RNA Therapeutics Market Size and Forecast 2026 to 2035
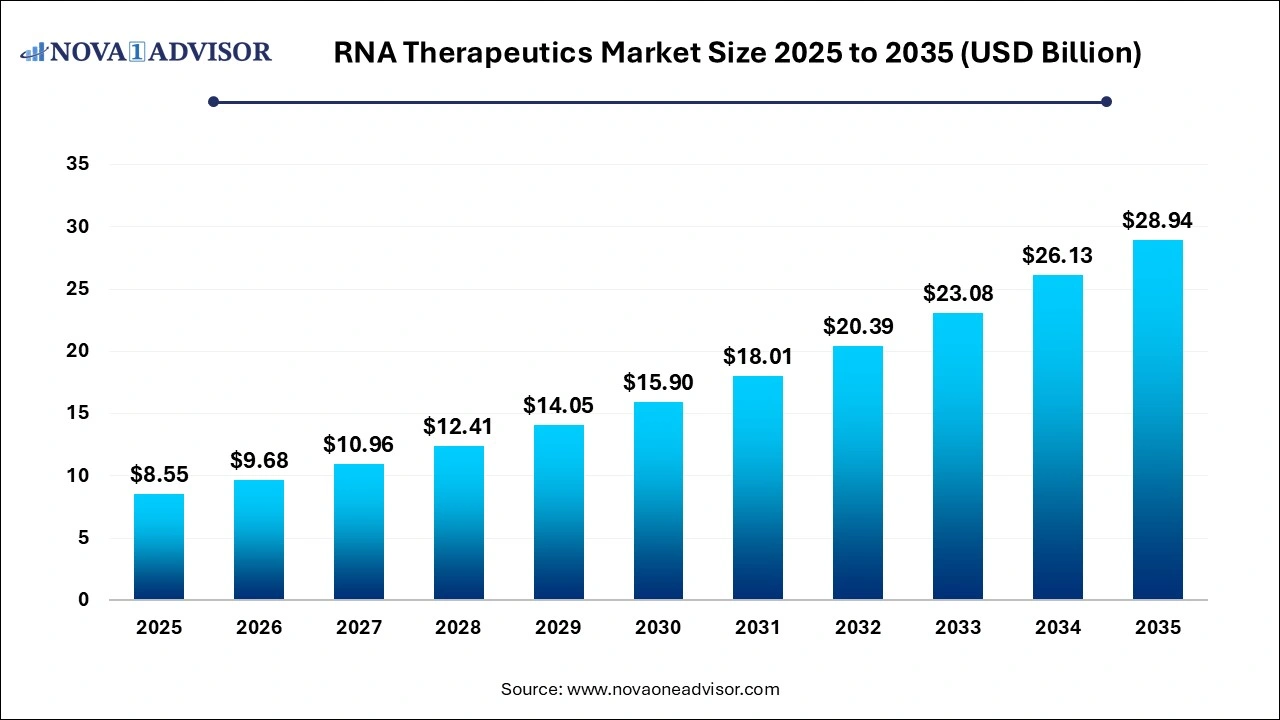
The global RNA therapeutics market was valued at USD 8.55 billion in 2025 and is projected to hit around USD 28.94 billion by 2035, growing at a CAGR of 12.97% during the forecast period 2026 to 2035. The growth of the market is attributed to the rising prevalence of chronic diseases, innovations in RNA technologies, and increasing investments in R&D.
RNA Therapeutics Market Key Takeaways
- North America dominated the RNA therapeutics market with the largest share in 2025.
- Asia Pacific is expected to grow at the fastest CAGR during the forecast period of 2026 and 2035.
- By type, the mRNA therapeutics segment led the market in 2025.
- By type, the RNA interference (RNAi) therapeutics segment is expected to expand at the highest CAGR in the coming years.
- By product, the vaccines segment held the largest share of the market in 2025.
- By product, the drugs segment is likely to grow at a significant rate over the forecast period.
- By indication, the infectious diseases segment dominated the market in 2025.
