Viral Vectors And Plasmid DNA Manufacturing Market Size, Share and Trends Analysis 2026 to 2035
The global viral vectors and plasmid DNA manufacturing market size was exhibited at USD 7.71 billion in 2025 and is projected to hit around USD 46.81 billion by 2035, growing at a CAGR of 19.76% during the forecast period 2026 to 2035.
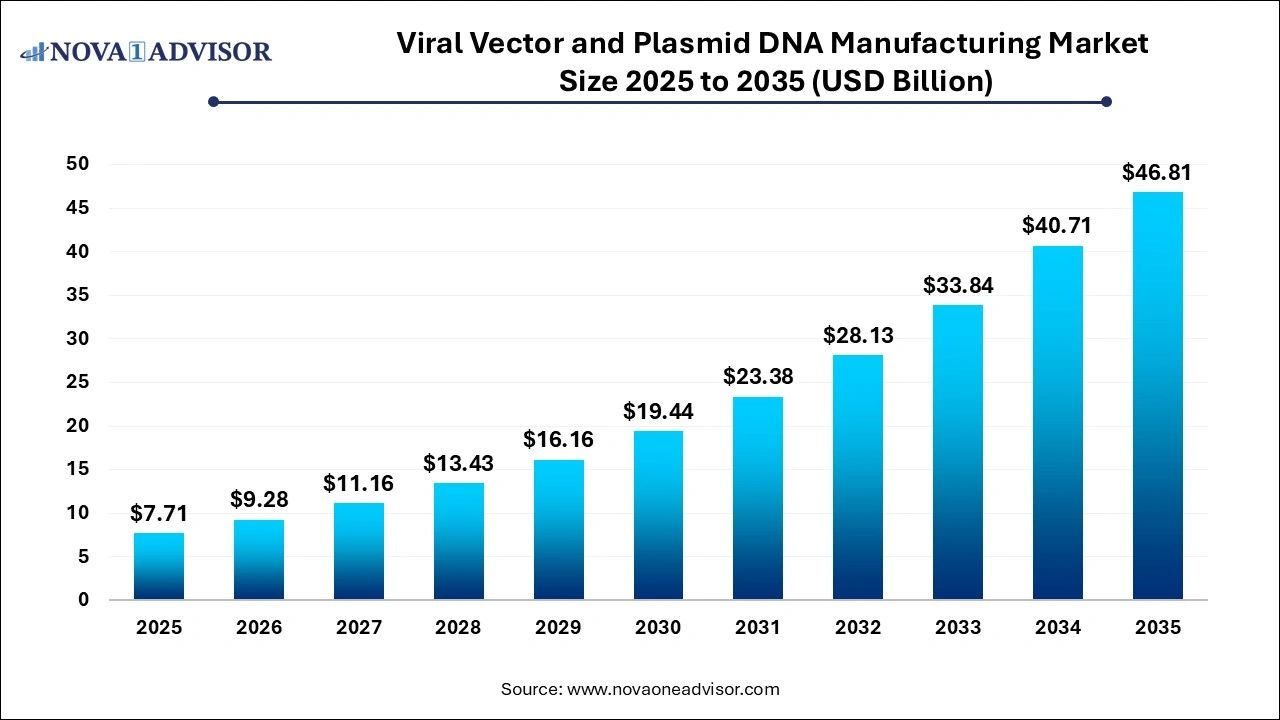
Key Pointers:
- The Adeno-associated virus (AAV) segment held the highest market share of 19.26% in 2025.
- The downstream processing segment held the highest market share of 54.31% in 2025.
- The vaccinology segment held the highest market share of 25.29% in 2022.
- The research institutes segment held the highest market share of 57.42% in 2025.
- The cancer segment held the highest market share of 38.32% in 2025.
- North America dominated the global market with a share of 49.79% in 2025.
