Lyophilization Equipment and Services Market Size, Growth and Trends 2026 to 2035
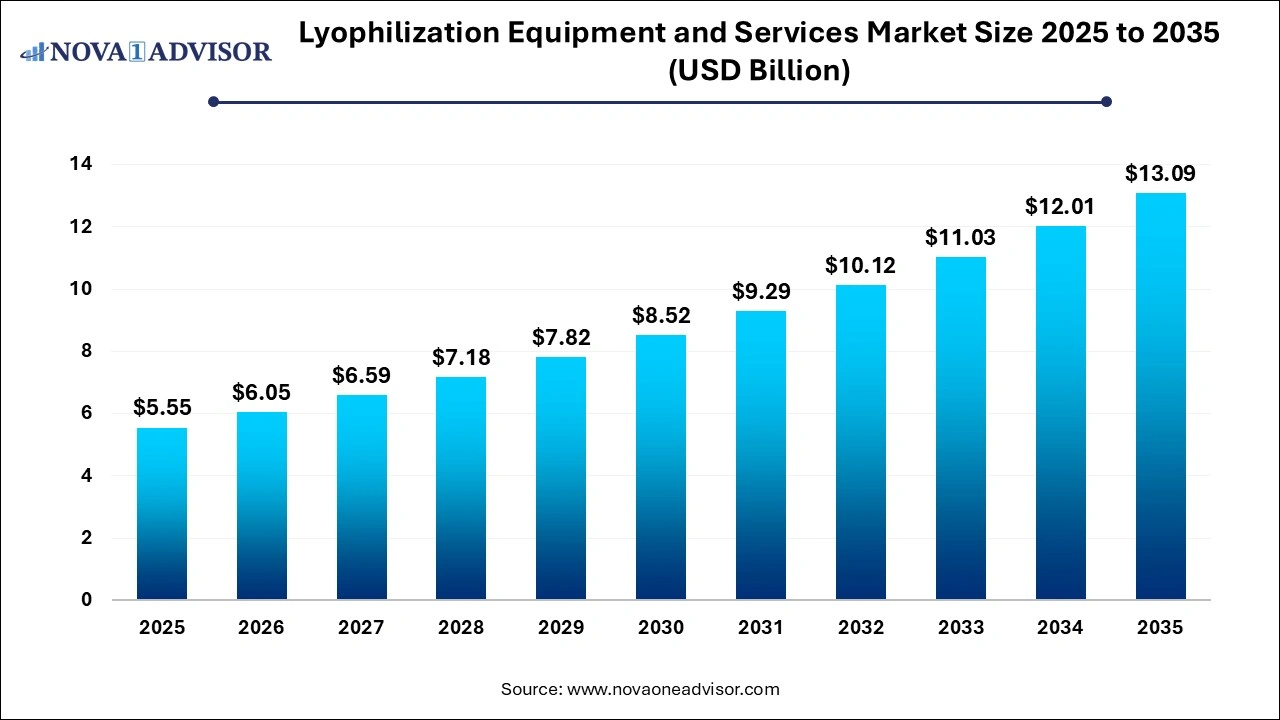
The global lyophilization equipment and services market was estimated at USD 5.55 billion in 2025 and is projected to reach USD 13.09 billion by 2035, growing at a CAGR of 8.96% over the forecast period 2026 to 2035. Market growth is driven by increasing prevalence of chronic diseases and the shift toward biologics and vaccines demand sophisticated freeze-drying to maintain potency, advancements in technology, modern equipment now features automated, energy-efficient designs, separate drying chambers to prevent cross-contamination, and real-time monitoring through IoT, and expanding outsourcing to CDMOs.
