Viral Capsid Development Market Summary
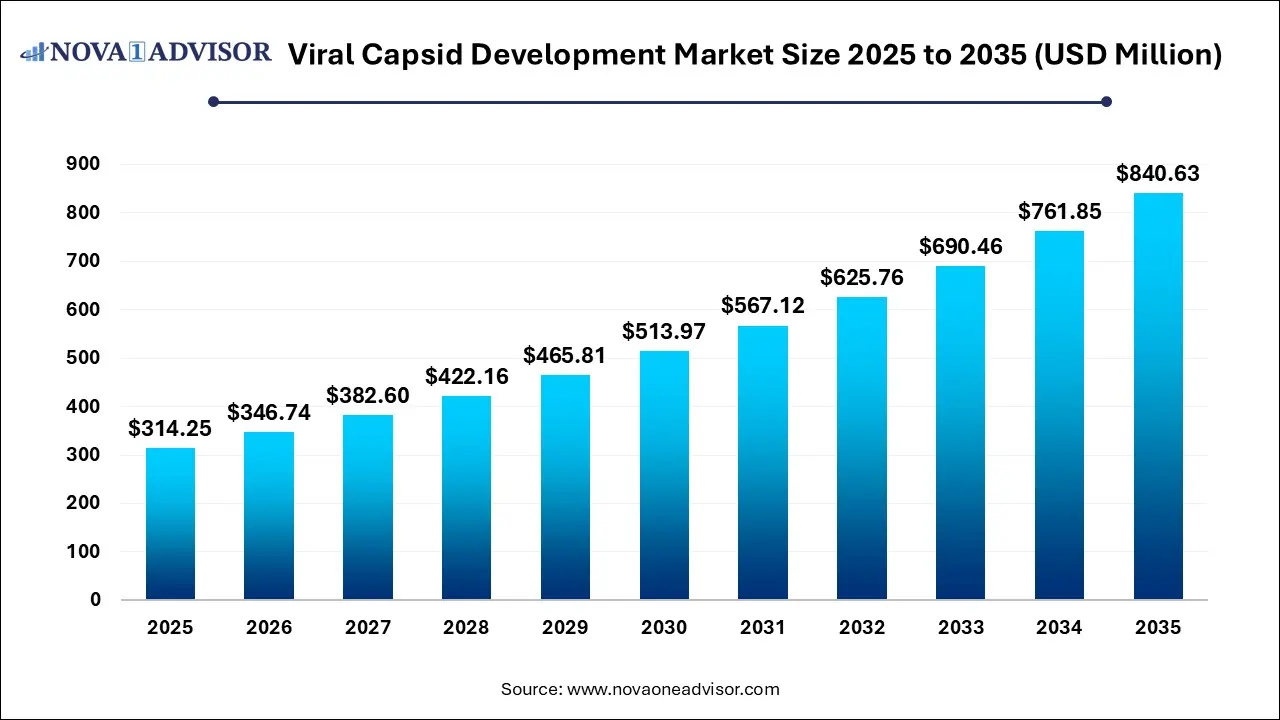
The global viral capsid development market size was estimated at USD 314.25 million in 2025 and is expected to reach USD 840.63 million in 2035, expanding at a CAGR of 10.34% over the forecast period of 2026-2035. The market growth is driven by increased regulatory approvals for gene-based treatments, innovation in technological advancements in engineering, and rising demand for large-scale, cost-effective manufacturing, which has led to increased partnerships with CDMOS.
