U.S. High Potency Active Pharmaceutical Ingredients Market Size and Growth
The U.S. high potency active pharmaceutical ingredients market size was valued at USD 32.65 billion in 2025 and is projected to surpass around USD 78.8 billion by 2035, registering a CAGR of 9.19% over the forecast period of 2026 to 2035.
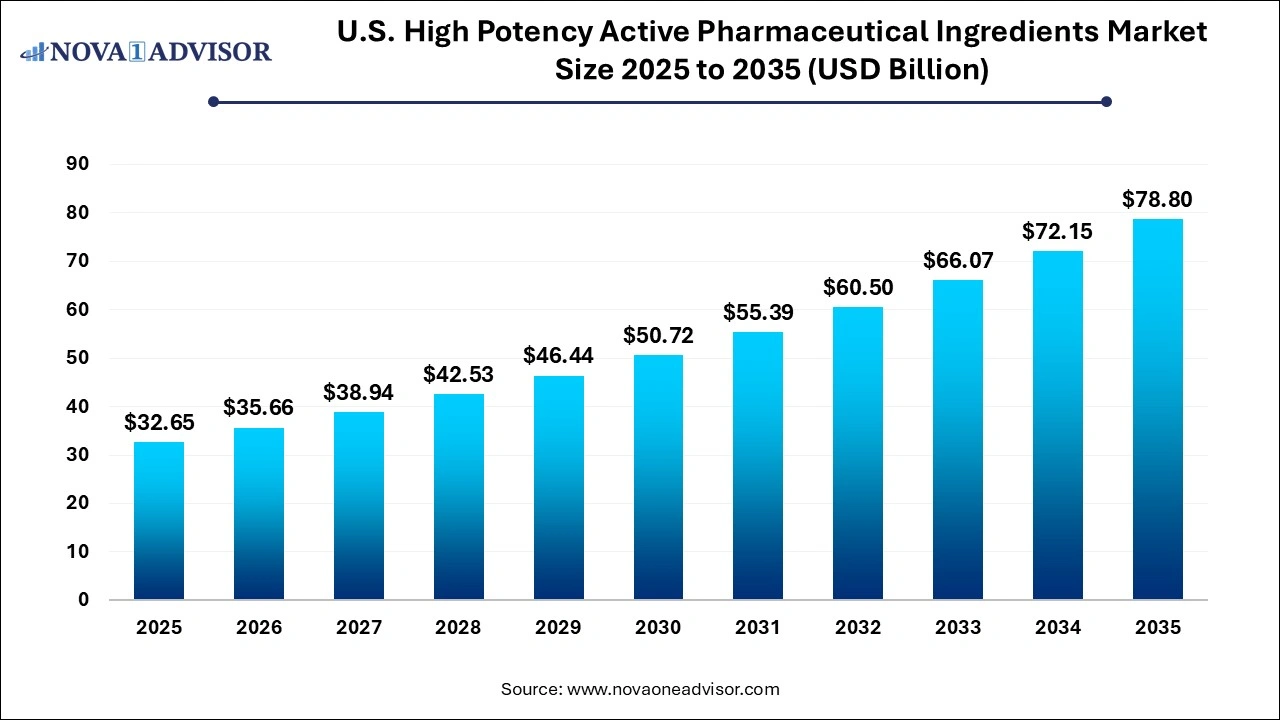
U.S. High Potency Active Pharmaceutical Ingredients Market Key Takeaways
- In the U.S. high potency active pharmaceutical ingredients market, the in-house segment predominates among manufacturers.
- The outsourced segment grows fastest in the U.S. high-potency active pharmaceutical ingredients market.
- In the U.S. high potency active pharmaceutical ingredients market, the oncology segment emerges as a dominant force, driving significant growth and innovation.
- The hormonal disorders segment fastest grow in the U.S. high-potency active pharmaceutical ingredient market.