- By indication, the rare genetic disorders/hereditary diseases segment is expected to expand at a rapid pace in the upcoming period.
- By end-user, the hospitals & clinics segment held the largest market share in 2025.
- By end-user, the research institutes segment is expected to grow at the fastest rate during the projection period.
Impact of AI on the RNA Therapeutics Market
AI is significantly transforming the RNA therapeutics market by accelerating drug discovery and development processes. It enables precise design and optimization of RNA molecules, improving efficacy and reducing off-target effects. AI-driven data analysis helps identify novel therapeutic targets and predict patient responses, enhancing personalized medicine approaches. Additionally, AI streamlines manufacturing by optimizing production workflows, increasing scalability and reducing costs. Overall, AI adoption is boosting innovation and speeding up the delivery of RNA-based treatments to patients.
- In August 2025, MIT researchers used AI to design nanoparticles that improve RNA vaccine and therapy delivery. By training a machine-learning model on thousands of existing particles, they predicted more effective materials and identified particles suited for different cell types, enabling the integration of new materials into delivery systems.
RNA Therapeutics Market Overview
The RNA therapeutics market focuses on developing treatments that use RNA molecules to target and modulate gene expression, offering a promising approach for various diseases including genetic disorders, infectious diseases, and cancer. RNA therapeutics provide advantages such as high specificity, the ability to target previously “undruggable” genes, and faster development times compared to traditional drugs. Their versatility enables personalized medicine and novel treatment options with potentially fewer side effects. Key growth drivers include advancements in RNA delivery technologies, increasing approvals of RNA-based therapies, and rising investments in research and development. Additionally, growing awareness of RNA therapeutics' potential and expanding applications across multiple disease areas are fueling market expansion.
What are the Major Trends in the RNA Therapeutics Market?
- Expansion of mRNA Vaccine Technologies: The success of mRNA vaccines, especially during the COVID-19 pandemic, has accelerated research and development in mRNA-based therapeutics for infectious diseases, cancer, and rare genetic disorders.
- Advancements in RNA Delivery Systems: Improved delivery methods such as lipid nanoparticles (LNPs) and novel vectors are enhancing the stability, efficiency, and targeting of RNA therapeutics, overcoming one of the key challenges in the field.
- Growth of RNA Interference (RNAi) Therapies: RNAi therapeutics are gaining traction due to their ability to silence specific genes, offering promising treatment options for genetic and rare diseases, which is driving significant investment and clinical pipeline growth.
Report Scope of RNA Therapeutics Market
| Report Coverage |
Details |
| Market Size in 2026 |
USD 9.68 Billion |
| Market Size by 2035 |
USD 28.94 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 12.97% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Type, By Product, By Indication, By End-User |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
RNA Therapeutics Market Dynamics
Drivers
Increasing Approvals of RNA Therapies
Increasing approvals of RNA therapies significantly drive the growth of the RNA therapeutics market by validating the safety and efficacy of these novel treatments. Regulatory greenlights encourage more investment and research, accelerating the development of new RNA-based drugs. Approved therapies also boost adoption among healthcare providers and patients. Additionally, successful approvals expand the range of treatable diseases, from rare genetic disorders to infectious diseases, broadening market potential. This momentum fosters innovation and partnerships across the biotech and pharmaceutical sectors, further propelling market expansion.