Lyophilization Equipment and Services Market Key Takeaways
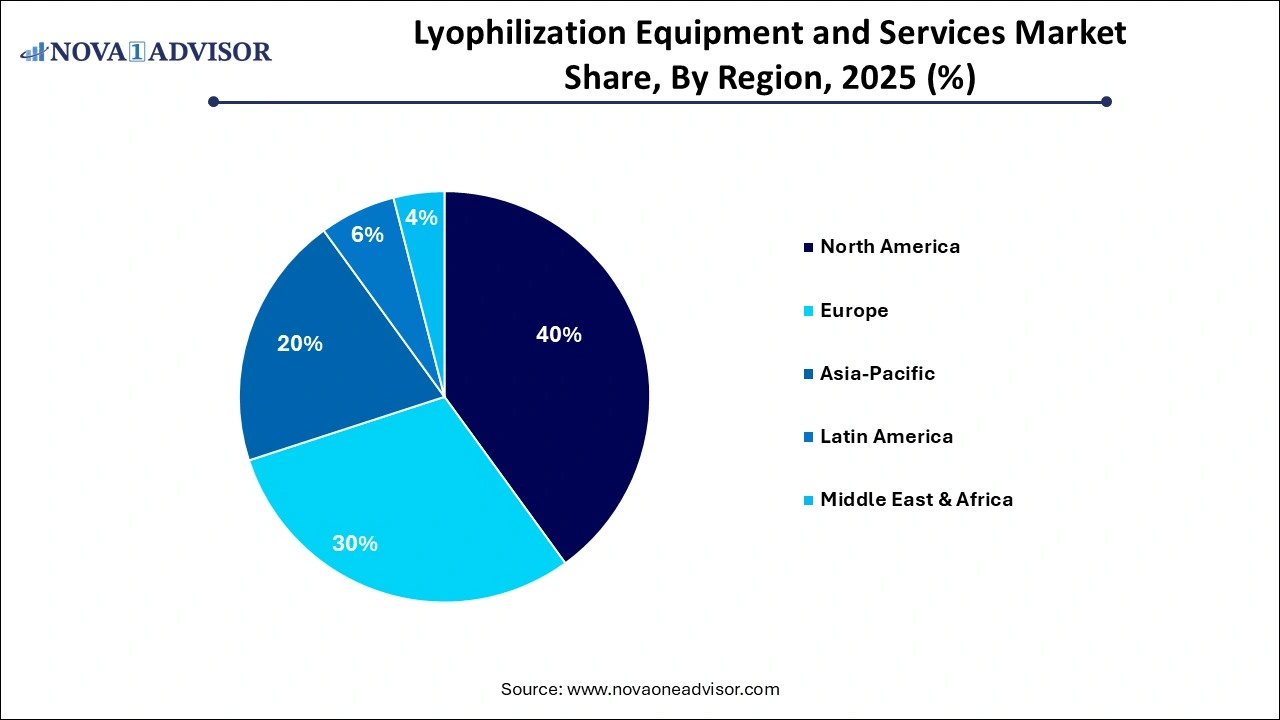
- By region, North America held the largest share of the lyophilization equipment and services market in 2025.
- By region, Asia Pacific is expected to experience the fastest growth between 2026 and 2035.
- By modality, the consumables and accessories segment led the market in 2025.
- By modality, the services segment is expected to expand at the highest CAGR over the projected timeframe.
- By scale of operation, the industry scale segment led the market in 2025.
- By scale of laboratory scale, the therapeutic segment is the fastest growing in the lyophilization equipment and services market.
- By application, the pharmaceutical and biotechnology companies segment dominated the lyophilization equipment and services market in 2025.
Artificial Intelligence The Next Growth Catalyst in Lyophilization Equipment and Services
AI and ML are transforming the lyophilization equipment and services industry by replacing traditional, manual, empirical methods with data-driven, intelligent process control, thereby enhancing product quality and consistency. These technologies analyze vast datasets to optimize freeze-drying cycles in real time, drastically reducing process variability and minimizing product loss during the production of sensitive biologics. AI-driven predictive maintenance allows manufacturers to preempt equipment failures and reduce maintenance costs, thereby increasing operational efficiency.
Strategic Overview of the Global Lyophilization Equipment and Services Industry
The lyophilization equipment and services market, which uses specialized freeze-drying technology to remove moisture from frozen products via sublimation, is experiencing significant growth. This market is vital for stabilizing heat-sensitive, labile materials, such as biopharmaceuticals, vaccines, and diagnostic reagents, by extending their shelf life, maintaining structural integrity, and allowing room-temperature storage. Beyond pharmaceuticals, the technology offers crucial advantages in the food industry, including the preservation of flavor, nutrition, and texture in fruits, ready-to-eat meals, and dairy products, alongside increased use in probiotics and nutraceuticals. Key growth factors propelling this market include the rising demand for biologics and injectables, the need for advanced cold-chain-independent logistics, and increasing CDMO outsourcing for high-complexity, large-scale production.