Viral Vectors And Plasmid DNA Manufacturing Market Report Scope
|
Report Coverage
|
Details
|
|
Market Size in 2023
|
USD 3.8 Billion
|
|
Market Size by 2032
|
USD 13.09 Billion
|
|
Growth Rate from 2023 to 2032
|
CAGR of 14.72%
|
|
Base year
|
2022
|
|
Forecast period
|
2023 to 2032
|
|
Segments covered
|
Vector type, workflow, application, end-use, disease, product type, region
|
|
Regional scope
|
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa
|
Viral vectors are tools for delivering genetic material into cells. Viruses have developed specific systems for transporting their DNA inside the cells they infect. Furthermore, retrovirus, adenovirus, lentivirus, herpes simplex virus, and others are among the viral vectors which can be employed to transfer genetic material into the genetic composition of cells.
The viral vectors and plasmid DNA manufacturing market saw a lucrative opportunity due to the COVID-19 pandemic. Furthermore, the market players are shifting their business focus toward the development of viral vectors for the SARS-CoV-2 vaccine. Viral vectors are commonly employed tools used in the discovery and development of vaccines against SARS-CoV-2. The ongoing COVID-19 pandemic has encouraged investment in this space in search of a vaccine as viral vector-based vaccines can be manufactured and designed relatively quickly with the use of the same building blocks. For instance, Companies such as Johnson and Johnson/Janssen (J&J); AstraZeneca/University of Oxford; Gamaleya Research Institute; and CanSino Biologics have developed viral vector-based vaccines.
The market for viral vectors and plasmid DNA manufacturing is growing due to the increasing prevalence of target ailments and diseases and the efficacy of viral vectors in gene therapy delivery. This increase is aided by continued research into viral vector-based cell and gene therapies, as well as financing for gene therapy advancement. In addition, an increase in the number of gene therapy-based discovery programs initiated by biotechnology and pharmaceutical companies is expected to drive the demand for scalable production of gene therapy vectors.
A rising number of patients opting for gene therapy is driving the global market. Due to a surge in gene therapy development, demand for plasmid DNA is skyrocketing. Thus, Adeno-associated virus (AAV), lentivirus, and other viral vector systems require pDNA (Plasmid DNA) to be manufactured. In addition, several genetic illnesses and infectious diseases are on the rise in different areas of the globe. For instance, as per UNAIDS data, 38.0 million people across the globe were living with HIV in 2019, and 1.7 million people were newly infected with the virus.
Additionally, rising demand for synthetic genes and untapped expanding market potential are projected to open up new chances for market players in the future. However, the risk of insertional mutagenesis and the high cost of gene treatments stymie market expansion. Furthermore, technical innovations that address the constraints posed by traditional vector production processes create an attractive potential for industry manufacturers.
Some of the prominent players in the Viral Vectors And Plasmid DNA Manufacturing Market include:
- Merck KGaA
- Lonza
- FUJIFILM Diosynth Biotechnologies
- Thermo Fisher Scientific
- Cobra Biologics
- Catalent Inc.
- Wuxi Biologics
- Takara Bio Inc.
- Waisman Biomanufacturing
- Genezen laboratories
- Batavia Biosciences
- Miltenyi Biotec GmbH
- SIRION Biotech GmbH
- Virovek Incorporation
- BioNTech IMFS GmbH
- Audentes Therapeutics
- BioMarin Pharmaceutical
- RegenxBio, Inc.
Segments Covered in the Report
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2032. For this study, Nova one advisor, Inc. has segmented the global Viral Vectors And Plasmid DNA Manufacturing market.
By Vector Type
- Adenovirus
- Retrovirus
- Adeno-Associated Virus (AAV)
- Lentivirus
- Plasmids
- Others
By Workflow
- Upstream Manufacturing
- Vector Amplification & Expansion
- Vector Recovery/Harvesting
- Downstream Manufacturing
By Application
- Antisense & RNAi Therapy
- Gene Therapy
- Cell Therapy
- Vaccinology
- Research Applications
By End-use
- Pharmaceutical and Biopharmaceutical Companies
- Research Institutes
By Disease
- Cancer
- Genetic Disorders
- Infectious Diseases
- Others
By Product Type
- Plasmid DNA
- Viral Vector
- Non-Viral Vector
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa (MEA)