Approved RNA Therapies as of Q3 2024
|
Product Name
|
Generic Name |
Year First Approved
|
Disease(s) |
Locations Approved |
Originator Company |
|
CSPC Pharmaceutical COVID-19 vaccine
|
COVID-19 vaccine, CSPC Pharmaceutical
|
2023 |
Infection, coronavirus, novel coronavirus prophylaxis |
China |
CSPC Pharmaceutical |
|
Sinocelltech COVID-19 vaccine
|
COVID-19 alpha/beta/delta/Omicron variants S-trimer quadrivalent recombinant protein vaccine
|
2023 |
Infection, coronavirus, novel coronavirus prophylaxis |
China, UAE, U.S. |
Sinocelltech |
| Izervay |
avacincaptad pegol sodium |
2023 |
Wet age-related macular degeneration |
U.S. |
Archemix |
| Qalsody |
tofersen |
2023 |
Amyotrophic lateral sclerosis |
U.S., EU |
Ionis Pharmaceuticals |
| ARCT-154 |
COVID-19 mRNA vaccine, Arcturus |
2023 |
Infection, coronavirus, novel coronavirus prophylaxis |
Japan |
Arcturus Therapeutics |
| Daichirona |
COVID-19 vaccine, Daiichi Sankyo |
2023 |
Infection, coronavirus, novel coronavirus prophylaxis J |
Japan |
Daiichi Sankyo |
| Wainua |
eplontersen |
2023 |
Transthyretin-related hereditary amyloidosis |
U.S., Canada |
Ionis Pharmaceuticals |
| Rivfloza |
nedosiran |
2023 |
Hyperoxaluria |
U.S. |
Dicerna Pharmaceuticals |
| SYS-6006.32 |
Bivalent COVID-19 mRNA vaccine, CSPC Pharmaceutical
|
2023 |
Infection, coronavirus, novel coronavirus prophylaxis |
China |
CSPC Pharmaceutical |
| RQ-3033 |
COVID-19 mRNA vaccine, Walvax Biotechnology
|
2023 |
Infection, coronavirus, novel coronavirus prophylaxis |
China |
Walvax Biotechnology |
| Rytelo |
imetelstat
|
2024 |
Myelodysplastic syndrome |
U.S. |
Geron |
| mRESVIA |
respiratory syncytial virus vaccine, Moderna Therapeutics
|
2024 |
Respiratory syncytial virus prophylaxis |
U.S., EU |
Moderna Therapeutics |
Rising Prevalence of Genetic and Chronic Diseases
The rising prevalence of genetic and chronic diseases drives the growth of the RNA therapeutics market by increasing the demand for innovative and targeted treatment options. RNA therapeutics offer the ability to specifically target the genetic basis of these diseases, providing more effective and personalized therapies compared to traditional treatments. As conditions like cancer, rare genetic disorders, and autoimmune diseases continue to affect large populations worldwide, there is a growing need for advanced therapies that can address underlying molecular causes. This demand encourages ongoing research, development, and adoption of RNA-based drugs, fueling market expansion. Additionally, improved diagnosis and awareness of these diseases further accelerate the adoption of RNA therapeutics.
Restraints
Manufacturing Complexities and High Development Costs
The production of RNA-based drugs requires specialized facilities, stringent quality control, and advanced technologies, making the process time-consuming and expensive. These complexities increase the risk of manufacturing errors and delays, limiting scalability and accessibility. Additionally, the high cost of research, development, and regulatory approval poses financial challenges, especially for smaller companies. As a result, these factors hinder widespread adoption and slow down market expansion despite the growing demand.
Stability Challenges and Regulatory Hurdles
RNA molecules are inherently unstable and prone to degradation, which complicates formulation and storage, impacting drug efficacy. Additionally, RNA therapies can trigger unintended immune responses, leading to safety concerns that require careful management. Strict and evolving regulatory requirements for approval add further complexity, prolonging development timelines and increasing costs. These challenges create barriers to entry and limit the rapid commercialization of new RNA-based treatments.
Opportunities
Development of Improved Delivery Systems
The development of improved delivery systems creates significant opportunities in the RNA therapeutics market by enhancing the stability, targeting, and efficiency of RNA-based drugs. Advanced delivery technologies, such as lipid nanoparticles and novel carriers, help protect RNA molecules from degradation and facilitate precise delivery to specific cells or tissues, increasing therapeutic effectiveness. These innovations also reduce potential side effects and immune reactions, improving patient safety and compliance. As a result, improved delivery systems enable the expansion of RNA therapies to treat a wider range of diseases, propelling market growth and attracting increased investment in research and development.