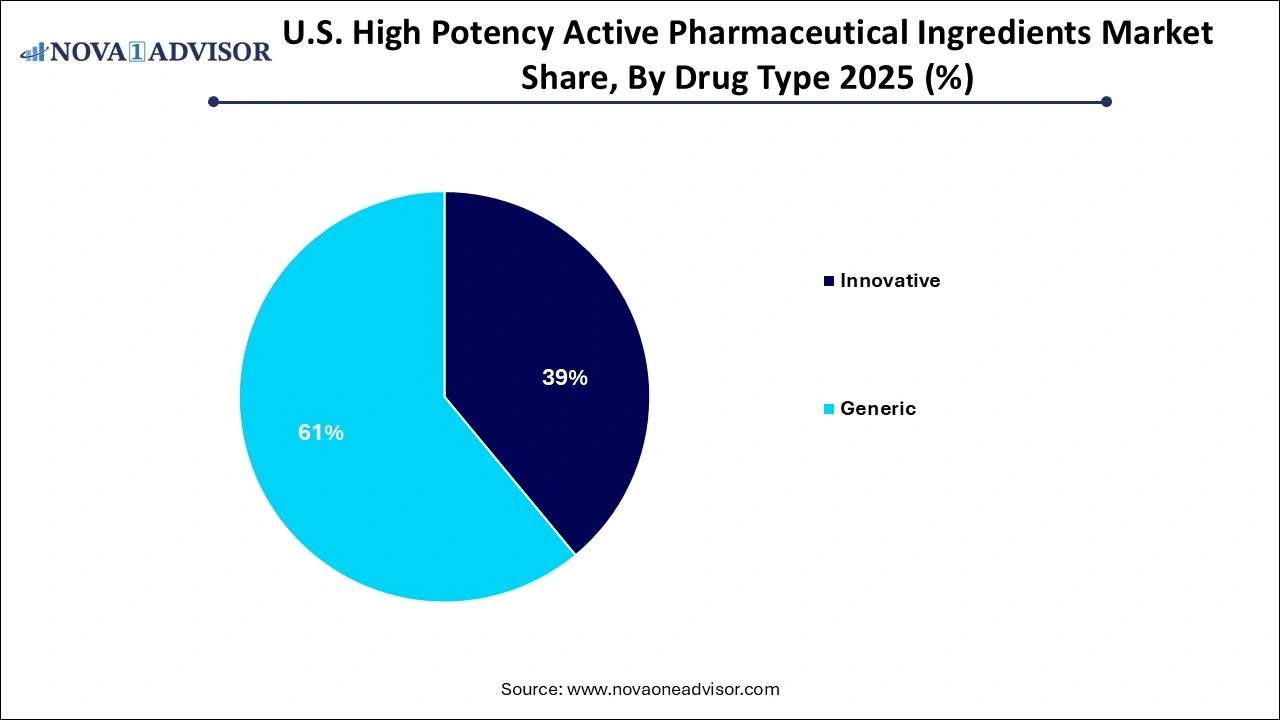
- In the U.S. high potency active pharmaceutical ingredients market, the innovative drugs segment holds dominance.
- The generic segment is the fastest-growing in the U.S. high potency active pharmaceutical ingredients market.
- In the U.S. high potency active pharmaceutical ingredients market, the synthetic segment emerges as the dominant force by product type.
U.S. High Potency Active Pharmaceutical Ingredients Market Outlook
- Market Growth Overview: The U.S. high potency active pharmaceutical ingredients market is expected to grow significantly between 2025 and 2034, driven by the increasing incidence of cancer and other chronic illnesses, improved technological advancements, and advanced therapies of precision medicine.
- Sustainability Trends: Sustainability trends involve the adoption of green chemistry, waste reduction, circularity, and environmental performance metrics.
- Major Investors: Major investors in the market include Pfizer, Novartis, Merck, Bristol-Myers Squibb, Sanofi, Lonza, CordenPharma, and CARBOGEN AMCIS.
Artificial Intelligence: The Next Growth Catalyst in U.S. High Potency Active Pharmaceutical Ingredients
AI is profoundly reshaping the U.S. high-potency active pharmaceutical ingredients industry by reducing the time and cost associated with complex drug discovery, development, and manufacturing. Machine learning algorithms and digital twins are enabling precise, AI-driven synthesis route optimization, which improves yield and purity while reducing safety risks and waste in the production of high-potency molecules. In the U.S., pharmaceutical companies are leveraging AI to simulate manufacturing scenarios, enabling continuous process verification and predictive maintenance of specialized, high-containment equipment.
