Sterile Filtration Market Size, Growth and Trends 2026 to 2035
The global sterile filtration market size was estimated at USD 7.99 billion in 2025 and is expected to reach USD 17.25 billion by 2035, growing at a CAGR of 8% from 2025 to 2035.
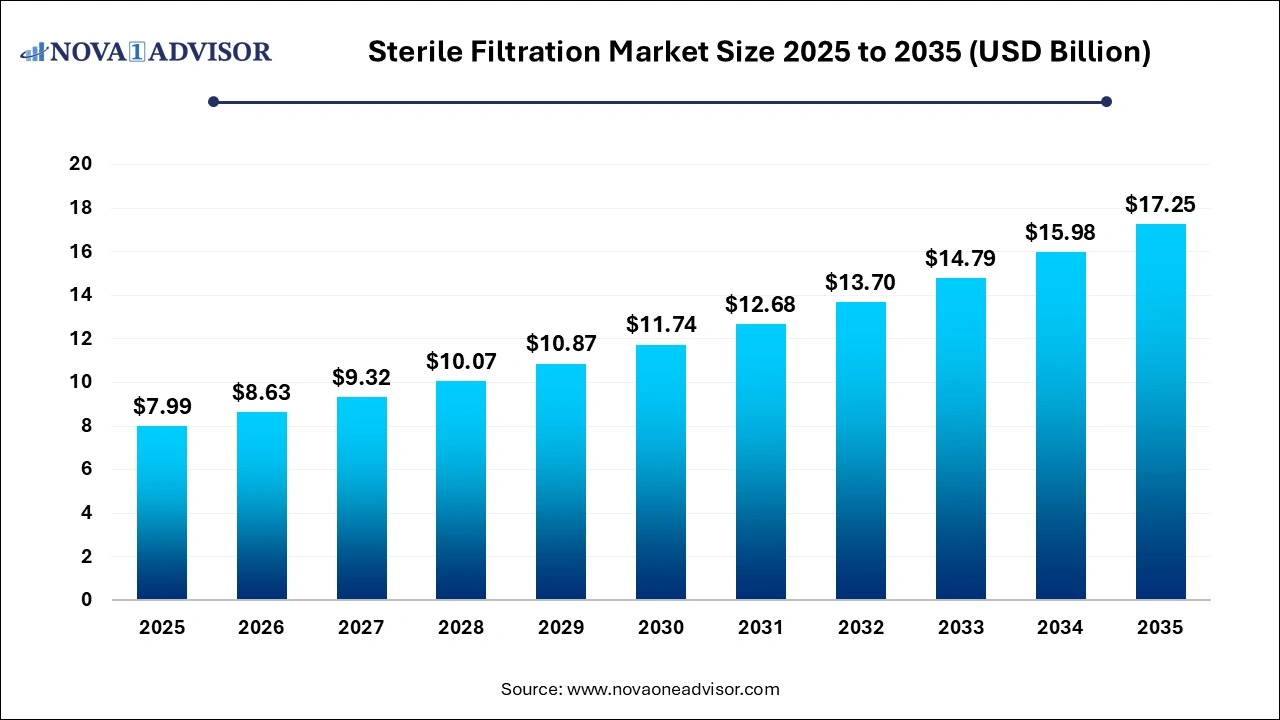
Sterile Filtration Market Outlook
- Market Growth Overview: The sterile filtration market is expected to grow significantly between 2026 and 2035, driven by the growing demand for biopharma and biological sectors, stringent regulatory compliance, and the shift toward a single-use system adoption.
- Sustainability Trends: Sustainability trends involve the rise of single-use systems, eco-friendly material development, and energy-efficient processes.
- Major Investors: Major investors in the market include Merck KGaA (MilliporeSigma), Danaher Corporation (Pall Corporation), and Sartorius AG.
Sterile Filtration Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 8.63 Billion |
| Market Size by 2035 |
USD 17.25 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 8% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Segments Covered |
By Product, By End-User, By Application, By Membrane Type, By Pore Size |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
Merck KGaA, Danaher Corporation, Sartorius AG |
Sterile Filtration Market Segmental Insights
By Product Insights
How did the Cartridge Filters Segment Dominate the Sterile Filtration Market?
The cartridge filters segment is driven by its widespread use, anchored by strict FDA and EMA regulatory mandates for high-integrity microbial removal in vaccine and drug manufacturing. As industry workflows shift toward single-use technologies, the demand for these scalable, PES-membrane-based formats continues to rise to ensure efficient, compliant, and sterile processing. Its role is the workhorse of large-scale production, offering the high surface area and throughput required for biopharmaceutical sterilization.
How did the Capsule Filters Segment Expect to Hold the Fastest-Growing Sterile Filtration Market in the Coming Years?
The capsule filters segment is driven by the industry-wide shift toward single-use technologies, which eliminate complex cleaning validation and reduce cross-contamination risks. Their ability to provide cost-effective scalability and faster changeovers makes them essential for the booming biologics and biosimilars pipelines managed by global CMOs. As manufacturing hubs in the Asia-Pacific region expand, these validated, high-performance filters have become the primary choice for meeting strict global regulatory standards in modern vaccine and therapy production.
By End-User Insights
How did the Pharmaceutical Companies Segment Account for the Largest Share in the Sterile Filtration Market?