Lyophilization Equipment and Services Market Outlook
- Market Growth Overview: The lyophilization equipment and services market is expected to grow significantly between 2025 and 2034, driven by the rising demand for biopharmaceuticals and vaccines, which is critical for stabilizing thermo-sensitive biologics and proteins. Innovation in technology, such as automated, high-capacity machinery, advanced vacuum systems, and superior control software are increasing efficiency and ensuring compliance with strict quality standards, and expansion in the food processing industry.
- Sustainability Trends: Sustainability trends are energy efficiency improvements; manufacturers are incorporating low-energy vacuum pumps, optimized airflow designs, and advanced monitoring systems to cut power usage, as freeze-drying is traditionally energy-intensive. Alternative refrigerants, heat recovery, waste minimization, and a circular approach.
- Major Investors: Major investors in the market include GEA Group AG, Tofflon Science and Technology Group, Thermo Fisher Scientific, IMA Industria Macchine Automatiche S.p.A., and SP Industries. They are accelerating market growth through strategic acquisitions, such as Syntegon's acquisition of Azbil Telstar, and by developing advanced, energy-efficient, and automated technology to meet increasing biopharmaceutical demand.
- Startup Economy: The startup economy in the market consists of agile technology firms and specialty contract service providers focused on automating processes, enhancing energy efficiency, and reducing the high cost of freeze-drying. These emerging companies, along with niche CDMOs, drive the market by offering modular, AI-enabled, and IoT-integrated equipment suitable for smaller, specialized biopharmaceutical batches.
Lyophilization Equipment and Services Market Report Scope
| Report Coverage |
Details |
| Market Size in 2026 |
USD 6.05 Billion |
| Market Size by 2035 |
USD 13.09 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 8.96% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Segments Covered |
By Modality Insights, Scale of Operation Insights, Application Insights |
| Regional Scope |
North America; Europe; Asia Pacific; Central and South America; the Middle East and Africa |
| Key Companies Profiled |
Tofflon Science and Technology Co., Ltd., Martin Christ Freeze-Drying Systems GmbH, Azbil Corporation, SP Industries (SP Scientific), GEA Group |
Lyophilization Equipment and Services Market Dynamics
Drivers
Expansion of the Biopharmaceutical Sectors
The rapid expansion of the biopharmaceutical sector, particularly the rising development of biologics, vaccines, and personalized medicines, acts as the primary driver for the lyophilization equipment and services industry. As these advanced therapies are often highly temperature-sensitive and unstable in liquid form, lyophilization is essential to ensure their long-term stability, shelf life, and efficacy. This growing need for stabilization has accelerated the demand for industrial-scale, technologically advanced lyophilizers that offer high-precision control. According to Pharma Now. The lyophilization is critical for biologics and injectable drugs; it transforms delicate, short-lived medicines into resilient, travel-ready warriors.
Rising of CDMO Outsourcing
According to Mantel Associates the outsourcing to CDMOs allows pharmaceutical companies to minimize capital investments in infrastructure, equipment, and personnel. It provides flexibility in scaling up or down manufacturing capacity based on demand, reducing costs and improving overall operational efficiency. To manage this increased outsourcing, CDMOs are aggressively investing in new, large-scale, and automated lyophilizers, which directly boost the sales of advanced equipment manufacturers. India's shift toward operating models reshapes an entire industry; in the global pharmaceutical is happening now, India's CDMOs sector sits squarely at its centre.
Restraint
High Capital Investment and Operational Costs
The market growth is hindered by creating significant barriers to entry, particularly for small and medium-sized enterprises. The initial setup cost for industrial-scale lyophilizers can reach millions of dollars, leading many companies to opt for lower-cost, alternative drying methods such as spray drying, even if they result in reduced product quality. The process is energy-intensive, requiring high utility expenditures for refrigeration and vacuum systems, which directly impacts operating margins in cost-sensitive industries.