Value Chain Analysis of the U.S. High Potency Active Pharmaceutical Ingredients Market
- R&D and Drug Discovery (Pre-clinical Phase: This stage involves identifying high-potency molecules for targeted therapies, particularly for oncology and autoimmune diseases, focusing on compounds that are effective at low dosages.
Key Players: Pfizer Inc., AbbVie Inc., Eli Lilly and Company, Roche, Bristol-Myers Squibb.
- Raw Material Sourcing and Intermediate Production: This stage involves sourcing complex, specialized chemical precursors and key raw materials required for synthetic or biotech HPAPI production.
Key Players: BASF Pharma Solutions, Lonza, Evonik Industries AG.
- Regulatory Approval and Quality Control: The manufacturing process must adhere to strict Current Good Manufacturing Practices (cGMP) and FDA regulations regarding cross-contamination, demanding precise validation and cleaning protocols.
Key Players: U.S. Food and Drug Administration (FDA), internal QA/QC teams at Lonza and Pfizer.
U.S. High Potency Active Pharmaceutical Ingredients Market Overview
The high potency active pharmaceutical ingredients (HPAPI) are more effective than conventional APIs at considerably lower dosage levels, but their handling issues are unique due to their potency. One significant reason anticipated to propel the market for highly potent active pharmaceutical ingredients is the increased incidence of cancer (HPAPIs). The WHO estimates that cancer is the second leading cause of mortality, accounting for nearly 9.6 million deaths in 2019, with tobacco use accounting for 22% of those deaths. The entire burden of the disease has been attributed, according to the CDC, to risk factors including lifestyle modifications like smoking, obesity, drinking alcohol, and exposure to UV radiation from the sun or tanning beds. Furthermore, it is predicted that the cost of cancer treatment will rise to USD 174 billion by the end of 2022, which will likely drive market expansion.
U.S. High Potency Active Pharmaceutical Ingredients Market Growth Factors
The U.S. High Potency Active Pharmaceutical Ingredients (HPAPI) market is experiencing significant growth driven by several key factors. Increasing prevalence of chronic diseases such as cancer, cardiovascular conditions, and autoimmune disorders is fueling the demand for potent medications that require HPAPIs. Additionally, advancements in biopharmaceutical technology and the growing trend of targeted therapy are boosting the development and adoption of HPAPIs. Stringent regulatory standards and the necessity for specialized manufacturing facilities are also propelling market expansion as pharmaceutical companies invest in state-of-the-art technologies to meet compliance and safety requirements. Furthermore, strategic collaborations and partnerships among key industry players are enhancing research and development capabilities, further driving innovation and growth in the HPAPI sector.
- Rising demand for drugs
- Increasing Focus on Precision Medicine and High-potency APIs
- Technological Advancements in High-potency API Manufacturing
- Increasing application of HPAPIs
- Rising prevalence of cancer
U.S. High Potency Active Pharmaceutical Ingredients Market Report Scope
| Report Attribute |
Details |
| Market Size in 2026 |
USD 35.66 Billion |
| Market Size by 2035 |
USD 78.8 Billion |
| Growth Rate From 2026 to 2035 |
CAGR of 9.21% |
| Base Year |
2025 |
| Forecast Period |
2026 to 2035 |
| Segments Covered |
By Product, By Manufacturer Type, By Drug Type, and By Application |
| Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
| Report Coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Key Companies Profiled |
BASF SE, CordenPharma, Dr. Reddy’s Laboratories Ltd., CARBOGEN AMCIS AG, Pfizer, Inc., Sun Pharmaceutical Industries, Ltd., Teva Pharmaceutical Industries Ltd., Albany Molecular Research, Inc., Sanofi S.A., Merck & Co., Inc., Novartis AG |
U.S. High Potency Active Pharmaceutical Ingredients Market Key Drivers
- Rising Demand for Oncology Drugs: The oncology segment is the largest and fastest-growing application area for HPAPIs, driven by the increasing prevalence of cancer. The development of targeted therapies, such as antibody-drug conjugates (ADCs), is also fuelling this demand .
- Increasing Clinical Trials and Approvals: There is a growing emphasis on precision medicine and the development of novel drugs. This includes significant R&D investments and clinical trials aimed at creating innovative treatments for various chronic and life-threatening diseases.
