Risk-based Monitoring Software Market Size and Trends 2026 to 2035
The global risk-based monitoring software market size was reached at USD 511.00 million in 2026 and is expected to exceed around USD 1,781.23 million by 2035, with a CAGR of 13.3% from 2026 to 2035.
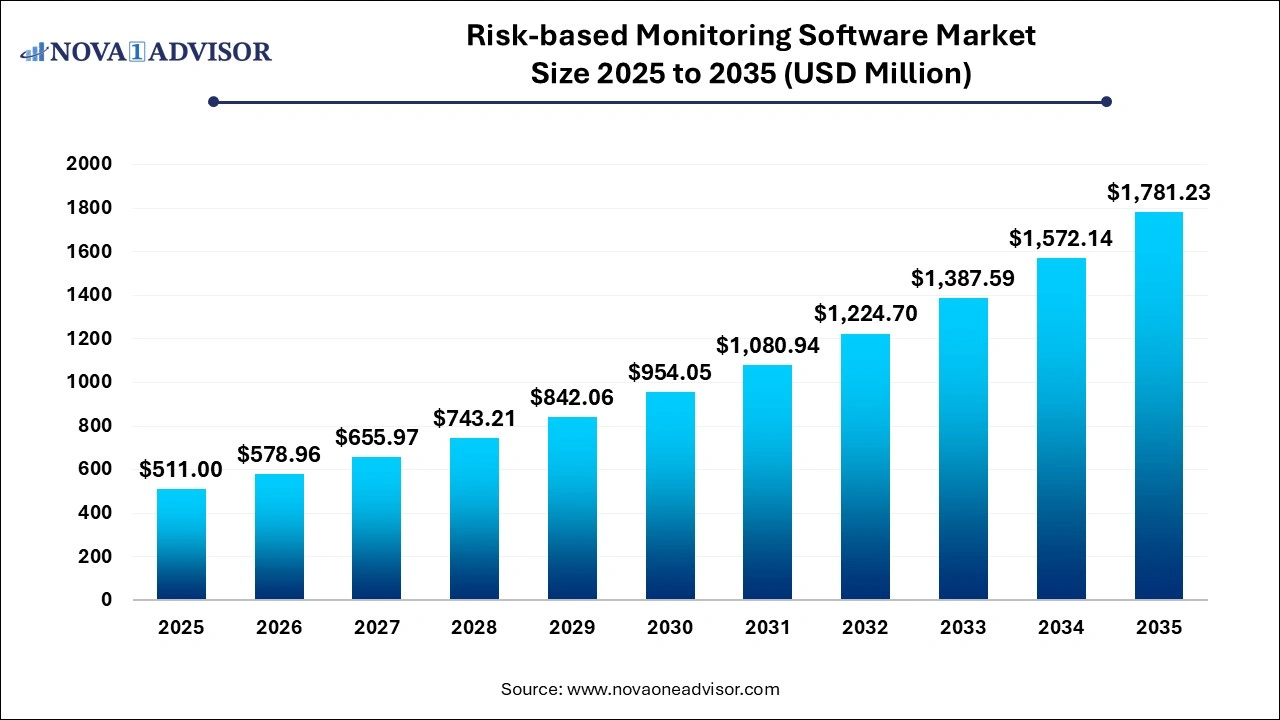
Risk-based Monitoring Software Market Outlook
- Market Growth Overview: The risk-based monitoring software market is expected to grow significantly between 2025 and 2034, driven by the rising complexity of clinical trials and cost reduction, integration of technological advancements, and shift to remote/hybrid trials.
- Sustainability Trends: Sustainability trends involve ESG integration and risk assessment, adoption of AI and ML, and decentralized and hybrid trials.
- Major Investors: Major investors in the market include Oracle, Medidata Solutions, Veeva Systems, Parexel, ArisGlobal, and IBM.
Risk-based Monitoring Software Market Segmental Insights
By Type Insights
How did the Enterprise RBM Software Segment Dominate the Risk-based Monitoring Software Market?
The enterprise RBM software segment is driven by integrating AI and machine learning with key risk indicators (KRIs). These robust solutions allow sponsors to detect anomalies and predict trial issues before they escalate. The high cost and time efficiency by optimizing on-site monitoring and ensuring strict regulatory compliance across complex, decentralized clinical trial environments.
How did the Side RBM Software Segment Expect to Hold the Fastest-Growing Risk-based Monitoring Software Market in the Coming Years?
The side RBM software segment is driven by it provides real-time, local insights that allow for the immediate resolution of site-specific data and safety issues. Its rapid adoption is fueled by the rise of decentralized and hybrid clinical trials, where robust remote management and AI-driven predictive analytics are essential for maintaining data integrity. By optimizing resource allocation and meeting strict FDA/EMA regulatory mandates, these tools significantly reduce the high costs associated with traditional manual on-site monitoring.
By Component Insights
How did the Software Segment Account for the Largest Share in the Risk-based Monitoring Software Market?