Viral Capsid Development Market Key Takeaways
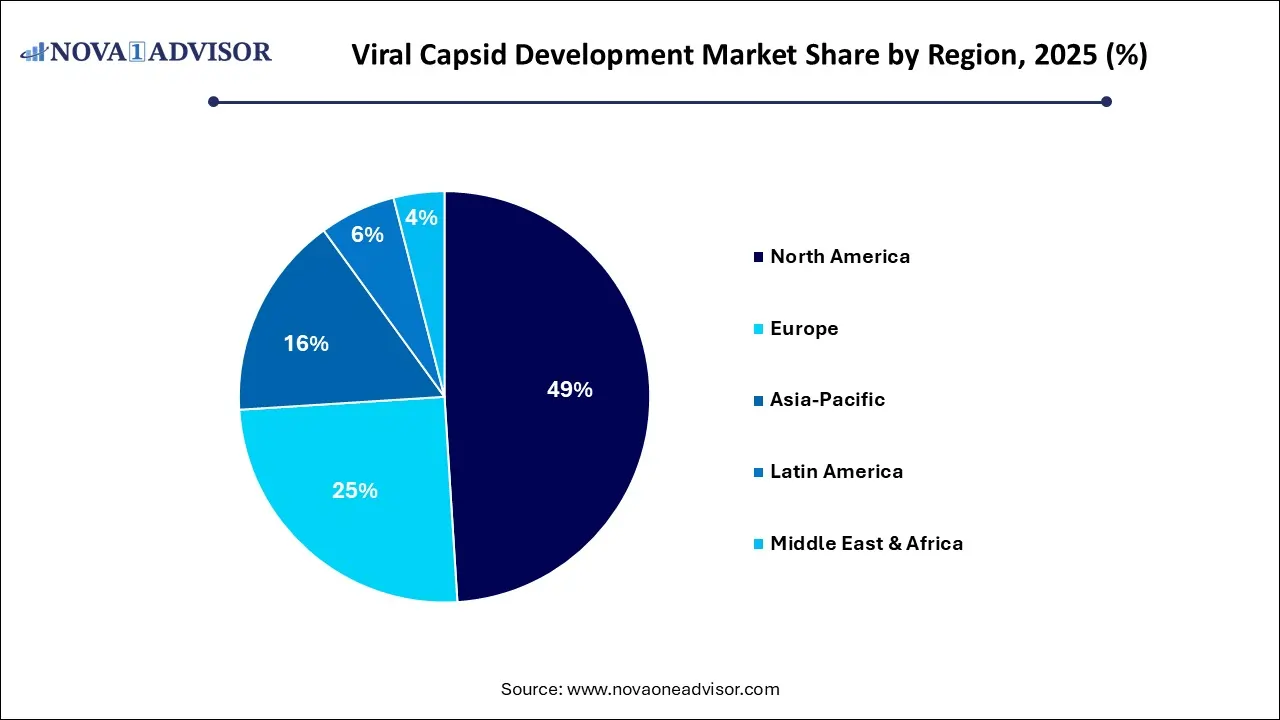
- By region, North America held the largest share of the viral capsid development market in 2025.
- By region, Asia Pacific is expected to experience the fastest growth between 2026 and 2035.
- By type, the adeno-associated virus (AAV) capsid segment led the market in 2025.
- By type, the lentiviral segment is expected to expand at the highest CAGR over the projected timeframe.
- By application, the gene therapy segment led the market in 2025.
- By application, the vaccine development segment is expected to expand at the highest CAGR over the projection period.
- By end use, the pharmaceutical and biotechnology companies segment led the market in 2025.
- By end use, the academic and research institute segment is the fastest growing in the viral capsid development market.
Artificial Intelligence: The Next Growth Catalyst in Viral Capsid Development
AI and ML are transforming the viral capsid development field by enabling in silico design of adeno-associated virus vectors, thereby significantly reducing reliance on time-consuming, traditional trial-and-error experimental methods. By analyzing large-scale datasets from next-generation sequencing, AI algorithms can predict which capsid variants will assemble properly and possess desired properties, such as high tissue specificity or low immunogenicity. These tools are, for example, helping to engineer capsids that evade pre-existing immunity, allowing for more effective gene therapies and the potential for re-dosing. Consequently, AI/ML is driving a shift toward highly optimized, programmable capsids that shorten developmental timelines by months, particularly in the competitive CDMO sector.
Strategic Overview of the Global Viral Capsid Development Industry
The viral capsid development market comprises a field that involves creating customized vectors using adeno-associated viruses (AAV) and lentiviruses for precise, tissue-specific, and efficient therapeutic delivery in gene therapies and vaccines. Key benefits include improved targeting of specific tissue, increased payload capacity, lower immune system recognition, and higher safety profiles, which enable lower dosages. The market growth is driven by the expanding portfolio of gene therapies, advancement in AI-driven capsid engineering and directed evolution, and increased investment in rare disease treatments.