Rising demand for Personalized Medicine
The rising demand for personalized medicine creates vast opportunities in the RNA therapeutics market by enabling treatments tailored to an individual’s genetic makeup. RNA-based therapies, such as mRNA and RNA interference, can be customized to target specific mutations or disease pathways, offering more precise and effective treatment options. This personalized approach improves patient outcomes and reduces adverse effects, driving greater adoption of RNA therapeutics. Additionally, advances in genomics and bioinformatics are accelerating the development of personalized RNA drugs, attracting significant investment and fueling market growth.
RNA Therapeutics Market Segment Insight
By Type Insight
Why Did the mRNA Therapeutics Segment Lead the RNA Therapeutics Market in 2025?
The mRNA therapeutics segment led the market in 2024 due to the unprecedented success of mRNA-based COVID-19 vaccines, which demonstrated the mRNA’s effectiveness and scalability on a global scale. This breakthrough boosted investor confidence and accelerated research into mRNA applications beyond infectious diseases, including cancer and genetic disorders. Additionally, advances in delivery technologies, such as lipid nanoparticles, improved the stability and efficiency of mRNA therapies. The versatility of mRNA in rapidly developing targeted treatments further solidified its dominant position in the market.
The RNA interference (RNAi) therapeutics segment is expected to grow at the fastest CAGR during the forecast period, owing to its ability to selectively silence disease-causing genes, offering targeted treatment for a variety of genetic and chronic conditions. Advances in delivery technologies have improved RNAi stability and reduced off-target effects, enhancing its clinical potential. Increasing approvals of RNAi-based drugs and ongoing clinical trials are fueling investor and industry interest. Additionally, growing research into expanding RNAi applications beyond rare diseases to more common conditions support strong future growth in this segment.
By Product Insight
How Does the Vaccines Segment Dominate the RNA Therapeutics Market in 2025?
The vaccines segment dominated the market while holding the largest share in 2025. This is primarily due to the widespread success and adoption of mRNA vaccines during the COVID-19 pandemic. These vaccines showcased the rapid development, high efficacy, and safety of RNA-based technologies, leading to increased global demand and acceptance. Additionally, ongoing research is expanding RNA vaccine applications to other infectious diseases, boosting market growth. The ability to quickly adapt RNA vaccines to emerging variants and pathogens further strengthens this segment’s market dominance.
The drugs segment is expected to grow at a significant rate in the upcoming period due to the expanding pipeline of RNA-based treatments targeting a wide range of diseases, including genetic disorders, cancers, and autoimmune conditions. Advances in delivery systems and molecular design are improving the efficacy and safety of RNA drugs, encouraging broader clinical adoption. Additionally, increased investment from pharmaceutical companies and growing collaborations with biotech firms are accelerating drug development. The versatility of RNA therapeutics to address complex diseases with unmet medical needs further drives strong growth in this segment.
By Indication Insight
What Made Infectious Diseases the Dominant Segment in the market in 2025?
The infectious diseases segment dominated the RNA therapeutics market in 2025 due to the widespread success of mRNA vaccines developed for COVID-19, which validated RNA technology as a powerful tool for rapid vaccine development. The urgent global need for effective infectious disease control accelerated investment and research in RNA-based vaccines and therapeutics. Moreover, RNA platforms allow for quick adaptation to emerging pathogens and variants, making them ideal for managing infectious outbreaks. This adaptability, combined with growing government and public health support, solidified infectious diseases as the leading segment in the market.
The rare genetic disorders/hereditary diseases segment is expected to expand at a rapid pace over the projection period. Advances in RNA technology, such as antisense oligonucleotides and RNA interference, offer precise gene modulation, making them ideal for correcting genetic mutations. The growing awareness and improved diagnostic capabilities are driving early detection and treatment demand. Supportive regulatory frameworks and orphan drug incentives also encourage the development of RNA therapies for rare diseases, fueling the growth of this segment. Moreover, the growth of the segment is attributed to the increasing development of novel RNA-based therapies targeting these previously untreatable conditions.