The software segment is driven by the increasing complexity of Phase III clinical trials and high data volumes. Its growth is fueled by web-based and cloud-based platforms that provide real-time risk assessment, predictive modeling, and enhanced productivity for global research teams. These advanced analytical tools are indispensable for maintaining strict regulatory compliance and preventing data breaches within modern, data-heavy R&D environments.
How did the Services Segment Expect to Hold the Fastest-Growing Risk-based Monitoring Software Market in the Coming Years?
The services segment is driven by the critical need for specialized consulting and process redesign as companies transition from traditional monitoring. High demand for regulatory expertise regarding ICH E6(R3) guidelines and the outsourcing of complex trial operations to CROs are major catalysts for this expansion. The integration of AI-driven analytics and cybersecurity services ensures that centralized, cloud-based systems remain compliant and secure across rapidly growing research hubs.
By Delivery Mode Insights
How did the Web-Based Segment Account for the Notable Share in the Risk-based Monitoring Software Market?
The web-based segment is driven by they provide instant data accessibility and high operational efficiency, allowing for real-time risk mitigation across decentralized trial sites. Their subscription-based models eliminate significant upfront infrastructure costs, offering a cost-effective and scalable solution that adjusts to changing study demands. They seamlessly integrate advanced AI and predictive analytics into existing workflows, ensuring robust data management for high-volume pharmaceutical research.
How did the Cloud-Based (Saas) Segment Expect to Hold the Fastest-Growing Risk-Based Monitoring Software Market in the Coming Years?
The cloud-based (SaaS) segment is driven by it eliminates massive upfront hardware costs, offering a scalable and cost-effective solution for both SMEs and large enterprises. Its ability to provide real-time data integration with EDC systems allows for proactive risk identification and faster, data-driven decision-making. Furthermore, built-in regulatory compliance features and the seamless integration of AI/ML predictive analytics make these platforms significantly more competitive than traditional, static monitoring tools.
By End-User Insights
How Did the Pharmaceutical & Biopharmaceutical Companies Segment Account for the Notable Share in the Risk-Based Monitoring Software Market?
The pharmaceutical & biopharmaceutical companies segment is driven by adopting RBM solutions; these firms can reduce on-site monitoring costs, which typically account for 30% of drug development expenses, through efficient centralized data analytics. A strategic focus on quality oversight and regulatory compliance with FDA/EMA mandates drives their heavy investment in AI-integrated monitoring tools to ensure data integrity.
How Did the Contract Research Organization Segment Expect to Hold the Fastest-Growing Risk-Based Monitoring Software Market in the Coming Years?
The contract research organization segment is driven by the strategic outsourcing of increasingly complex R&D pipelines. Their rapid adoption of AI-integrated RBM tools and sophisticated software for decentralized clinical trials allows for superior remote monitoring and faster trial completion. By utilizing these platforms to ensure strict regulatory compliance and patient safety, CROs have become indispensable partners for pharmaceutical firms looking to mitigate risk in high-stakes global studies.
Key Players in the Risk-based Monitoring Software Market
- Oracle: Oracle provides comprehensive, AI-driven risk management and compliance cloud applications that, when integrated with their Oracle Clinical One platform, enable continuous, data-driven monitoring of study sites and trial data. Their solutions allow for centralized, automated risk assessments and real-time visibility into key risk indicators (KRIs) across global clinical trials.
- Medidata Solutions: As a Dassault Systèmes company, Medidata offers the Medidata Clinical Cloud™, which features unified RBM tools like Medidata Detect and Rave TSDV to automate data surveillance, identify anomalies, and manage site performance.
- Parexel: Parexel contributes through its Risk-based Quality Management (RBQM) services, which utilize central statistical monitoring to identify risks and guide on-site monitoring activities.
- Bioclinica: Bioclinica offers an integrated technology suite featuring Medical Imaging, Electronic Data Capture (EDC), and Interactive Response Technology (IRT) to support RBM workflows. Bio-Optronics: Bio-Optronics contributes to the market with its eClinical software solutions, specifically designed to automate study management and improve operational efficiency.
Unravelling the Critical Segments
This research report offers market revenue, sales volume, production assessment and prognoses by classifying it on the basis of various aspects including product type, application/end-user, and region. Further, this research study investigates market size, production, consumption and its development trends at global, regional, and country level for period 2017 to 2027 and covers subsequent region in its scope:
By Type
- Enterprise RBM Software
- Site RBM Software
By Component
By Delivery Mode
- Web-based (On-demand)
- Licensed Enterprise (On-premise)
- Cloud-based (SaaS)
By End-User
- Pharmaceutical & Biopharmaceutical Companies
- CROs
- Medical Device Companies
- Other End Users
By Region
- North America
- Europe
- Germany
- France
- United Kingdom
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa (MEA)
- GCC
- North Africa
- South Africa
- Rest of Middle East & Africa