Opportunities
Continuous Manufacturing Adoption
The expansion of lyophilization equipment and services creates a significant opportunity, according to Freeze Drying Systems Pvt. Ltd., by demanding advanced, automated, and high-efficiency systems that replace traditional batch-wise processing. This shift creates substantial opportunities for equipment manufacturers to provide cutting-edge solutions, including continuous freeze-dryers with integrated process analytical technology (PAT), designed to reduce processing times by up to 10 times and minimize product variability. The transition also fuels the expansion of contract development and manufacturing organizations (CDMOs), as pharmaceutical companies seek specialized services to implement these complex, expensive technologies without bearing the full capital expenditure.
Cell and Gene Therapies (CGTs)
The market is creating immersive opportunities by creating an urgent need to stabilize fragile, temperature-sensitive products such as viral vectors, mRNA, and engineered cells. As the number of FDA-approved CGTs rises, manufacturers require advanced, specialized freeze-drying equipment that ensures long-term viability, portability, and reduced dependence on costly cold-chain logistics. The complexity of these biologics creates high demand for tailored lyophilization cycles, which drives the need for contract development and manufacturing organizations (CDMOs) with specialized expertise in small-batch and aseptic processing.
How Macroeconomic Variables Influence the Lyophilization Equipment and Services Market?
- Economic Growth and GDP: Economic growth and rising GDP generally lead to positive growth. By increasing disposable income, which boosts demand for processed, high-quality, and freeze-dried food products. Expanding economies also spur increased healthcare spending and R&D investment, leading to higher adoption of freeze-drying technology for pharmaceutical biologics, vaccines, and injectables.
- Inflation & Drug Pricing Pressures: It can negatively affect the growth of the lyophilization equipment and services market by increasing the high capital, energy, and operational costs associated with the technology, which squeezes profit margins for pharmaceutical manufacturers. While rising operational expenses create financial barriers, these pressures simultaneously drive a shift toward outsourcing to contract development and manufacturing organizations (CDMOs) for cost-efficient solutions and increased adoption of automated, energy-efficient equipment to manage long-term costs.
- Exchange Rates: Exchange rate fluctuations can both positively and negatively affect, depending on the currency fluctuation direction, acting primarily as a driver when a weaker local currency makes exported equipment more competitive. Conversely, a strengthening local currency can act as a restraint by making imported high-tech freeze-drying systems more expensive, increasing operational costs for local manufacturers.
Lyophilization Equipment and Services Market Segment Insights
By Modality Insights
Why did the Consumables & Accessories Segment Dominate the Lyophilization Equipment and Services Market in 2025?
The consumables and accessories segment dominated the market, accounting for the largest share in 2025. The high-turnover, recurring demand for essential components like vials and stoppers is required for every production cycle. This steady consumption is further amplified by the surge in biologics and vaccines, which require sterile, high-quality materials to ensure drug stability. Stricter regulatory standards and the rise of CDMO outsourcing have shifted the market toward single-use accessories to prevent contamination and manage large-scale operations. The equipment is a one-time purchase, and consumables provide the continuous revenue stream necessary to sustain the expanding global biopharma infrastructure.
- In January 2025, Akums Drugs & Pharmaceuticals Ltd. launched a new state-of-the-art sterile manufacturing facility dedicated to lyophilized products to meet rising global demand for stable, freeze-dried pharmaceuticals. The facility, which is now operational, offers expanded production capabilities for lyophilized injectables, vials, and other sterile products.
The services segment is expected to grow at the fastest CAGR during the projection period, driven by the need to bypass high capital costs while accessing specialized expertise for complex biologics and vaccines. Global demand for thermostable logistics and strict regulatory compliance has shifted the burden of sterile manufacturing to these high-capacity service providers. Furthermore, the expansion of food preservation and rapid industrial growth in the Asia-Pacific region has broadened the market beyond pharmaceuticals.
- In August 2025, Cytiva partnered with Argonaut Manufacturing Services in the EU and US to expand lyophilization and reagent stabilization services, specifically aimed at improving localized manufacturing and cost-efficiency for diagnostic developers.
By Scale of Operation Insights
How Does the Industry Scale Segment Lead the Lyophilization Equipment and Services Market in 2025?
The industry scale segment led the lyophilization equipment and services market in 2025 due to providing the massive capacity and high-throughput automation required for the global distribution of biologics and vaccines. By integrating sophisticated technology, these large-scale systems ensure product stability and batch-to-batch consistency while reducing long-term operating costs. The shift toward CDMO outsourcing further bolsters this segment, as service providers invest heavily in industrial-sized infrastructure to support multiple pharmaceutical clients.