Market Outlook
- Market Growth Overview: The viral capsid development market is expected to grow significantly between 2025 and 2035, driven by the rising number of gene therapy products in clinical trials and FDA approvals, advancement in capsid engineering, and increased funding from pharma, biotech, and venture capital, along with expanding CDMO capacity, which is accelerating developments. Advancements in technology are addressing the need for better capsid stability and targeting, driving the development of novel viral vectors.
- Sustainability Trends: Sustainability trends are single-use technology reduce water consumption and cross-contamination, but they generate significant plastic waste. Artificial intelligence and digital twins are increasingly used to optimize viral capsid manufacturing. Researchers are employing green chemistry principles, such as using biodegradable, non-toxic alternatives for transfection and purification processes. The industry is moving from transient transfection methods to stable cell line development for large-scale production, reducing raw material waste and improving batch consistency.
- Major Investors: Major investors in the market include pharmaceutical and biotechnology companies, such as Lonza, Merck KGaA, and Charles River. They fuel expansion by acquiring speciality firms to integrate advanced cell-line development and manufacturing capabilities. Their investments prioritize Adeno-associated virus (AAV) capsids for gene therapy and vaccine development, focusing on enhancing delivery efficiency and reducing immunogenicity.
- Startup Economy: The startup economy in the market is rapidly expanding, driven by the targeting of neurodegenerative & ocular diseases. A large percentage of these startups are focused on breaching the blood-brain barrier (CNS) or treating retinal diseases. Large pharma and biotech are actively acquiring or partnering with these startups. Growing focus on designer AAV capsids for CNS-based gene therapies develops engineered AAV capsids with tailored tropisms for targeted gene therapy.
Viral Capsid Development Market Report Scope
| Report Attribute |
Details |
| Market size value in 2026 |
USD 346.74 Million |
| Revenue forecast in 2035 |
USD 840.63 Million |
| Growth Rate |
CAGR of 10.34% from 2026 to 2035 |
| Base year for Estimation |
2025 |
| Forecast period |
2026 - 2033 |
| Quantitative units |
Revenue in USD million/billion, and CAGR from 2026 to 2035 |
| Segments covered |
Type, Application, End-use, Region |
| Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
| Key companies profiled |
PackGene Biotech Inc.; Sartorius AG; Revvity; VectorBuilder; AAVnerGene; AskBio Inc.; Voyager Therapeutics; uniQure N.V.; Vigene Biosciences; Capsigen. |
Market Dynamics
Drivers
Surge in Gene Therapy Adoption
The creating in urgent demand for safer, more efficient, and tissue-specific delivery vehicles. As the clinical pipeline for gene therapies expands, particularly for rare genetic disorders, developers are heavily investing in custom-engineered adeno-associated virus capsids to improve targeting, reduce immune responses, and allow for lower, more effective dosing. The industry is rapidly advancing from natural capsids to next-generation engineered variants, utilizing directed evolution and artificial intelligence to create specialized, tissue-specific shells.
Advancement in Vector Engineering
Enabling the creation of custom, high-performance vectors, techniques such as directed evolution and AI-driven design, allow researchers to engineer capsids with superior tissue-specific targeting, improved transduction efficiency, and enhanced immune evasion capabilities. The shift towards engineered rather than naturally occurring capsids is critical for expanding gene therapy applications into complex diseases like neuromuscular and CNG disorders, which directly accelerates R&D investments.
Restraint
High Manufacturing Costs and Complexity
The market growth is hindered by creating sustainable barriers to entry and limiting commercial scalability. The production process involves intricate, multi-step procedures, including transfection and purification, which are often costly, time-consuming, and suffer from low, variable yields. The need for specialized, high-end, good manufacturing practice (GMP) compliant facilities and expensive raw materials like plasmid DNA places a heavy financial burden on smaller biotech companies. These complexities lead to long developmental timelines and significant delays in bringing new therapies to market.