- Growth in Biologics and Biosimilars: The production of biologics and biosimilars requires high potency APIs due to their complex molecular structures, which further boosts market growth.
Segmental Insights
By Manufacturers Insights
How did the Outsourced segment grow fastest in the U.S. high-potency active pharmaceutical ingredients market?
The outsourced segment growth is driven by the substantial cost and technical complexity of in-house manufacturing, which pushes many drug developers to seek specialized external partners. This outsourcing model allows pharmaceutical companies to focus their resources on core competencies like drug discovery and clinical development, leveraging the established expertise of contract development and manufacturing organizations (CDMOs).
By Application Insights
How did the hormonal disorders segment fastest grow in the U.S. high-potency active pharmaceutical ingredient market?
The increasing prevalence of these conditions in aging populations and women, coupled with enhanced diagnostic capabilities. This demand is broad, covering diverse treatments for thyroid disorders, diabetes, and reproductive health, and these therapies often present a more affordable alternative to complex hormonal disorder treatments.
By Drug Type Insights
How did the generic segment is fastest growth in the U.S. high potency active pharmaceutical ingredients market?
The generic drug type segment is driven by the increasing healthcare costs and patient demand for cheaper treatments, particularly for chronic diseases. The FDA’s generic drug program and abbreviated new drug application process facilitate, and rising therapeutic demand fuel the market growth.
By Product Insights
How did the biotech segment experience the fastest growth in the U.S. high potency active pharmaceutical ingredients market?
The transition toward targeted therapies and personalized medicine, which demand highly complex and effective molecules, such as monoclonal antibodies and oligonucleotides. This growth is further propelled by the strategic focus on Antibody-Drug Conjugates (ADCs) and significant R&D investments, which offer superior clinical success rates for severe autoimmune and genetic conditions.
U.S. High Potency Active Pharmaceutical Ingredients Market Recent advancements
- In March 2024, Noramco Group Launched, Integrating Halo Pharma and Purisys , Noramco Group, a North American pharmaceutical supply chain services provider, has been unveiled, consolidating the strengths of subsidiaries Halo Pharma and Purisys. This integration aims to streamline logistics, enhance regulatory compliance, and bolster domestic production capabilities in the pharmaceutical industry.
- In Lupin shares surged over 3.39% as the US FDA greenlit the launched of Mirabegron extended-release (ER) tablets, 25 mg. This approval marks a significant milestone for the pharmaceutical major, opening avenues for the introduction of this medication in the United States market.
U.S. High Potency Active Pharmaceutical Ingredients Market Top Key Companies:
- BASF SE: BASF strengthens its position in the U.S. HPAPI market by providing specialized, high-quality ingredients and utilizing its new GMP Solution Center in Wyandotte, Michigan, for advanced drug formulation.
- CordenPharma: CordenPharma operates as a top-tier CDMO in the U.S. market, specializing in the development and manufacturing of complex HPAPIs, peptides, and lipids for targeted therapies.
- Dr. Reddy’s Laboratories Ltd.: Dr. Reddy’s contributes to the U.S. market by leveraging its expertise in producing complex, high-quality, and affordable generic HPAPIs for therapeutic areas like oncology and cardiology.
- CARBOGEN AMCIS AG: CARBOGEN AMCIS provides comprehensive CDMO services for HPAPIs, including specialized capabilities in Antibody Drug Conjugates (ADCs) and oncology, with high-containment manufacturing to handle potent materials safely.
- Pfizer, Inc.: Pfizer contributes to the HPAPI market by leveraging its extensive infrastructure to develop and manufacture advanced, high-potency drugs, particularly in the oncology and specialty sectors.
- Sun Pharmaceutical Industries, Ltd.: Sun Pharma is a major global player supplying high-potency APIs and formulations to the U.S. market, focusing on complex, targeted treatments for oncology and other critical diseases.
U.S. High Potency Active Pharmaceutical Ingredients Market Report Segmentation
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the U.S. High Potency Active Pharmaceutical Ingredients market.
By Product
By Manufacturer Type
By Drug Type
By Application
- Oncology
- Hormonal Disorders
- Glaucoma
- Others