The laboratory scale segment is projected to grow at the highest CAGR in the coming years. This is mainly because of the surge in biopharmaceutical research and development for complex molecular, such as mRNA vaccines, which have made laboratory-scale units indispensable for optimizing delicate formulations at the earliest stages. The industry's shift toward personalized medicine further drives demand for these high-precision, flexible benchtop units that can handle small, targeted batches more efficiently than industrial systems. Integration of AI-driven controls and automation has transformed these tools into sophisticated platforms that minimize human error and accelerate the development of heat-sensitive products.
By Application Insights
How Does the Pharmaceutical and Biotechnology Companies Segment Lead the Lyophilization Equipment and Services Market in 2025?
The pharmaceutical and biotechnology companies segment led the lyophilization equipment and services market in 2025 due to a surge in biologics and vaccine manufacturing, which has made lyophilization indispensable for maintaining the stability and efficacy of complex, heat-sensitive drugs. As pharmaceutical companies expand R&D and large-scale production, they increasingly rely on CDMO outsourcing to access specialized equipment without the burden of high capital investment. These services are further enhanced by technological advancements in automated, continuous freeze-drying, which improve process efficiency and reduce energy consumption.
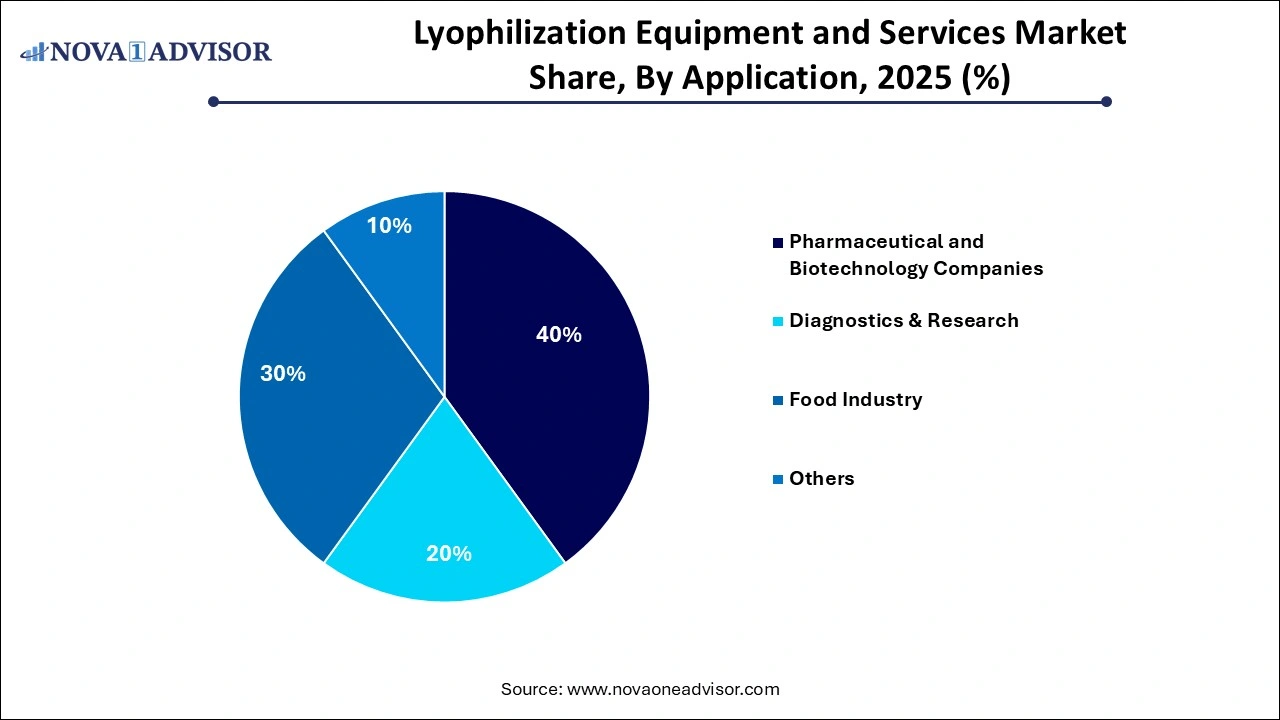
The food industry segment is projected to grow at the highest CAGR in the coming years. This is mainly because the shift toward premium nutrition and convenience has made lyophilization essential for preserving the original taste and nutrients of food without artificial additives. The rapid urbanization in emerging markets, where rising incomes fuel the consumption of high-quality instant meals and health snacks. Modern technological advancements in automation and energy efficiency have lowered operational costs, allowing producers to scale these high-end preservation methods profitably.