- In August 2025, Ionis Pharmaceuticals received FDA approval for DAWNZERA™ (donidalorsen), the first and only RNA-targeted therapy for preventing hereditary angioedema (HAE) attacks in patients aged 12 and older. DAWNZERA targets plasma prekallikrein (PKK) and is self-administered via autoinjector every 4 or 8 weeks.
By End-User Insight
Why Did the Hospitals & Clinics Contribute the Largest Share of the RNA Therapeutics Market in 2025?
The hospitals & clinics segment dominated the market with the largest share in 2024 due to the widespread administration of RNA-based vaccines and therapies in clinical settings worldwide. These healthcare facilities serve as primary points for delivering cutting-edge treatments, including mRNA vaccines for COVID-19 and RNA drugs for various diseases. Increased patient awareness and demand for innovative, personalized treatments further boosted the segment’s growth. Additionally, hospitals and clinics benefit from established infrastructure and trained personnel, enabling efficient deployment and monitoring of RNA therapeutics.
The research institutes segment is expected to grow at the fastest CAGR during the forecast period due to increasing investments in RNA technology and expanding research activities focused on developing novel RNA-based treatments. These institutes play a critical role in early-stage discovery and innovation, driving breakthroughs in RNA interference, mRNA therapies, and antisense oligonucleotides. Additionally, growing collaborations between academia, biotech firms, and pharmaceutical companies are accelerating the translation of research into clinical applications. The rising focus on personalized medicine and rare genetic disorders further fuels research demand, positioning research institutes as key growth drivers in the market.
Regional Insights
What Made North America the Dominant Region in the RNA Therapeutics Market?
North America dominated the RNA therapeutics market while holding the largest share in 2025. This is primarily due to its robust biotechnology and pharmaceutical infrastructure, which supports extensive research, development, and commercialization of RNA-based therapies. The region benefits from significant government funding, strong intellectual property protection, and a high concentration of leading biotech companies and research institutes. Early adoption of innovative RNA technologies, especially during the COVID-19 pandemic with mRNA vaccines, further propelled market growth. Additionally, favorable regulatory frameworks and a large patient population with access to advanced healthcare contribute to North America’s leadership in this market.
The U.S. is a major contributor to the North America RNA therapeutics market, driven by its strong biotech and pharmaceutical sectors. The country hosts numerous leading companies pioneering RNA-based therapies and vaccines, supported by substantial government funding and advanced research infrastructure. Additionally, the U.S. benefits from a well-established regulatory environment and early adoption of innovative technologies, fueling market growth.
- As of January 2024, the U.S. has 21 FDA-approved RNA-based therapies and at least 131 more in clinical trials, with RNAi and mRNA making up 77% of the pipeline. These therapies span 15 therapeutic areas, including rare diseases, and offer the potential for infrequent dosing, such as once or twice a year. While 80% of candidates are in early-stage trials, 29 are in late-stage development, signaling strong potential for upcoming approvals.
What Makes Asia Pacific the Fastest-Growing Market for RNA Therapeutics?
Asia Pacific is expected to experience the fastest growth in the market during the projection period due to increasing investments in biotechnology and pharmaceutical research across countries like China, Japan, and South Korea. The region benefits from a large patient population, growing healthcare infrastructure, and rising awareness of advanced therapies. Additionally, government initiatives and favorable regulatory reforms are encouraging innovation and local manufacturing capabilities. The expanding presence of global and regional biotech companies further accelerates market development, making Asia Pacific a rapidly emerging hub for RNA therapeutics.
China is leading the charge in the Asia Pacific RNA therapeutics market due to its substantial investments in biotechnology and strong government support for innovation in life sciences. The country has rapidly expanded its research infrastructure and fostered collaborations between academic institutions, biotech firms, and pharmaceutical companies. Additionally, China’s large patient population and growing healthcare access create significant demand for advanced RNA-based therapies. Regulatory reforms aimed at accelerating drug approvals further boost the development and commercialization of RNA therapeutics.