Opportunities
Next-Generation Engineered Capsids
Overcoming the limitations of natural serotypes, such as poor targeting and high immunogenicity. Through advanced techniques like rational design, AI-guided engineering, and directed evolution, these custom vectors can be precisely tailored to improve tissue-specific delivery, enhance transduction efficiency, and allow for lower, safer dosing in patients. This increased precision and safety expand the application of viral vectors to a broader range of diseases, particularly in neurology, cardiology, and ophthalmology. The engineered capsids are being designed to facilitate more efficient, scalable, and cost-effective manufacturing processes, directly addressing the production bottlenecks that have historically limited the market.
Automation and Process Intensification
The market is creating immersive opportunities by addressing traditional bottlenecks in gene therapy manufacturing, such as low yields, high costs, and limited reproducibility. The integration of AI-driven design, high-throughput screening, and automated purification allows for rapid optimization of capsids, accelerating development from discovery to clinical trials. By closing manufacturing processes and minimizing human intervention, these advancements not only enhance batch-to-batch consistency but also reduce the cost of goods, meeting stringent regulatory standards.
How Macroeconomic Variables Influence the Viral Capsid Development Market?
Economic Growth and GDP
Economic growth and rising GDP generally lead to positive growth. By fueling increased investment in biotechnology R&D and expanding healthcare infrastructure for gene therapies. Higher disposable incomes and economic stability allow for greater expenditure on advanced medical treatments, which accelerates the demand for viral vectors, specifically Adeno-associated virus (AAV) capsids. While high manufacturing costs and complex regulatory requirements can pose minor, localized constraints, robust economic conditions generally act as a significant driver for market expansion and innovation.
Inflation & Drug Pricing Pressures
It can negatively affect the growth of the viral capsid development market by increasing input costs for raw materials, labor, and manufacturing while simultaneously reducing profit margins and limiting investment in R&D. Although the demand for advanced gene therapies continues to rise, these economic pressures force companies to reconsider investment in expensive, high-risk, long-term development programs, particularly due to policies like the U.S. inflation reduction act.
Exchange Rates
Exchange rate fluctuations can negatively affect, by introducing volatility in the cost of international manufacturing, raw materials, and specialized, technology-driven research. While a weakening local currency can technically boost export revenues for domestic firms, the high reliance on specialized global suppliers and imported technology components means that currency depreciation often leads to inflated operating costs.
Segment Outlook
Type Insights
Why Did the Adeno-Associated Virus (AAV) Capsid Segment Dominate the Viral Capsid Development Market in 2025?
The adeno-associated virus (AAV) capsid segment dominated the market with the largest share in 2025. This is due to engineering next-generation AAV vectors that offer superior safety and precise tissue targeting through AI and machine learning. These innovators solve critical delivery hurdles, such as crossing the blood-brain barrier, which has solidified AAV9's dominant market share. By developing scalable manufacturing processes, startups lower entry barriers for biopharma, accelerating high-demand treatments for neurological and rare genetic disorders.
The lentiviral segment is expected to grow at the fastest CAGR during the projection period, driven by optimizing vectors for CAR-T and autoimmune therapies, ensuring the stable, long-term gene expression required for curative treatments. By pioneering stable producer cell lines, these innovators have shifted manufacturing from costly batch processes to consistent, scalable industrial models. This technical maturation is driving massive R&D investment, particularly in the Asia-Pacific region, where startups are rapidly expanding the clinical pipeline. Their ability to lower production hurdles while enhancing therapeutic durability makes lentiviral technology a cornerstone of the next-generation immunotherapy economy.
Application Insights
How Does the Gene Therapy Segment Lead the Viral Capsid Development Market in 2025?
The gene therapy segment led the viral capsid development market in 2025 due to the engineering-optimized AAV vectors that achieve unprecedented tissue specificity and immune evasion. The critical delivery technology is required for personalized gene therapies to treat rare diseases safely. Heavy investment from the biopharma sector, now over 50% of end-use, is accelerating the transition of these engineered capsids from lab to clinic. The startup economy turns complex biological vectors into scalable, high-value assets that underpin the future of targeted, long-term medicine.