Lyophilization Equipment and Services Market Regional Insights
What Made North America the Dominant Region in the Lyophilization Equipment and Services Market?
North America maintained dominance in the lyophilization equipment and services market while holding the largest share in 2025. The region’s dominance is attributed to its status as a global hub for biopharmaceutical R&D, where the surge in mRNA vaccines and biologics necessitates advanced stabilization. The region benefits from a high concentration of specialized CDMOs and a robust food innovation sector, supported by strict regulatory oversight and advanced infrastructure. These factors ensure North America remains the primary leader in the global lyophilization landscape.
- In May 2024, Governor Hochul announced the groundbreaking of IMA Life North America’s $30 million, 80,000-square-foot expansion project in Tonawanda, New York, designed to increase production of pharmaceutical freeze-drying equipment. The new facility, supported by over $1.1 million in state tax credits, will create 70 new jobs and strengthen the production of equipment for partners like Pfizer and Moderna.
U.S. Lyophilization Equipment And Services Market Trends
The U.S. is a major contributor to the market in North America due to the rapid expansion of biologics and vaccines, and advanced lyophilization is essential for maintaining the stability of temperature-sensitive drugs. The high concentration of CDMOs is investing in large-scale, automated industrial units to reduce capital burdens for pharmaceutical firms. The technological upgrades, such as AI-driven predictive maintenance and energy-efficient microwave-assisted drying, which enhance process precision and speed-to-market.
What Makes Asia Pacific the Fastest-Growing Area in the Market?
Asia Pacific is expected to grow at the fastest rate in the coming years. This is due to the biopharmaceutical boom and the rapid expansion of regional manufacturing hubs in China and India. Strategic government initiatives, such as India’s PLI scheme and the launch of massive CDMO facilities, such as Akums, are significantly strengthening local production capacity for vaccines and biologics. The high adoption of IoT-enabled and energy-saving freeze-drying systems to manage rising operational costs.
China Lyophilization Equipment And Services Market Trends
China is a key player in the Asia Pacific lyophilization equipment and services market due to the shift toward CDMO outsourcing and the adoption of IoT-driven automation to ensure strict regulatory compliance and process efficiency. The strong push for energy-saving technologies and government-backed modernization of the food sector is meeting the rising consumer demand for premium, shelf-stable products. The region’s ability to blend high-tech innovation with cost-effective manufacturing is making it the primary engine for global lyophilization expansion.
How is the Opportunistic Rise of Europe in the Lyophilization Equipment and Services Market?
Europe is experiencing a strategic growth in the market due to the increasing need for stability in biologics and vaccines, with the pharmaceutical sector rising as a hub for high-quality, technically advanced, and energy-efficient lyophilization equipment. The increasing outsourcing to specialized services to reduce capital expenditure, with services witnessing the fastest growth within the region. Growing demand for advanced freeze-drying technology for premium, organic, and long-shelf-life products and high safety and quality standards favor sophisticated European-manufactured equipment, enhancing the region’s market position.
United Kingdom Lyophilization Equipment And Services Market Trends
The United Kingdom is leading the market in Europe due to the stability requirements of next-generation biologics and cell gene Therapy. To combat high energy costs and strict regulations, the industry is prioritizing automation, PAT-driven efficiency, and sustainable cooling technologies. This shift is further fueled by a heavy reliance on specialized CDMOs, allowing firms to scale production while maintaining the rigorous precision needed for modern medicine.
Lyophilization Equipment And Services Market Supply Chain Analysis
- R&D and Formulation Development: This stage involves determining the appropriate formulation (solvents, cryoprotectants) and cycle parameters for the product, such as freezing rates and vacuum levels, to ensure stability.
Key Players: Coriolis Pharma, Lyophilization Technology, Inc., ProJect Pharmaceutics, Vetter Pharma.