Region-Wise Growth Overview of the RNA Therapeutics Market:
| Region |
Market Size (2024) |
Projected CAGR (2025-2034) |
Key Growth Drives |
Key Challenges |
Market Outlook |
| North America |
USD 3.1 Bn |
~6.65% |
Advanced R&D capabilities, robust biotech infrastructure, strong funding support |
High development costs, stringent regulatory requirements |
Dominant and steadily growing market |
| Asia Pacific |
USD 2.2 Bn |
~7.89% |
Rising healthcare investments, large patient base, supportive government policies |
Infrastructure gaps, price sensitivity in some markets |
Fastest-growing and rapidly emerging region |
| Europe |
USD 1.8 Bn |
~10.76% |
Healthcare innovation, collaborative R&D, regulatory facilitation (e.g., EMA) |
Slow regulatory pathways, reimbursement constraints |
Stable growth |
| Latin America |
USD 0.6 Bn |
~5.24% |
Expanding healthcare infrastructure, growing demand for advanced therapies |
Limited funding, regulatory complexity |
Emerging market with strong growth potential |
| MEA |
USD 0.4 Bn |
~4.14% |
Government-backed healthcare projects, increasing biotech interest |
Infrastructure deficiencies, skilled workforce shortages |
Underpenetrated but promising growth |
RNA Therapeutics Market Value Chain Analysis
1. Research & Development (R&D)
This stage involves discovery, design, and optimization of RNA-based molecules such as mRNA, siRNA, and antisense oligonucleotides. Companies invest heavily in preclinical studies, gene target validation, and RNA molecule engineering to improve efficacy, stability, and delivery. Innovations at this level are critical to building a robust clinical pipeline.
- Key Players: Moderna, Alnylam Pharmaceuticals, Ionis Pharmaceuticals, and BioNTech
2. Manufacturing (Raw Materials & RNA Synthesis)
This stage includes the production of high-quality raw materials such as nucleotides, enzymes, plasmids, and lipids, as well as RNA synthesis under GMP conditions. Manufacturers ensure the RNA molecules are synthesized with high purity, and many also offer lipid nanoparticle (LNP) formulations for drug delivery. Quality control and scalability are vital at this stage.
- Key Players: Thermo Fisher Scientific, Danaher (Cytiva), TriLink BioTechnologies, and Wacker Chemie AG
3. Formulation & Drug Delivery
Formulation focuses on delivering RNA molecules effectively into target cells, often using lipid nanoparticles (LNPs), polymers, or viral vectors. Delivery technology significantly impacts therapeutic outcomes, making this a crucial and innovation-driven segment. Companies in this stage work to optimize stability, targeting, and safety profiles of RNA drugs.
- Key Players: Arcturus Therapeutics, Precision NanoSystems (acquired by Danaher), and CordenPharma
4. Clinical Trials & Regulatory Approval
Once a drug candidate passes preclinical studies, it enters human clinical trials (Phases I–III) followed by regulatory review. This phase involves patient recruitment, data collection, and compliance with regulatory standards set by agencies like the FDA and EMA. Late-stage approvals are often a major turning point in commercial success.
- Key Players: Moderna, Alnylam Pharmaceuticals, Ionis Pharmaceuticals, and CureVac
5. Commercialization & Distribution
After regulatory approval, products are scaled up for commercialization. This stage includes product launch, marketing, distribution, and post-market surveillance. Partnerships between biotech innovators and pharmaceutical giants help in expanding global access and infrastructure support for RNA therapies.
- Key Players: Pfizer, Moderna, BioNTech, and Novartis
6. End-Use (Hospitals, Clinics, Research Institutes)
Hospitals, clinics, and research institutions serve as primary points for RNA therapeutic administration and continued study. These stakeholders play a vital role in patient access, clinical feedback, and long-term safety monitoring, helping refine existing treatments and support further innovation.
- Key Stakeholders: Mayo Clinic, Cleveland Clinic, NIH, and Academic Research Centers
Key Players Competing in the RNA Therapeutics Market
A global leader in mRNA technology, Moderna played a pivotal role in commercializing mRNA vaccines, particularly for COVID-19. The company continues to expand its RNA pipeline targeting infectious diseases, cancer, and rare conditions.