The vaccine development segment is projected to grow at the highest CAGR in the coming years. This is mainly because of leveraging AI and machine learning to design optimized capsids for infectious diseases like HIV and RSV. Their ability to rapidly engineer high-titer AAV and viral vector platforms is critical for global pandemic preparedness and fast, scalable immunization. By focusing on targeted, safer delivery systems, these innovators reduce off-target effects and improve clinical success rates for both public health and private biotech. This technical agility, backed by global health initiatives, is turning advanced capsid engineering into the primary infrastructure for the world's future vaccine R&D.
End Use Insights
How did the Pharmaceutical and Biotechnology Companies Segment dominate the Viral Capsid Development Market in 2025?
The pharmaceutical and biotechnology companies segment dominated the viral capsid development market in 2025 by bringing the gap between high-risk R&D and commercial-scale manufacturing through specialized AAV engineering. By securing massive capital for clinical trials in oncology and neurology, these firms have turned next-generation capsids into high-value therapeutic assets. Their focus on in-house optimization and efficient cell culture systems has lowered production costs while drastically improving gene delivery to the CNS and eyes.
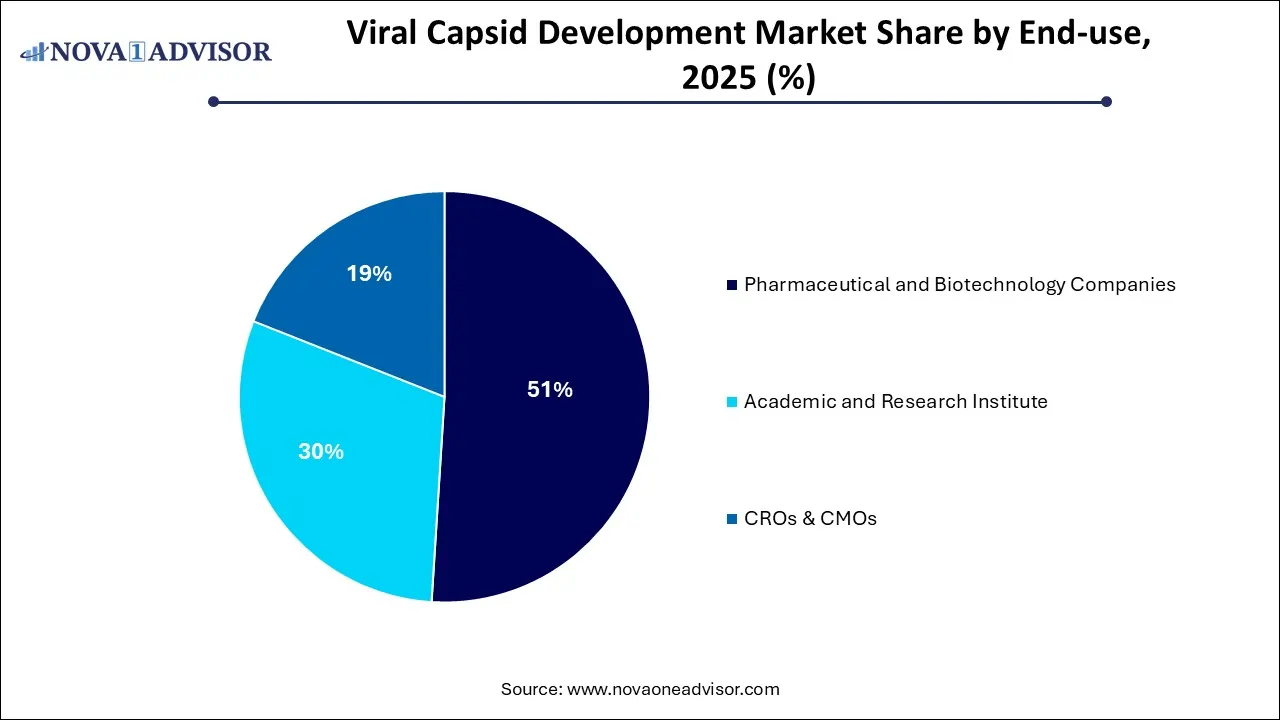
The academic and research institute segment is expected to expand at the highest CAGR in the coming years. This is primarily due to leveraging government grants and CRISPR technologies to de-risk high-quality, specialized capsids for rare diseases. By bridging the skills gap and collaborating with industry, academia provides the trained talent and validated prototypes that startups scale into commercial assets. This synergy transforms laboratory discoveries into clinical-grade technologies, research institutions pioneer novel AAV serotypes, and AI-driven design to solve safety and targeting hurdles.