- Equipment Component Manufacturing: Specialized suppliers provide essential components for lyophilization equipment, such as vacuum systems, refrigeration units, sensors for Process Analytical Technology (PAT), and high-efficiency loading systems.
Key Players: SP Industries (SP Scientific), Labconco Corporation, Edwards Vacuum, Cuddon Freeze Dry, Zirbus Technology GmbH.
- Distribution and Post-Sales Support: This final stage involves delivering the sensitive equipment to end-users (pharma, food processing, research institutions) along with installation, validation, maintenance, and technical training services.
Key Players: SP Scientific, Labconco Corporation, Tofflon, GEA Group, Martin Christ.
Lyophilization Equipment and Services Market Companies
- Tofflon Science and Technology Co., Ltd.: Tofflon is the world’s largest manufacturer of pharmaceutical freeze dryers, providing high-capacity, cGMP-compliant systems for vaccines, biologics, and antibiotics.
- Martin Christ Freeze-Drying Systems GmbH: Martin Christ is a global technology leader who uniquely covers the entire equipment spectrum, ranging from small laboratory units to pilot systems and full-scale production lines.
- Azbil Corporation Operating through its subsidiary Azbil Telstar, the company specializes in high-end lyophilization equipment specifically designed for sensitive pharmaceuticals like antibody drugs and vaccines.
- SP Industries (SP Scientific): SP Scientific is a leading provider of highly specialized R&D and pilot-scale freeze dryers, often credited with setting the industry standard for cycle development and formulation testing.
- GEA Group: GEA is a global powerhouse that focuses on large-scale industrial lyophilization plants, offering fully integrated lines that include automated loading and unloading systems (ALUS).
Recent Developments
- In July 2024, GEA introduced sustainable, energy-efficient innovations in pharmaceutical freeze-drying, featuring the LYOAIR® natural refrigerant cooling system and atmospheric spray freeze-drying that reduces energy usage by roughly 50%. Further developments included microwave-based continuous processing and the LYOVAC® ECO Mode for real-time energy optimization. Read the full press release at GEA.
- In August 2025, Cytiva and Argonaut Manufacturing Services partnered to expand cGMP lyophilization and reagent stabilization services for diagnostic assay developers in the U.S. and Europe. The collaboration focuses on reducing cold-chain dependency for molecular diagnostics by providing localized, small-to-large-scale services.
Exclusive Analysis on the Lyophilization Equipment and Services Market
The global lyophilization equipment and services market is leveraged by the increasing demand for stabilization of sensitive biologics, vaccines, and injectables in the pharmaceutical industry. Segmental analysis indicates that the pharmaceutical and biotechnology segment dominates, fueled by the rising pipeline of complex molecules, monoclonal antibodies, and personalized medicine. Technological advancements, including automated, high-efficiency tray-style freeze-dryers and continuous freeze-drying systems, are enhancing process control, improving product quality, and shortening cycle times. The high capital investment requirements and technical expertise are boosting the adoption of outsourced lyophilization services (CDMOs), particularly for specialized and clinical-scale applications.
Segments Covered in the Report
By Modality
- Devices
- Tray Lyophilizers
- Shelf Lyophilizers
- Rotary Lyophilizers
- Others
- Consumables & Accessories
- Services
By Scale of Operation
- Laboratory Scale
- Pilot Scale
- Industry Scale
By Application
- Pharmaceutical and Biotechnology Companies
- Diagnostics & Research
- Food Industry
- Others
By Region
- North America
- U.S.
- Canada
- Mexico
- Rest of North America
- South America:
- Brazil
- Argentina
- Rest of South America
- Europe
- Western Europe
- Germany
- Italy
- France
- Netherlands
- Spain
- Portugal
- Belgium
- Ireland
- UK
- Iceland
- Switzerland
- Poland
- Rest of Western Europe
- Eastern Europe
- Austria
- Russia & Belarus
- Türkiye
- Albania
- Rest of Eastern Europe
- Asia Pacific:
- China
- Taiwan
- India
- Japan
- Australia and New Zealand,
- ASEAN Countries (Singapore, Malaysia)
- South Korea
- Rest of APAC
- MEA:
- GCC Countries
- Saudi Arabia
- United Arab Emirates (UAE)
- Qatar
- Kuwait
- Oman
- Bahrain
- South Africa
- Egypt
- Rest of MEA