A pioneer in RNA interference (RNAi) therapeutics, Alnylam has multiple FDA-approved RNAi drugs (e.g., Onpattro, Givlaari). It focuses on rare genetic, cardio-metabolic, and hepatic diseases.
The company specializes in antisense oligonucleotide (ASO) therapies and has a robust RNA drug pipeline. Ionis has partnered with companies like Biogen and AstraZeneca for broader therapeutic reach.
Known for its co-development of the first approved mRNA COVID-19 vaccine with Pfizer, BioNTech is expanding its mRNA platform into oncology and other infectious diseases.
It focuses on developing mRNA-based vaccines and therapeutics, including treatments for COVID-19, influenza, and cancer. CureVac is also investing in next-gen mRNA technology for better stability and efficacy.
- Arrowhead Pharmaceuticals
It develops RNAi-based drugs primarily for liver-targeted diseases. Arrowhead has partnerships with major pharma players like Janssen and Takeda.
Sirnaomics specializes in RNAi therapeutics with a focus on oncology and fibrotic diseases. The company is advancing a proprietary delivery platform to enhance therapeutic efficiency.
The company is a leader in RNAi therapeutics, targeting diseases with high unmet need, particularly in hematology and cardiovascular disorders. The company uses GalNAc-conjugation for targeted delivery to the liver.
- Translate Bio (Acquired by Sanofi)
It brought expertise in mRNA-based therapies for infectious and rare diseases. Now part of Sanofi’s growing RNA therapeutics capabilities.
It develops mRNA vaccines and therapeutics using its proprietary LUNAR® delivery platform. Arcturus is also advancing self-amplifying mRNA (saRNA) technology to lower dosing needs.
Recent Developments
- In January 2025, Inverna Therapeutics, a Danish biotech co-founded by the University of Southern Denmark and Argobio, has launched as a pioneering RNA therapeutics company focused on transforming outcomes in severe genetic diseases, starting with Huntington’s disease.
- In September 2024, Aurora Biosynthetics, an advanced RNA therapeutics manufacturer, officially launched to meet rising demand for RNA therapeutics across Asia-Pacific. The company offers end-to-end GMP production of plasmid DNA, mRNA, and lipid nanoparticles, marking a new era in RNA therapy manufacturing.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the RNA therapeutics market.
By Type
- RNA Interference (RNAi) Therapeutics
- mRNA Therapeutics
- Antisense Oligonucleotide (ASO) Therapeutics
- Others
By Product
By Indication
- Rare Genetic Disorders/Hereditary Diseases
- Infectious Diseases
- Others (Metabolic Disorders, Auto-immune Disorders, and Oncology)
By End-User
- Research Institutes
- Hospitals & Clinics
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa (MEA)
List of Tables
- Global RNA Therapeutics Market Size (USD Billion), 2024–2034
- Global RNA Therapeutics Market Share by Type, 2024 & 2034
- Global RNA Therapeutics Market Share by Product, 2024 & 2034
- Global RNA Therapeutics Market Share by Indication, 2024 & 2034
- Global RNA Therapeutics Market Share by End-User, 2024 & 2034
- North America RNA Therapeutics Market Size, by Country, 2024–2034
- U.S. RNA Therapeutics Market Size, by Type, 2024–2034
- U.S. RNA Therapeutics Market Size, by Product, 2024–2034
- U.S. RNA Therapeutics Market Size, by Indication, 2024–2034
- U.S. RNA Therapeutics Market Size, by End-User, 2024–2034
- Canada RNA Therapeutics Market Size, by Type, 2024–2034
- Mexico RNA Therapeutics Market Size, by Type, 2024–2034