Regional Analysis
What Made North America the Dominant Region in the Market?
North America maintained dominance in the viral capsid development market while holding the largest share in 2025. The region’s dominance is attributed to integrating world-class biotech hubs with FDA-led regulatory guidance, supported by significant venture capital and academic research. This ecosystem facilitates rapid scaling from discovery to high-volume manufacturing, overcoming critical yield and purity hurdles for advanced gene therapies. The combination of specialized talent, robust funding, and high market demand for therapies targeting the CNS and liver solidifies the region's leadership. This synergy of infrastructure, investment, and clear regulatory pathways drives high-volume, high-quality production to meet growing clinical needs.
U.S. Viral Capsid Development Market Trends
The U.S. is a major contributor to the market in North America due to making AI-driven capsid design the industry standard, drastically reducing trial-and-error to enhance tissue targeting and payload capacity. By shifting from adherent to scalable suspension cell lines, these innovators are slashing production costs while solving the empty capsid problem to ensure higher therapeutic efficacy. This technological leap, combined with a move toward immune-evasive, programmable vectors, allows for the repeat dosing essential for long-term cures.
What Makes Asia Pacific the Fastest-Growing Area in the Market?
Asia Pacific is expected to grow at the fastest rate in the coming years. This is due to leveraging a unique combination of localized, cost-effective manufacturing and rapid clinical expansion. With the aggressive government incentives and streamlined regulations, startups are transforming the region into a premier hub for academic-industry collaborations and high-volume clinical trials. This infrastructure-first approach reduces the cost-per-dose barrier, making advanced capsid engineering accessible and accelerating the region’s rise as a global biotech powerhouse.
China Viral Capsid Development Market Trends
China is a key player in the Asia Pacific viral capsid development market due to adopting single-use bioreactors and modular platforms; these firms are reducing contamination risks and enabling the flexible, small-batch manufacturing essential for personalized medicine. Strong state backing and a focus on international out-licensing are driving these startups to meet global quality standards, positioning them as primary partners for global biotech firms. This infrastructure-led growth, fueled by outsourced CMC functions, is transforming China into a global hub for high-volume, targeted gene therapy delivery.
How is the Opportunistic Rise of Europe in the Viral Capsid Development Market?
Europe is experiencing a strategic growth in the market due to leveraging a world-class academic foundation to pioneer precision-engineered capsids that minimize immune responses in rare disease therapies. With a surging pipeline of over 350 clinical trials, the region is scaling rapidly through aggressive M&A and CDMO expansions, such as Oxford Biomedica’s recent strategic acquisitions. By integrating AI-driven optimization and automation, European firms are drastically improving vector yields while reducing the high costs associated with bespoke manufacturing.
United Kingdom Viral Capsid Development Market Trends
The United Kingdom is leading the market in Europe due to combining academic-led research with advanced in-silico modeling to simulate and optimize capsid performance before production. By integrating automation and high-throughput screening, these firms are rapidly identifying candidates that offer superior tissue targeting and reduced immunogenicity for oncology and neurology. This technical precision, backed by strategic consolidations and specialized manufacturing investments, has turned the UK into a primary hub for high-efficiency viral vector development.
Viral Capsid Development Market Supply Chain Analysis
This initial stage focuses on creating novel, synthetic, or engineered viral capsids using rational design, AI, and directed evolution to improve tissue targeting, reduce immunogenicity, and increase transduction efficiency.
Key Players: Latus Bio, VectorBuilder, Shape Therapeutics, Biogen, Capsigen.
- Manufacturing and Production
This involves producing the engineered capsids at scale, utilizing advanced systems like suspension cell culture, transient transfection, or insect cell-based production. It covers Upstream and Downstream processes.
Key Players: Sartorius AG, PackGene Biotech Inc., Revvity (Vigene Biosciences), Lonza, Thermo Fisher Scientific.