The pharmaceutical companies segment is driven by the massive production volumes of monoclonal antibodies and vaccines, which require rigorous, high-capacity sterile filtration. Adherence to strict cGMP guidelines necessitates the continuous use of validated membrane filters and disposable cartridges to ensure absolute sterility and patient safety. As manufacturing expands into emerging markets, the high-volume demand for these essential consumables remains the primary driver of market share and industrial scale.
How did the Biopharmaceutical Companies Segment Expect to Hold the Fastest-Growing Sterile Filtration Market in the Coming Years?
The biopharmaceutical companies segment is driven by the rapid expansion of manufacturing hubs in the Asia Pacific region and a strategic shift toward single-use capsule technologies to ensure flexible, high-throughput processing. With the strict regulatory safety standards and a rising reliance on specialized CDMOs, this segment remains the primary engine for advanced filtration innovation.
By Application Insights
How did the Bioburden Reduction Segment Account for the Largest Share in the Sterile Filtration Market?
The bioburden reduction segment is driven by the rising production of complex biologics and vaccines, which require specialized cartridge filters to remove impurities and cell debris. As a mandatory step for final product reliability, bioburden reduction remains the highest-volume application for sterile filtration consumables in large-scale pharmaceutical operations. The bioburden reduction application is rooted in its role as a critical in-process control, ensuring microbial levels are minimized before final sterilization to meet strict FDA and global regulatory mandates.
How did the Fill-Finish Processes Segment Expect to Hold the Fastest-Growing Sterile Filtration market in the Coming Years?
The fill-finish processes segment is driven by the critical need for terminal sterilization and stringent contamination control in vaccines and biologics. The rapid adoption of single-use, closed-processing systems is accelerating this growth by integrating high-performance filters for venting and gas management in automated lines. As specialized CDMOs scale up high-volume production for next-generation therapies, the demand for advanced, validated filtration technology at this final manufacturing stage continues to surge.
By Membrane Type Insights
How did the PES (Polyethersulfone) Sgment Account for the Largest Share in the Sterile Filtration Market?
The PES (Polyethersulfone) segment is driven by its high porosity and superior flow rates, which ensure efficient high-flux filtration in large-scale processing. Their low protein-binding nature is critical for biopharmaceuticals, as it minimizes the loss of expensive active ingredients during the sterilization of biological fluids. Supported by a long-standing clinical track record and FDA compliance, PES remains the most reliable and durable material for meeting the rigorous thermal and chemical demands of modern drug manufacturing.
How did the PES & PVDF Segment Expect to Hold the Fastest-Growing Sterile Filtration Market in the Coming Years?
The PES & PVDF segment is driven by the its low protein binding and high-flux performance. Their superior technical properties, including high thermal stability and chemical resistance, make them indispensable for meeting strict global regulatory sterility standards. As the industry shifts toward single-use disposable systems, these high-performance materials are increasingly integrated into cartridge filters to ensure faster validation and safer, more efficient manufacturing.
By Pore Size Insights
How did the 0.2-0.22 μm segment account for the largest share in the Sterile Filtration market?
The 0.2-0.22 μm segment is driven by its essential role in final drug product filtration and security assurance, effectively removing bacteria, such as Brevundimonas diminuta. Available in high-performance materials like PES and PVDF, these filters are indispensable for the high-volume bioprocessing of vaccines and complex biologics. The global regulatory gold standard for sterilising-grade filtration in the pharmaceutical and biotech sectors.
How did the 0.1 μm Segment Expect to Hold the Fastest-Growing Sterile Filtration Market in the Coming Years?
The 0.1 μm segment is driven by the critical need to remove Mycoplasma and ultrafine contaminants that bypass traditional 0.22 μm filters. This acceleration is fueled by stringent Annex 1 regulatory revisions and the booming production of mRNA vaccines and cell therapies, which demand the highest levels of sterility assurance. By offering a superior balance of high permeability and low pressure loss, these advanced PES membranes allow manufacturers to maximize throughput while ensuring the absolute purity of complex, high-value biologics.
Key Players in the Sterile Filtration Market
- Merck KGaA: Merck KGaA provides a comprehensive portfolio of Millipore Express sterilizing-grade membranes and Mobius single-use assemblies, which are essential for maintaining sterility in biologics and vaccine production.
- Danaher Corporation: Danaher, operating through its Pall Corporation and Cytiva brands, provides integrated sterile filtration solutions, including advanced membranes and single-use technologies that are crucial for high-capacity vaccine and biologics manufacturing.
- Sartorius AG: Sartorius focuses on high-performance sterile filters, such as Sartopore and Sartobran PES membranes, specifically designed to meet rigorous standards like EU GMP Annex 1 for microbial retention and sterile filtration.
Segments Covered in the Report
By Product
- Membrane
- Capsules Filters
- Cartridge Filters
- Syringe Filters
- Bottle-top & Table-top Filtration Systems
- Accessories
By End user
- Pharmaceutical Companies
- Biopharmaceutical Companies
- Food & Beverage Companies
- Academics Institutes & Research Laboratories
- CMOs & CROs
By Aapplication
- Fill-finish Processes
- Bioburden Reduction
- Buffer Filtration & Cell Culture Media Preparation
- Water Purification
- Air Filtration
By Membrane Type
- PES
- PVDF
- Nylon
- PFTE
- MCE & CA
- Other Materials
By Membrane Pore Size
- 0.1 μm
- 0.2–022 μm
- 0.45 μm
By Region
- North America
- Europe
- Asia Pacific
- LATAM
- MEA