- North America RNA Therapeutics Market Size, by Product, 2024–2034
- North America RNA Therapeutics Market Size, by Indication, 2024–2034
- North America RNA Therapeutics Market Size, by End-User, 2024–2034
- Europe RNA Therapeutics Market Size, by Country, 2024–2034
- Germany RNA Therapeutics Market Size, by Type, 2024–2034
- France RNA Therapeutics Market Size, by Type, 2024–2034
- U.K. RNA Therapeutics Market Size, by Type, 2024–2034
- Italy RNA Therapeutics Market Size, by Type, 2024–2034
- Rest of Europe RNA Therapeutics Market Size, by Type, 2024–2034
- Europe RNA Therapeutics Market Size, by Product, 2024–2034
- Europe RNA Therapeutics Market Size, by Indication, 2024–2034
- Europe RNA Therapeutics Market Size, by End-User, 2024–2034
- Asia Pacific RNA Therapeutics Market Size, by Country, 2024–2034
- China RNA Therapeutics Market Size, by Type, 2024–2034
- Japan RNA Therapeutics Market Size, by Type, 2024–2034
- South Korea RNA Therapeutics Market Size, by Type, 2024–2034
- India RNA Therapeutics Market Size, by Type, 2024–2034
- Southeast Asia RNA Therapeutics Market Size, by Type, 2024–2034
- Rest of Asia Pacific RNA Therapeutics Market Size, by Type, 2024–2034
- Asia Pacific RNA Therapeutics Market Size, by Product, 2024–2034
- Asia Pacific RNA Therapeutics Market Size, by Indication, 2024–2034
- Asia Pacific RNA Therapeutics Market Size, by End-User, 2024–2034
- Latin America RNA Therapeutics Market Size, by Country, 2024–2034
- Brazil RNA Therapeutics Market Size, by Type, 2024–2034
- Rest of Latin America RNA Therapeutics Market Size, by Type, 2024–2034
- Latin America RNA Therapeutics Market Size, by Product, 2024–2034
- Latin America RNA Therapeutics Market Size, by Indication, 2024–2034
- Latin America RNA Therapeutics Market Size, by End-User, 2024–2034
- Middle East & Africa RNA Therapeutics Market Size, by Country, 2024–2034
- Turkey RNA Therapeutics Market Size, by Type, 2024–2034
- GCC Countries RNA Therapeutics Market Size, by Type, 2024–2034
- Africa RNA Therapeutics Market Size, by Type, 2024–2034
- Rest of Middle East & Africa RNA Therapeutics Market Size, by Type, 2024–2034
- Middle East & Africa RNA Therapeutics Market Size, by Product, 2024–2034
- Middle East & Africa RNA Therapeutics Market Size, by Indication, 2024–2034
- Middle East & Africa RNA Therapeutics Market Size, by End-User, 2024–2034
- Global RNA Therapeutics Market Outlook, 2024–2034 (USD Billion)
- Global RNA Therapeutics Market Share, by Type, 2024
- Global RNA Therapeutics Market Share, by Product, 2024
- Global RNA Therapeutics Market Share, by Indication, 2024
- Global RNA Therapeutics Market Share, by End-User, 2024
- Global RNA Therapeutics Market Share, by Type, 2034
- Global RNA Therapeutics Market Share, by Product, 2034
- Global RNA Therapeutics Market Share, by Indication, 2034
- Global RNA Therapeutics Market Share, by End-User, 2034
- North America RNA Therapeutics Market Share, by Country, 2024
- U.S. RNA Therapeutics Market Share, by Type, 2024
- Europe RNA Therapeutics Market Share, by Country, 2024
- Germany RNA Therapeutics Market Share, by Type, 2024
- Asia Pacific RNA Therapeutics Market Share, by Country, 2024
- China RNA Therapeutics Market Share, by Type, 2024
- Latin America RNA Therapeutics Market Share, by Country, 2024
- Brazil RNA Therapeutics Market Share, by Type, 2024
- Middle East & Africa RNA Therapeutics Market Share, by Country, 2024
- GCC Countries RNA Therapeutics Market Share, by Type, 2024
- Comparative Growth Rate of RNA Therapeutics Market across Regions, 2024–2034