- Clinical Development and Regulatory Approval
This stage includes clinical trials (Phases I-III) to validate the safety and efficacy of the capsid-based therapies in humans. It requires strict compliance with regulatory standards (GMP) to obtain FDA/EMA approval for commercialization.
Key Players: Novartis, Pfizer, Roche
Viral Capsid Development Market Companies
- Revvity contributes to the viral capsid market by leveraging the expertise of SIRION Biotech to provide advanced AAV capsid engineering and directed evolution services that enhance tissue tropism. Their capabilities include generating customized, high-performance capsid libraries and providing comprehensive analytical, QC, and manufacturing services to accelerate gene therapy development.
- VectorBuilder offers a one-stop-shop platform that combines AI-driven in silico design with high-throughput in vivo screening in non-human primates to create, optimize, and validate novel AAV capsids. They specialize in engineering capsids with improved tissue specificity, such as blood-brain barrier penetration, and provide end-to-end CDMO services for large-scale production of these tailored vectors.
- AAVnerGene specializes in the development of customized AAV capsids using their proprietary ATHENA platforms (I, II, and III) that apply directed evolution, random peptide insertion, and barcoding technology to improve targeting and efficiency.
- PackGene Biotech Inc. operates as a leading CRO/CDMO providing end-to-end Adeno-Associated Virus (AAV) vector services, utilizing a proprietary -Alpha 293 high-yield platform to significantly increase production efficiency and reduce costs for gene therapies.
- Sartorius AG provides a comprehensive, end-to-end technology portfolio that supports the entire viral capsid production process, from upstream cell culture in bioreactors to downstream purification using advanced membrane chromatography.
Recent Developments
- In September 2025, Abselion has launched the AAVX and AAV9 Total Capsid Quantification Kits for its Amperia benchtop platform, incorporating Thermo Fisher Scientific’s CaptureSelect affinity reagents for improved, high-specificity AAV titer measurements. The new kits streamline AAV development by offering a ready-to-use, sandwich-style electrochemical immunoassay that eliminates the need for extensive in-house optimization and complex optics.
- In August 2025, ProBio launched end-to-end current good manufacturing practice (CGMP) adeno-associated virus (AAV) manufacturing services at its 128,000-sq.-ft. facility in Hopewell, N.J., targeting accelerated development timelines. This site provides integrated services from plasmid DNA production to aseptic fill and finish, offering scalable batch sizes from 50 L to 200 L to support both clinical and commercial gene therapy programs.
- In November 2025, Latus Bio expanded its AI/ML strategy to accelerate AAV gene therapy development, leveraging an extensive in vivo database from non-human primates to optimize capsid discovery and payload design. The company has added experts Dr. Pranam Chatterjee and Dr. Philip M. Kim to its scientific advisory board to advance this data-powered predictive engine.
Exclusive Analysis on the Viral Capsid Development Market
The global viral capsid development market is leveraging an in vivo database of unique delivery zip codes (capsid-tissue-cell pairings). Latus Bio is moving away from blind screening. Their strategy, supported by protein modeling experts like Drs. Pranam Chatterjee and Philip Kim, uses large language models (LLMs) to predict how a capsid will behave in a human body before it is ever synthesized. The FDA’s OTAT has streamlined the path for engineered capsid, evidenced by the approval of Kebilidi, an AAV-based therapy targeting the brain.
From an analyst’s perspective, the market’s growth vector is being catalyzed by the transition from random screening to AI-driven rational design, which utilizes massive in vivo datasets to predict precise tissue targeting and shorten R&D timelines. The advanced manufacturing transitions, specifically the move to scalable suspension cell lines and high-precision quantification tools that solve the empty capsid purity hurdle. Significant capital inflow from Big Pharma, evidenced by billion-dollar licensing deals for proprietary AAV platforms, has transformed engineered capsids into high-value intellectual property assets.
Segments Covered in the Report
By Type
- Adeno-associated Virus (AAV) Capsids
- Adenoviral
- Lentiviral
- Others
By Application
- Gene Therapy
- Vaccine Development
- Cancer Therapy
- Others
By End-use
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institute
- CROs & CMOs
By Regional
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa



