services, including process development and GMP production. The company is expanding its global capacity to meet rising demand in gene therapy.
- Charles River Laboratories
The company specializes in testing, QC, and analytical services for viral vectors and plasmid DNA. It plays a critical role in ensuring regulatory compliance and product safety throughout the development lifecycle.
- Wuxi Advanced Therapies (WuXi AppTec)
It provides comprehensive CDMO services for gene therapy, including viral vector production, plasmid DNA manufacturing, and analytical testing. WuXi supports global clients with modular manufacturing platforms.
- Vigene Biosciences (now part of Charles River)
The company engages in producing AAV, adenovirus, and lentivirus vectors at research, preclinical, and GMP scales. It supports gene therapy developers with high-throughput and flexible manufacturing options.
A key player in gene therapy, Bluebird Bio manufactures its own viral vectors for clinical and commercial use. It is vertically integrated to control quality and supply chain in-house.
Recent Developments
- In August 2025, Wacker Chemie AG and Estonia-based Gearbox Biosciences have announced a strategic collaboration to co-develop scalable, antibiotic-free technologies for plasmid DNA (pDNA) manufacturing. The partnership combines Wacker’s GMP biologics expertise with Gearbox’s innovative microbial platforms to improve efficiency and cost-effectiveness in producing pDNA for mRNA therapies, gene therapies, and vaccines.
- In April 2025, ProBio launched GMP plasmid DNA manufacturing at its Hopewell, NJ facility, offering clinical-grade production from cell bank to batch release in just three months. Serving both U.S. and European markets, the site integrates process development, analytics, and project management to streamline timelines. The service ensures high-purity plasmids (≥85% supercoiled content) and reduces contamination risk, with GMP-like plasmids delivered in four weeks.
- In June 2024, Bionova Scientific announced a $100 million investment to expand into plasmid DNA production with a new 100,000 sq ft facility in The Woodlands, Texas. Set to begin operations in Q1 2025, the site will offer pDNA development and production, with GMP manufacturing expected by early 2026.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2034. For this study, Nova one advisor, Inc. has segmented the viral vectors & plasmid DNA manufacturing market.
By Vector Type
- Adenovirus
- Plasmid DNA
- Lentivirus
- Retrovirus
- AAV
- Others
By Application
- Gene Therapy
- Antisense &RNAi
- Cell Therapy
- Vaccinology
By Workflow
- Upstream Processing
- Vector Recovery/Harvesting
- Vector Amplification & Expansion
- Downstream Processing
- Fill-finish
- Purification
By End-User
- Biopharmaceutical and Pharmaceutical Companies
- Research Institutes
By Disease
- Genetic Disorders
- Cancer
- Infectious Diseases
- Others
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)
List of Tables
- Global Viral Vectors & Plasmid DNA Manufacturing Market Size (USD Billion), 2024–2034
- Global Market Share by Vector Type, 2024 & 2034
- Global Market Share by Application, 2024 & 2034
- Global Market Share by Workflow, 2024 & 2034
- Global Market Share by End-User, 2024 & 2034
- Global Market Share by Disease, 2024 & 2034
- North America Market Size, by Country, 2024–2034
- U.S. Market Size, by Vector Type, 2024–2034
- U.S. Market Size, by Application, 2024–2034
- U.S. Market Size, by Workflow, 2024–2034
- U.S. Market Size, by End-User, 2024–2034
- U.S. Market Size, by Disease, 2024–2034
- Canada Market Size, by Vector Type, 2024–2034
- Mexico Market Size, by Vector Type, 2024–2034
- North America Market Size, by Application, 2024–2034
- North America Market Size, by Workflow, 2024–2034
- North America Market Size, by End-User, 2024–2034
- North America Market Size, by Disease, 2024–2034
- Europe Market Size, by Country, 2024–2034
- Germany Market Size, by Vector Type, 2024–2034
- France Market Size, by Vector Type, 2024–2034
- U.K. Market Size, by Vector Type, 2024–2034
- Italy Market Size, by Vector Type, 2024–2034
- Rest of Europe Market Size, by Vector Type, 2024–2034
- Europe Market Size, by Application, 2024–2034
- Europe Market Size, by Workflow, 2024–2034
- Europe Market Size, by End-User, 2024–2034
- Europe Market Size, by Disease, 2024–2034
- Asia Pacific Market Size, by Country, 2024–2034
- China Market Size, by Vector Type, 2024–2034
- Japan Market Size, by Vector Type, 2024–2034
- South Korea Market Size, by Vector Type, 2024–2034
- India Market Size, by Vector Type, 2024–2034
- Southeast Asia Market Size, by Vector Type, 2024–2034
- Rest of Asia Pacific Market Size, by Vector Type, 2024–2034
- Asia Pacific Market Size, by Application, 2024–2034
- Asia Pacific Market Size, by Workflow, 2024–2034
- Asia Pacific Market Size, by End-User, 2024–2034
- Asia Pacific Market Size, by Disease, 2024–2034
- Latin America Market Size, by Country, 2024–2034
- Brazil Market Size, by Vector Type, 2024–2034
- Rest of Latin America Market Size, by Vector Type, 2024–2034
- Latin America Market Size, by Application, 2024–2034
- Latin America Market Size, by Workflow, 2024–2034
- Latin America Market Size, by End-User, 2024–2034
- Latin America Market Size, by Disease, 2024–2034
- Middle East & Africa Market Size, by Country, 2024–2034
- Turkey Market Size, by Vector Type, 2024–2034
- GCC Countries Market Size, by Vector Type, 2024–2034
- Africa Market Size, by Vector Type, 2024–2034
- Rest of Middle East & Africa Market Size, by Vector Type, 2024–2034
- Middle East & Africa Market Size, by Application, 2024–2034
- Middle East & Africa Market Size, by Workflow, 2024–2034
- Middle East & Africa Market Size, by End-User, 2024–2034
- Middle East & Africa Market Size, by Disease, 2024–2034
- Global Market Outlook, 2024–2034 (USD Billion)
- Global Market Share, by Vector Type, 2024
- Global Market Share, by Application, 2024
- Global Market Share, by Workflow, 2024
- Global Market Share, by End-User, 2024
- Global Market Share, by Disease, 2024
- Global Market Share, by Vector Type, 2034
- Global Market Share, by Application, 2034
- Global Market Share, by Workflow, 2034
- Global Market Share, by End-User, 2034
- Global Market Share, by Disease, 2034
- North America Market Share, by Country, 2024
- U.S. Market Share, by Vector Type, 2024
- Europe Market Share, by Country, 2024
- Germany Market Share, by Vector Type, 2024
- Asia Pacific Market Share, by Country, 2024
- China Market Share, by Vector Type, 2024
- Latin America Market Share, by Country, 2024
- Brazil Market Share, by Vector Type, 2024
- Middle East & Africa Market Share, by Country, 2024
- GCC Countries Market Share, by Vector Type, 2024
- Comparative Growth Rate of Viral Vectors & Plasmid DNA Manufacturing Market across Regions, 2024–2034


